REVIEW: New Insight into the Role of AhR in Lung Carcinogenesis
Dinara A. Akhmetova1,a*, Vadim V. Kozlov2,3, and Ludmila F. Gulyaeva1,2
1Novosibirsk National Research State University, 630090 Novosibirsk, Russia2Research Institute of Molecular Biology and Biophysics, Federal Research Center for Fundamental and Translational Medicine, Siberian Branch of the Russian Academy of Sciences, 630117 Novosibirsk, Russia
3Department of Thoracic Oncology № 3, Novosibirsk Regional Clinical Oncology Center, 630108 Novosibirsk, Russia
* To whom correspondence should be addressed.
Received June 24, 2022; Revised September 6, 2022; Accepted September 13, 2022
Lung cancer (LC), one of the most common malignant neoplasms, is the leading cause of high cancer mortality worldwide. Smoking is a risk factor for almost all histological types of LC. Benzo[a]pyrene (BaP), one of the main constituents of tobacco smoke, can cause cancer. It has been established that its toxic effects can develop in the following ways: genotoxic (formation of adducts with DNA) and non-genotoxic or epigenetic. The latter is less known, although it is known that BaP activates aryl hydrocarbon receptor (AhR), which regulate transcription of many target genes, including microRNAs, which can lead to initiation and enhancement of the malignant cell transformation. Recent studies are evaluating the role of AhR in the regulation of immune checkpoints, as cigarette smoke and BaP induce the AhR-regulated expression of PD-L1 (CD274) in lung epithelial cells in vitro and in vivo. In addition, kynurenine (a metabolite of tryptophan) has been found to stimulate the PD-1 (CD279) expression in cytotoxic T cells by activating AhR. Recent studies confirm great importance of AhR expressed in malignant cells for suppression of antitumor immunity. All this makes us rethink the role of AhR in lung carcinogenesis and investigate the mechanisms of its activation by exogenous and endogenous ligands. This review highlights the current understanding of the functional features of AhR and its role in the LC pathogenesis.
KEY WORDS: lung cancer, AhR, cigarette smoke, benzo[a]pyrene, PD-1, PD-L1DOI: 10.1134/S0006297922110013
Abbreviations: AhR, aryl hydrocarbon receptor; ARNT, AhR nuclear translocator; BaP, benzo[a]pyrene; DREs, dioxin response elements; Kyn, kynurenine; LC, lung cancer; miR, microRNA; NSCLC, non-small cell LC; PAHs, polycyclic aromatic hydrocarbons; PD-1, programmed cell death 1; PD-L1, programmed cell death 1 ligand; TCDD, 2,3,7,8-tetrachlorodibenzo-n-dioxin.
INTRODUCTION
Lung cancer (LC) continues to be the leading cause of cancer deaths worldwide. According to the World Health Organization (WHO), 1,800,000 people died from LC in 2020 [1]. Smoking is a risk factor for almost all histological types of LC [2]. The components of tobacco smoke include polycyclic aromatic hydrocarbons (PAHs), nitrosamines, and radioactive elements that contribute to the emergence of lung cancer [3].
According to international agency for research on cancer (IARC), benzo[a]pyrene (BaP), part of the cigarette smoke, belongs to the group 1 human carcinogens, i.e., can cause cancer [4]. Toxic effects of BaP are realized via two main mechanisms: genotoxic (BaP oxidation leading to formation of the electrophilic metabolites that can bind to DNA) and non-genotoxic (binding to aryl-hydrocarbon receptor (AhR) followed by activation of the target genes). Typically, such genes have xenobiotic response elements (XREs), originally known as dioxin response elements (DREs) on their distal promoters.
The BaP-mediated activation of AhR leads to induction of the cytochrome P450 complex genes, namely CYP1A and CYP1B. In early research the role of AhR in LC carcinogenesis was focused on initiation of LC through activation of cytochrome P450 complex genes. Another proposed mechanism was the AhR-dependent DNA adduct formation. In addition, the role of AhR in the development of chronic inflammation was studied, which could contribute to the emergence of cancer [5].
It has shown that BaP, which is a component of cigarette smoke, in addition to the classical AhR target genes (CYP1A, CYP1B), induces expression of the immune checkpoints, which comprise a system of inhibitory mechanisms that regulate autoimmune processes. These include proteins such as PD-L1 (programmed cell death 1 ligand, CD274) and PD-1 (programmed cell death 1, CD279). PD-L1 is a ligand of the PD-1 receptor. Interaction of PD-L1 and PD-1 contributes to neutralization of T cells and inhibition of cytokine production [6].
