MINI-REVIEW: A Window to the Potassium World. The Evidence of Potassium Energetics in the Mitochondria and Identity of the Mitochondrial ATP-Dependent K+ Channel
Dmitry B. Zorova
Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119992 Moscow, Russia
Received April 23, 2022; Revised July 4, 2022; Accepted July 4, 2022
The conclusions made in the three papers published in Function by Juhaszova et al. [Function, 3, 2022, zqab065, zqac001, zqac018], can be seen as a breakthrough in bioenergetics and mitochondrial medicine. For more than half a century, it has been believed that mitochondrial energetics is solely protonic and is based on the generation of electrochemical potential of hydrogen ions across the inner mitochondrial membrane upon oxidation of respiratory substrates, resulting in the generation of ATP via reverse transport of protons through the ATP synthase complex. Juhaszova et al. demonstrated that ATP synthase transfers not only protons, but also potassium ions, with the generation of ATP. This mechanism seems logical, given the fact that in eukaryotic cells, the concentration of potassium ions is several million times higher than the concentration of protons. The transport of K+ through the ATP synthase was enhanced by the activators of mitochondrial ATP-dependent K+ channel (mK/ATP), leading to the conclusion that ATP synthase is the material essence of mK/ATP. Beside ATP generation, the transport of osmotically active K+ to the mitochondrial matrix is accompanied by water entry to the matrix, leading to an increase in the matrix volume and activation of mitochondrial respiration with the corresponding increase in the ATP synthesis, which suggests an advantage of such transport for energy production. The driving force for K+ transport into the mitochondria is the membrane potential; an excess of K+ is exported from the matrix by the hypothetical K+/H+ exchangers. Inhibitory factor 1 (IF1) plays an important role in the activation of mK/ATP by increasing the chemo-mechanical efficiency of ATP synthase, which may be a positive factor in the protective anti-ischemic signaling.
KEY WORDS: mitochondria, ATP synthase, mitochondrial ATP-dependent potassium channel, potassium ions, protons, bioenergetics, transport, rotation, membrane potential, ischemiaDOI: 10.1134/S0006297922080016
INTRODUCTION
In the biological, and in particular, biochemical world, there is a dilemma of creating of two types of systems: one of them is based on the highest level of specificity, leading to a high selectivity of its components (e,g., hormone-receptor system), and the other is based on the overwhelming quantitative predominance (for example, non-selective buffering of ions and organic molecules by albumins. Note that although it is generally recognized that albumin is present in the extracellular environment, e.g., the bloodstream, due to the fact that it is synthesized on cellular ribosomes, albumin must remain inside the cell for at least some time in the immature form [1]). A combination of selective and non-selective strategies in the interaction of biological molecules has been discussed very superficially. But if we consider mitochondrial energetics, which after awarding two Nobel prizes (in 1978 and 1997), has been recognized exclusively as protonic, is seems reasonable to raise the following question. How can the system transporting protons, the concentration of which in the cytosol is ~10–7 M (pH 7.2-7.4), ignore the presence of another monovalent cation, potassium, whose cytosolic concentration is millions times higher ([K+] = 1.4 × 10–1 M). To be fair, we should mention that prokaryotes living in the environment with a high sodium content, comprise both the Na+ translocating pump of the electron transport chain [2, 3] and the ATP synthase utilizing the Na+ gradient [4]. Therefore, the proven cases of monovalent metal cation transport instead of a proton exist in the energetics of living organisms, which is quite logical, because transport systems are organized in such a way that a high selectivity for a proton could, in principle, be overcome by the quantitative predominance of another monovalent cation. Hence, we can assumed an existence of not only protonic, but also potassium mitochondrial energetics.
THE DISCOVERY OF POTASSIUM ENERGETICS
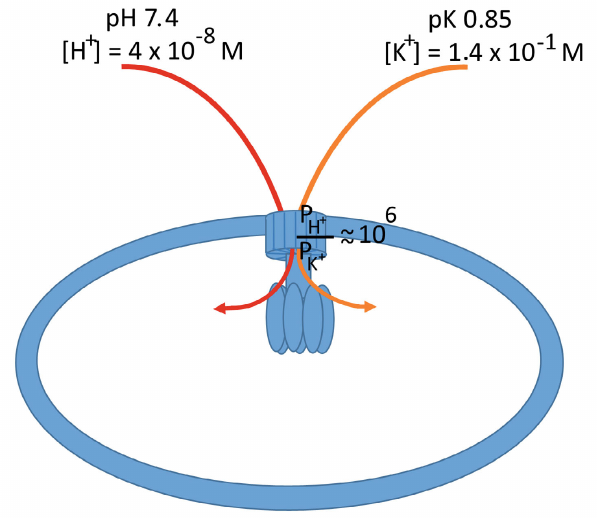
The basal permeability of the ATP synthase channel for protons and potassium ions (PH+ and PK+) was estimated as 5.2 ± 0.9 × 10–11 and 8.7 ± 2.9 × 10–17 m3/s, respectively. Addition of diazoxide, an activator of the mitochondrial ATP-dependent K+ channel (mK/ATP), increased these values approximately 3.5 times to 2.2 ± 1.3 × 10–10 and 3.0 ± 1.4 × 10–16 m3/s, respectively, thus maintaining the selectivity of the FO channel at ~106 : 1 with a strong preference for the proton transport over potassium ions (figure).
Schematic representation of the inner mitochondrial membrane with the ATP synthase complex. Protons and potassium ions are transported through the rotary part of the complex immersed in the membrane. The permeability of the complex differs significantly for the transported ions, with a strong preference for protons over potassium ions (~106 : 1). The intracellular K+ contents exceed the proton content more than 10 million times, which allows the K+ transport through the ATP synthase associated with the ATP synthesis. Moreover, due to the fact that potassium ions are osmotically active in comparison with protons, their transport to the mitochsondrial matrix is accompanied by the transport of water molecules (see the text), which activates respiration and promotes ATP synthesis
The question was raised and investigated in three consecutive papers published in Function [5-7], and the answer was that potassium energetics in the mitochondria does exist. It is based on the ability of mitochondrial ATP synthase for the potassium ion transport coupled with ATP synthesis. Moreover, such transport not only exists, but also prevails over proton transport – it was shown that per each transported H+, mitochondrial ATP synthase conducts 3.7 K+, and transfer of both these ions is associated with ATP synthesis. Basal potassium ion transport was detected upon reconstitution of mitochondrial ATP synthase in the liposomes (in the presence of potassium gradient), which increased after addition of diazoxide to the medium. This increase was blocked by venturicidin (ATP synthase proton channel inhibitor) and by the ATP-dependent K+ channel inhibitor 5-hydroxydecanoate. It means that, firstly, the ATP synthase complex is the material basis of mK/ATP and, secondly, it can transport potassium ions. Moreover, diazoxide equally enhanced the transport of K+ and H+ through the synthase, which was one of the facts confirming that protons and potassium ions use the same route in the ATP synthase complex.
Until recently, the nature of mK/ATP has been a subject of extensive search that have resulted in many discoveries and disappointments [8, 9]. The importance of identifying the nature of mK/ATP was undeniable, because activation of this channel can afford cardio- and neuroprotection [9-12], which makes mK/ATP activators potential drugs, in particular, the anti-ischemic agents.
One of the remarkable methodological aspects of the studies was an indirect proof of the rotary mechanism of the ATP synthase complex. Back in 1981, future Nobel laureate Paul Boyer wrote about a possible existence of the rotary component in this complex