At first, the connection between AhR and immune system was investigated through the 2,3,7,8-tetrachlorodibenzo-n-dioxin (TCDD)-dependent activation of AhR, which has a suppressive effect on animal and human T cells [7, 8]. It has recently been shown that kynurenine (Kyn), a metabolite of tryptophan, can also activate AhR and lead to PD-1 expression in the cytotoxic CD8+ T cells, which may contribute to the processes helping a tumor to evade immune system and to enhance its malignant phenotype [9]. Therefore, AhR activation facilitates survival of the transformed malignant cells by blocking anti-tumor immunity.
Much of the AhR-related research has gone far beyond toxicology. There is an increasing body of data showing epigenetic and immunological functions of the receptor. This review presents new data on the role of AhR in lung carcinogenesis, shows association of AhR with anti-tumor immune response, and describes the AhR-regulated microRNAs that could serve as potential biomarkers of LC.
CONTRIBUTION OF SMOKING TO THE DEVELOPMENT OF LUNG CANCER
Small cell and non-small cell LC are the most common histological types of LC. Small cell LC, which accounts for 15% of all cases of LC, has the most unfavorable prognosis. The 5-year survival rate is less than 5%, and for the untreated patients, the median survival is 2-4 months [10]. Non-small cell LC (NSCLC) accounts for 85% of all cases of LC. The 5-year survival rate is approximately 15%. NSCLC has three main subtypes depending on the cellular composition and localization of the oncological process: adenocarcinoma, squamous cell carcinoma, and large cell carcinoma [2].
One of the main risk factors for NSCLC is cigarette smoking [2]. Tar of the tobacco smoke contains such carcinogenic compounds as BaP, methylcholanthrene, tobacco-specific N-nitrosamines, N-methyl-N-nitrosourea, as well as metals (nickel, arsenic, lead, chromium) and radioactive elements (210polonium, 228thorium, 210lead) [3].
BaP is one of the most studied carcinogens in the tobacco smoke. The content of BaP in the smoke of one cigarette is 22.92-26.27 ng, according to the IARC [4]. BaP can bind to the AhR and activate it [11]. The result of such activation is translocation of AhR into the nucleus and induction of transcription of the AhR-dependent target genes, such as cytochrome P450 complex genes (CYP1A1, CYP1A2) and others. This interaction can lead to activation of carcinogens [12], formation of benzo[a]pyrene-7,8-diol-9,10-epoxide-N2-deoxyguanosine (BPDE-dG) in the human bronchial cells [13], and can also contribute to oxidative stress and inflammation [14].
In the case of reduced activity of enzymes in the second phase of xenobiotic metabolism, BaP epoxide metabolites could bind to DNA and form adducts [15], which, in turn, could lead to initiation of carcinogenesis in the lung tissue. Moreover, the BaP-dependent activation of AhR stimulates MAPK signaling, disruption of which leads to the induction of cellular processes such as proliferation, differentiation, and apoptosis [16].
Thus, substances in the cigarette smoke change metabolism of the lung cells and activate and/or disrupt transmission of the cell signals from receptors, which contributes to the development of malignant lung neoplasia.
STRUCTURE AND FUNCTIONS OF THE AhR
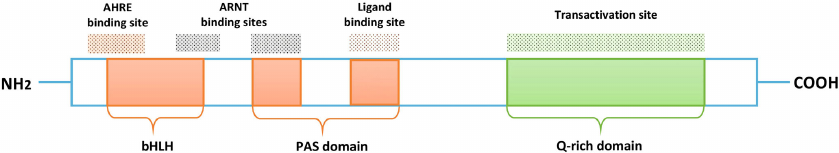
AhR is a member of the family of helix-loop-helix/Per-ARNT-Sim (PAS) transcription factors [17, 18] and consists of three functional parts: basic Helix-Loop-Helix (bHLH) domain, PAS domain (A and B), and Q-domain (Fig. 1).
Fig. 1. Functional structure of AhR. N-terminus of the receptor, which includes bHLH and PAS domains, provides heterodimerization of AhR with its nuclear translocator ARNT and promotes AhR binding to the promoter region of the target genes. bHLH mediates binding of AhR (at the AHRE binding site) and ARNT, followed by AhR binding to the promoter region. PAS domain is responsible for maintaining secondary interactions with ARNT and promotes formation of the AhR/ARNT heterodimer, moreover, the domain contains the AhR ligand binding site. Q-rich domain is responsible for transcription activation.
Both exogenous and endogenous ligands can initiate AhR activation. Exogenous ligands include PAHs, food phytochemical compounds, such as indole-3-carbinol, and indoles produced by microbiota [13]. Endogenous ligands include eicosanoids (such as lipoxin A4, bilirubin) and tryptophan metabolites, namely 6-formylindolo[3,2-b]carbazole and Kyn [19].
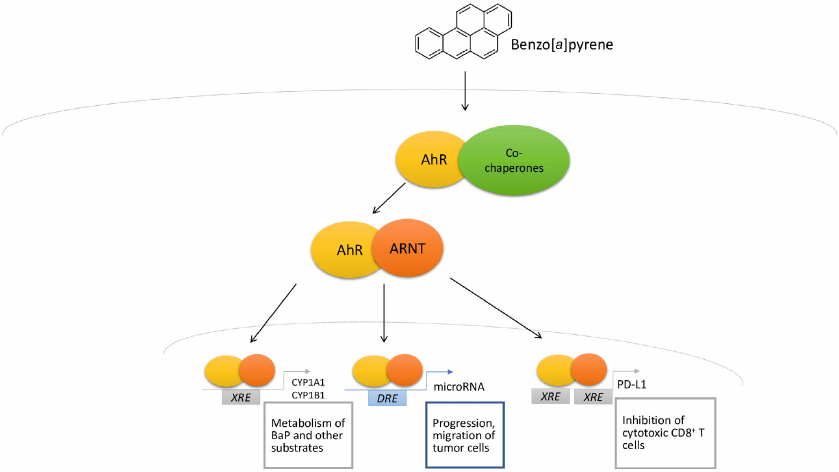
After binding to the ligand, the receptor translocates to the nucleus, where it heterodimerizes with AhR nuclear translocator (ARNT). The AhR/ARNT heterodimer is responsible for expression of many genes, including the cytochrome P450 complex genes (CYP1A1, CYP1A2, CYP1B1), TIPARP (gene coding TCDD Inducible Poly [ADP-Ribose] Polymerase) gene, as well as many others. Expression of the target genes is regulated through interaction with the canonical (5′-TNGCGTG-3′) and non-canonical receptor binding sites (Fig. 2). These binding sites are called XREs or DREs [17].
Fig. 2. Mechanism of BaP-mediated AhR activation.
The highest level of AhR expression is recorded in the human placenta, followed by the lungs, heart, pancreas, and liver. Whereas in the kidneys, brain, and skeletal muscles, it is expressed at low levels [20].
AhR is responsible for differentiation of T- and B-cell subpopulations, activates production of pro-inflammatory cytokines, regulates expression of transcription factors STAT3 and NF-kB, modulates response to microbial and viral pathogens, and is also involved in metabolism and bioactivation of xenobiotics [18]. AhR plays a significant role in pathogenesis of the chronic obstructive pulmonary disease (COPD) and asthma by regulating T cell differentiation. AhR increases mucin production in the NCI-H292 cell line in chronic bronchitis [5]. AhR has been shown to suppresses the cigarette smoke-induced acute lung inflammation [21]. Therefore, AhR controls immune, inflammatory, homeostatic, and toxic cell responses to endogenous and exogenous stimuli.
There is a controversial view on the role of AhR in initiation and progression of cancer. Some studies have shown that AhR could be a tumor suppressor under certain circumstances. For example, a certain dose of TCDD-dependent activation of AhR can suppress proliferation of cancer cells [22]. However, studies on the A549 human adenocarcinoma cell line showed that AhR activates DNA synthesis and subsequent cell proliferation of these cells, supposedly, through interaction with ARNT [23].
Global ChIP analysis of gene expression in the mouse liver revealed 14,446 and 974 regions enriched with AhR binding sites (false discovery rate 1%) at 2 and 24 h after TCDD injection, respectively [24].
In the human genome, 3915 AhR-binding sites have been identified [25]. Many of these binding sites require experimental validation to identify the AhR-regulated genes and elucidate their role in biological processes.
AhR performs many biological functions necessary for normal functioning of the body due to its structure and features of binding sites. Special attention is attracted to its role in carcinogenesis and LC development, elucidation of which requires understanding of molecular mechanisms of this process.
EXPRESSION OF THE AhR TARGET GENES IN LUNG TUMORS
AhR, known as a xenobiotic sensor, is activated by high-affinity low-molecular compounds and induces expression of its target genes. Exposure to PAH-containing substances primarily induces expression of the AhR-dependent enzymes of xenobiotic metabolism phases I and II in the body, such as CYP1A1, CYP1A2, CYP1B, glutathione S transferase, glucuronic transferase, NADPH, quinone oxidoreductase, aldo-keto reductase, and epoxide hydrolase [15].
CYP1A1 plays a significant role in cancerogenic activation through formation of highly reactive epoxides in the lungs by oxidizing PAHs [12]. Several studies show association of the polymorphic variants of the CYP1A1 gene, namely: allelic variant of CYP1A1*3 (T3205C) and CYP1A1*4 (Thr461Asn), with the increased activity of the enzyme, leading to increase in the formation of DNA-adducts and, as a result, to LC [12, 26]. A different variant of the same allele appears in the certain LC subtypes. For example, the CYP1A1*4 (Thr461Asn) allele is associated with the risk of developing small cell LC in the Spanish population [12, 27], and there is also evidence of association of the CYP1A1*2A (3801T→C) and CYP1A1*2C (2455A→G) alleles with NSCLC [26].
CYP1A2 can activate cigarette smoke procarcinogens. The CYP1A2*1D and CYP1A2*1F alleles are associated with the squamous cell LC [28]. There are different data regarding the allelic variant of CYP1A2 (163C→A). One study showed its association with adenocarcinoma in smokers [29], and in the 2013 meta-analysis the allelic variant of CYP1A2 (163C→A) was associated with the risk of squamous cell LC in Caucasians [30].
AhR can regulate expression of CYP1B1, which mediates bioactivation of tobacco procarcinogens and carcinogenic estrogens, facilitating their conversion into the reactive intermediate metabolites. These metabolites can cause damages in DNA by forming DNA-adducts. Changes in the CYP1B1 expression were observed in various histological types of NSCLC [31]. The allelic variant CYP1B1 Leu432Val is associated with the increased risk of LC in smokers [32].
EXPRESSION OF AhR-REGULATED microRNAs
AhR can regulate expression of microRNAs, small non-coding RNAs (~18-22 nt), dysregulation of which could lead to cancer [33]. AhR activation by berberine results in the increase of oncogenic miR-21-3p in the breast cancer cell line [34]. AhR controls basal expression of miR-196a in the mouse lung fibroblasts. Moreover, this receptor plays an important role in the regulation of microRNAs expressed in the lungs as was observed in the AhR gene knockout mice. AhR suppresses expression of the oncogenic miR-96, the level of which increases with chronic exposure to cigarette smoke [35].
Promoters of some intergenic microRNAs as well as their host genes contain AhR binding sites – DREs. Previously, it was believed that AhR performs some protective functions against the harmful effects of cigarette smoke, bypassing the classical binding to DREs [35]. A search for such elements was carried out in the genomes of rats, mice, and humans using in silico methods, and 154 genes with DREs were found in the human genome [36]. Therefore, the AhR-dependent regulation of microRNA expression could be mediated by binding to DREs. Further experimental studies are needed to confirm relevance of this mechanism. In particular, it was shown that the expression of miR-22, miR-29a, miR-126a, and miR-193b is significantly increased in the lungs of male rats, while the level of miR-483 is increased in the female rats during prolonged exposure to BaP. It has been suggested that the sex-dependent epigenetic effects of BaP could be based on the differential expression of microRNAs potentially regulated by AhR [37].
Changes in the microRNA levels in response to cigarette smoke can lead to progression of LC. AhR, in this case, could act as a negative regulator: for example, a reduced level of AhR leads to increase in the miR-96 expression. Chronic exposure to cigarette smoke in the AhR–/– mice has been shown to cause a dramatic increase in the level of miR-96 [35].
Decrease in the miR-335 levels as a response to the cigarette smoke-mediated increase in AhR levels was demonstrated [38]. Therefore, AhR can act as a “double-edged sword”, because the increase/decrease of AhR could contribute to deregulation of such important targets as microRNAs. At the same time, AhR can act as a repressor of some oncogenic microRNAs.
ROLE OF AhR IN FORMATION OF IMMUNOLOGICAL STATUS OF THE
TUMOR
Immunotherapy is used to treat LC, especially squamous cell LC. Immunotherapy is a type of cancer treatment that uses substances derived from living organisms. Monoclonal antibodies against cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), PD-1, and PD-L1 are used for LC treatment [39]. Such antibodies block inhibition of T cells, thereby contributing to antitumor immune response.
The immune checkpoint genes PD-1 and PD-L1 are important AhR target genes [6, 9, 39]. PD-1 is expressed directly in T- and B-cells and its ligand, PD-L1, is often expressed in neutrophils, epithelial, stromal cells, and tumors [39]. Interaction between PD-1 and PD-L1 leads to inhibition of the antitumor immune response and to increase in the tumor malignancy.
Recent studies have shown that AhR ligands can enhance expression of the immune checkpoint genes. There have been clinical observations that the smokers with NSCLC respond better to immunotherapy than the non-smokers [39]. These observations led to better understanding of the increase in the expression level of PD-L1 in the lung epithelial cells in vitro and in vivo upon exposure to cigarette smoke and BaP [6]. Immunotherapy based on the anti-PD-L1 monoclonal antibodies or AhR deficiency significantly suppressed the BaP-induced LC in mice. Further examination of the tumor specimens revealed that among 37 patients with NSCLC treated with anti-PD-1 therapy with pembrolizumab, 13/16 (81.3%) patients, who demonstrated partial response to the therapy or disease stabilization, had high AhR levels, whereas 12/16 (75%) patients with the progressing disease have low levels of AhR in the tumor tissues. AhR inhibitors have demonstrated significant antitumor activity and synergism with anti-PD-L1 antibodies in the mouse models of LC [6].
These results demonstrate that tobacco smoke allows lung epithelial cells to escape adaptive immunity promoting tumorigenesis, and the level of AhR predicts response to immunotherapy and is an attractive therapeutic target.
Another mechanism of AhR-mediated immunosuppression may be synthesizing of excess of Kyn, which is catalyzed by indoleamine 2,3-dioxygenase (IDO). Kyn can enhance the PD-L1 gene expression in macrophages, dendritic, and other immunocompetent cells, where AhR is active [40]. For example, AhR has recently been shown to be involved in the IFNγ-controlled expression of PD-L1 and IDO in the oral squamous carcinoma cells [9]. Moreover, distal promoter of the PD-L1 gene contains 2 XREs-like AhR binding elements, which promote activation of this gene expression by the AhR ligands [39].
Similar mechanisms were demonstrated for the autoimmune disease systemic lupus erythematosus (SLE). Recently, participation of PD-1 and PD-L1 in pathogenesis and development of SLE through AhR activation was demonstrated. In this case, kynurenine plays a significant role, its level increases in the serum of SLE patients. AhR is important for maintaining immune tolerance and development of various immune subpopulations, which emphasizes potential role of AhR in the PD-1 immunobiology [41].
Squamous cell LC is characterized by frequent increase in the level of PD-L1, which helps the tumor to avoid immune surveillance [42]. It was shown that the deficit of phosphatase and tensin homolog (PTEN) and/or p53 promotes the PD-L1 gene overexpression [6]. Activation of the PD-1 gene is stimulated in the tumor-repopulating cells (TRCs via intercellular Kyn exchange, which activates AhR [9].
INF-γ produced by CD8+ T cells stimulates release of high concentrations of this TRC-produced tryptophan metabolite, which is transported to the neighboring cytotoxic CD8+ T cells by the SLC7A8 and PAT4 transporters. Kyn induces and activates AhR and thereby increases the PD-1 gene expression. The Kyn-AhR pathway has been confirmed both in mice with tumors and in cancer patients, and its blockade can increase effectiveness of the antitumor-adoptive T cell therapy [9]. Therefore, such mechanism of the PD-1 gene activation may have potential immunotherapeutic applications.
Complex signaling interactions of the AhR–microRNA–PD-L1 axis should be noted, since microRNAs, especially the AhR-regulated microRNAs, are directly involved in regulation of the immune checkpoints and may contribute to the tumor growth and progression. Thus, the AhR-dependent oncogenic miR-193 and miR-93 target PD-L1 mRNA and regulate its level in the cell [37, 43]. Thus, disturbance of the level of expression of these microRNAs could lead to the development of cancer, including LC. Cigarette smoke causes a significant decrease in the content of many oncogenic microRNAs, as well as microRNAs that regulate the PD-L1 mRNA, which leads to initiation and further progression of LC [44].
Hence, AhR plays an important role in inhibition of anti-tumor immunity by regulating expression of PD-1 and PD-L1 genes. The immunosuppressive role of AhR in tumors may facilitate development of a new therapeutic strategy for the treatment of LC.
DISCUSSION
Scientific discoveries of the last decades have greatly enhanced our understanding of the role of AhR in lung carcinogenesis. Previously, this transcription factor was considered to play a key role at the initial stages of carcinogenesis through activation by the ligands of exogenous origin, such as BaP and other PAHs. This leads to increase in the expression of its target genes, primarily cytochrome P450, family 1. The result of such events is production of electrophilic PAH metabolites, which causes formation of DNA-adducts and emergence of mutations. Active investigation of AhR ligands has led to identification of new endogenous compounds, such as the tryptophan metabolite kynurenine, which has significantly expanded our understanding of the role of this receptor, especially in the immune response. The study of the expression of the AhR-regulated genes has expanded our knowledge about the role of AhR in physiological processes. The immune checkpoint genes PD-1 and PD-L1 are worth mentioning in this regard, which contain DREs and could be activated by AhR. Cigarette smoking, which leads to activation of this receptor, contributes to the “escape” of the tumor from the immune system. One of the therapeutic approaches in the treatment of lung cancer is immunotherapy. Understanding the role of AhR in shaping immunological status of the tumor will help improve cancer therapy and discover new promising treatments.
Contributions. D. A. Akhmetova, L. F. Gulyaeva – concept of the review article; D. A. Akhmetova, V. V. Kozlov, L. F. Gulyaeva – collection and analysis of literature, writing and editing the text of the review; D. A. Akhmetova – creation of illustrations.
Funding. This work was financially supported by the Russian Science Foundation (grant no. 22-15-00065, “Search for new targets for the diagnosis and therapy of squamous cell lung cancer”).
Ethics declarations. The authors declare no conflict of interest in financial or any other sphere. This article does not contain any studies with human participants or animals performed by any of the authors.
Open access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
REFERENCES
1.World Health Organization (WHO) (2020) Estimated
number of deaths in 2020, worldwide, both sexes, all ages (excl. NMSC),
Glob. Cancer Obs., 144.
2.Molina, J. R., Yang, P., Cassivi, S. D., Schild, S.
E., and Adjei, A. A. (2008) Non-small cell lung cancer: epidemiology,
risk factors, treatment, and survivorship, Mayo Clin. Proc.,
83, 584-594, doi: 10.4065/83.5.584.
3.Dyubkova T. P. (2008) The chemical composition of
tobacco smoke: toxic and carcinogenic effects on the human body [in
Russian], Med. Panorama, 38.
4.International Agency for Research on Cancer (IARC)
(2014) Chemical agents and related occupations, IARC Monogr.,
100, 423-428.
5.Tsay, J. J., Tchou-Wong, K. M., Greenberg, A. K.,
Pass, H., and Rom, W. N. (2013) Aryl hydrocarbon receptor and lung
cancer, Anticancer Res., 33, 1247-1256.
6.Wang, G. Z., Zhang, L., Zhao, X. C., Gao, S. H.,
Qu, L. W., et al. (2019) The Aryl hydrocarbon receptor mediates
tobacco-induced PD-L1 expression and is associated with response to
immunotherapy, Nat. Commun., 10, 1125, doi:
10.1038/S41467-019-08887-7.
7.Xue, P., Fu, J., and Zhou, Y. (2018) The aryl
hydrocarbon receptor and tumor immunity, Front. Immunol.,
3, 9-28, doi: 10.3389/fimmu.2018.00286.
8.Disner, G. R., Lopes-Ferreira, M., and Lima, C.
(2021) Where the aryl hydrocarbon receptor meets the microRNAs:
literature review of the last 10 years, Front. Mol. Biosci.,
8, 725044, doi: 10.3389/fmolb.2021.725044.
9.Liu, Y., Liang, X., Dong, W., Fang, Y., Lv, J., et
al. (2018) Tumor-repopulating cells induce PD-1 expression in
CD8+ T cells by transferring kynurenine and AhR activation,
Cancer Cell, 33, 480-494, doi:
10.1016/J.CCELL.2018.02.005.
10.Yang, S., Zhang, Z., and Wang, Q. (2019) Emerging
therapies for small cell lung cancer, J. Hematol. Oncol.,
12, 47, doi: 10.1186/S13045-019-0736-3.
11.Yoshikawa, Y., Izawa, T., Hamada, Y., Takenaga,
H., Wang, Z., et al. (2021) Roles for B[a]P and FICZ in subchondral
bone metabolism and experimental temporomandibular joint osteoarthritis
via the AhR/Cyp1a1 axis, Sci. Rep., 11, 14927,
doi: 10.1038/s41598-021-94470-4.
12.San Jose, C., Cabanillas, A., Benitez, J.,
Carrillo, J. A., Jimenez, M., et al. (2010) CYP1A1 gene
polymorphisms increase lung cancer risk in a high-incidence region of
Spain: a case control study, BMC Cancer, 10, 463, doi:
10.1186/1471-2407-10-463.
13.Alexandrov, K., Rojas, M., and Satarug, S. (2010)
The critical DNA damage by benzo(a)pyrene in lung tissues of smokers
and approaches to preventing its formation, Toxicol. Lett.,
198, 63-68, doi: 10.1016/j.toxlet.2010.04.009.
14.Hanyu, S., Jie, L., and Haiqing, G. (2021)
Benzo(α)pyrene induces oxidative stress and inflammation in human
vascular endothelial cells through AhR and NF-κB pathways,
Microvasc. Res., 137, 104179, doi:
10.1016/j.mvr.2021.104179.
15.Moorthy, B., Chu, C., and Carlin, D. J. (2015)
Polycyclic aromatic hydrocarbons: from metabolism to lung cancer,
Toxicol. Sci., 145, 5-15, doi: 10.1093/TOXSCI/KFV040.
16.Vázquez-Gómez, G., Rocha-Zavaleta,
L., Rodríguez-Sosa, M., Petrosyan, P., and Rubio-Lightbourn, J.
(2018) Benzo[a]pyrene activates an AhR/Src/ERK axis that
contributes to CYP1A1 induction and stable DNA adducts formation in
lung cells, Toxicol. Lett., 289, 54-62, doi:
10.1016/j.toxlet.2018.03.012.
17.Nebert, D. W. (2017) Aryl hydrocarbon receptor
(AHR): ‘pioneer member’ of the basic-helix/loop/helix
per-arnt-sim (bHLH/PAS) family of ‘sensors’ of foreign and
endogenous signals, Prog. Lipid Res., 67, 38-57, doi:
10.1016/J.PLIPRES.2017.06.001.
18.Shinde, R., and McGaha, T. L. (2018) The aryl
hydrocarbon receptor: connecting immunity to the microenvironment,
Trends Immunol., 39, 1005-1020, doi:
10.1016/j.it.2018.10.010.
19.Bock, K. W. (2019) Aryl hydrocarbon receptor
(AHR):from selected human target genes and crosstalk with transcription
factors to multiple AHR functions, Biochem. Pharmacol.,
168, 65-70, doi: 10.1016/J.BCP.2019.06.015.
20.Zhu, K., Meng, Q., Zhang, Z., Yi, T., He, Y.,
et al. (2019) Aryl hydrocarbon receptor pathway: role,
regulation and intervention in atherosclerosis therapy, Mol. Med.
Rep., 20, 4763, doi: 10.3892/MMR.2019.10748.
21.Thatcher, T. H., Maggirwar, S. B., Baglole, C.
J., Lakatos, H. F., Gasiewicz, T. A., et al. (2007) Aryl hydrocarbon
receptor-deficient mice develop heightened inflammatory responses to
cigarette smoke and endotoxin associated with rapid loss of the nuclear
factor-kappaB component RelB, Am. J. Pathol., 170,
855-864, doi: 10.2353/ajpath.2007.060391.
22.Wang, K., Li, Y., Jiang, Y. Z., Dai, C. F.,
Patankar, M. S., et al. (2013) An endogenous aryl hydrocarbon receptor
ligand inhibits proliferation and migration of human ovarian cancer
cells, Cancer Lett., 340, 63-71, doi:
10.1016/j.canlet.2013.06.026.
23.Shimba, S., Komiyama, K., Moro, I., and Tezuka,
M. (2002) Overexpression of the aryl hydrocarbon receptor (AhR)
accelerates the cell proliferation of A549 cells, J. Biochem.,
132, 795-802, doi: 10.1093/oxfordjournals.jbchem.a003289.
24.Dere, E., Lo, R., Celius, T., Matthews, J., and
Zacharewski, T. R. (2011) Integration of genome-wide computation DRE
search, AhR ChIP-chip and gene expression analyses of TCDD-elicited
responses in the mouse liver, BMC Genomics, 12, 365, doi:
10.1186/1471-2164-12-365.
25.Yang, S. Y., Ahmed, S., Satheesh, S. V., and
Matthews, J. (2018) Genome-wide mapping and analysis of aryl
hydrocarbon receptor (AHR) - and aryl hydrocarbon receptor repressor
(AHRR)-binding sites in human breast cancer cells, Arch.
Toxicol., 92, 225-240, doi: 10.1007/S00204-017-2022-X.
26.Wright, C. M., Larsen, J. E., Colosimo, M. L.,
Barr, J. J., Chen, L., et al. (2010) Genetic association study of
CYP1A1 polymorphisms identifies risk haplotypes in nonsmall cell lung
cancer, Eur. Respir. J., 1, 152-159, doi:
10.1183/09031936.00120808.
27.Schwarz, D., Kisselev, P., Cascorbi, I., Schunck,
W. H., and Roots, I. (2001) Differential metabolism of benzo[a]pyrene
and benzo[a]pyrene-7,8-dihydrodiol by human CYP1A1 variants,
Carcinogenesis, 22, 453-459, doi:
10.1093/carcin/22.3.453.
28.Singh, A. P., Pant, M. C., Ruwali, M., Shah, P.
P., Prasad, R., et al. (2010) Polymorphism in cytochrome P450
1A2 and their interaction with risk factors in determining risk of
squamous cell lung carcinoma in men, Cancer Biomark, 8,
351-359, doi: 10.3233/CBM-2011-0224.
29.B’chir, F., Pavanello, S., Knani, J.,
Boughattas, S., Arnaud, M. J., et al. (2009) CYP1A2 genetic
polymorphisms and adenocarcinoma lung cancer risk in the Tunisian
population, Life Sci., 84, 779-784, doi:
10.1016/j.lfs.2009.03.008.
30.Deng, S. Q., Zeng, X. T., Wang, Y., Ke, Q., and
Xu, Q. L. (2013) Meta-analysis of the CYP1A2-163C>A polymorphism and
lung cancer risk, Asian Pacific J. Cancer Prev., 14,
3155-3158, doi: 10.7314/APJCP.2013.14.5.3155.
31.Elfaki, I., Mir, R., Almutairi, F. M., and Abu
Duhier, F. M. (2018) Cytochrome P450: polymorphisms and roles in
cancer, diabetes and atherosclerosis, Asian Pac. J. Cancer
Prev., 19, 2057-2070, doi:
10.22034/APJCP.2018.19.8.2057.
32.Chen, P. F., He, X. F., Huang, G. H., Wang, W.,
and Qiu, Z. H. (2016) Association between the CYP1B1 polymorphisms and
lung cancer risk: a meta-analysis, Technol. Cancer Res. Treat.,
15, 73-82, doi: 10.1177/1533034615598866.
33.Lin, S., and Gregory, R. I. (2015) MicroRNA
biogenesis pathways in cancer, Nat. Rev. Cancer, 15,
321-333, doi: 10.1038/nrc3932.
34.Lo, S. N., Wang, C. W., Chen, Y. S., Huang, C.
C., Wu, T. S., et al. (2017) Berberine activates aryl hydrocarbon
receptor but suppresses CYP1A1 induction through miR-21-3p stimulation
in MCF-7 breast cancer cells, Molecules, 2017, 22, doi:
10.3390/molecules22111847.
35.Rogers, S., Souza, A. R., Zago, M., Iu, M.,
Guerrina, N., et al. (2017) Aryl hydrocarbon receptor (AhR)-dependent
regulation of pulmonary miRNA by chronic cigarette smoke exposure,
Sci Rep., 7, 40539, doi: 10.1038/srep40539.
36.Ovchinnikov, V. Y., Antonets, D. V., and
Gulyaeva, L. F. (2018) The search of CAR, AhR, ESRs binding sites in
promoters of intronic and intergenic microRNAs, J. Bioinform.
Comput. Biol., 16, 1-20, doi: 10.1142/S0219720017500299.
37.Filippov, S. V., Yarushkin, A. A., Yakovleva, A.
K., Kozlov, V. V., and Gulyaeva, L. F. (2020) The effect of
benzo[a]pyrene on the expression of AhR-regulated microRNA in
lungs of female and male rats, Biochemistry (Moscow)
Supplement Ser. B Biomedical Chemistry, 14, 347-355, doi:
10.1134/S199075082004006X.
38.Zhang, S., Kim, K. H., Jin, U. H., Pfent, C.,
Cao, H., et al. (2012) Aryl hydrocarbon receptor agonists induce
microRNA-335 expression and inhibit lung metastasis of estrogen
receptor negative breast cancer cells, Mol. Cancer Ther.,
11, 108-118, doi: 10.1158/1535-7163.MCT-11-0548.
39.Alsaab, H. O., Sau, S., Alzhrani, R., Tatiparti,
K., Bhise, K.,et al. (2017) PD-1 and PD-L1 checkpoint signaling
inhibition for cancer immunotherapy: mechanism, combinations, and
clinical outcome, Front. Pharmacol., 8, 561, doi:
10.3389/fphar.2017.00561.
40.Kenison, J. E., Wang, Z., Yang, K., Snyder, M.,
Quintana, F. J., et al. (2021) The aryl hydrocarbon receptor suppresses
immunity to oral squamous cell carcinoma through immune checkpoint
regulation, Proc. Natl. Acad. Sci. USA, 118, e2012692118,
doi: 10.1073/pnas.2012692118.
41.Curran, C. S., Gupta, S., Sanz, I., and Sharon,
E. (2019) PD-1 immunobiology in systemic lupus erythematosus,
J. Autoimmun., 97, 1-9, doi:
10.1016/j.jaut.2018.10.025.
42.Bylicki, O., Paleiron, N., Rousseau-Bussac, G.,
and Chouaïd, C. (2018) New PDL1 inhibitors for non-small cell lung
cancer: focus on pembrolizumab, OncoTargets Ther., 11,
4051-4064, doi: 10.2147/OTT.S154606.
43.Fan, J., Yin, Z., Xu, J., Wu, F., Huang, Q.,
et al. (2020) Circulating microRNAs predict the response to
anti-PD-1 therapy in non-small cell lung cancer, Genomics,
112, 2063-2071, doi: 10.1016/J.YGENO.2019.11.019.
44.Iqbal, M. A., Arora, S., Prakasam, G., Calin, G.
A., and Syed, M. A. (2019) MicroRNA in lung cancer: role, mechanisms,
pathways and therapeutic relevance, Mol. Aspects Med.,
70, 3-20, doi: 10.1016/J.MAM.2018.07.003.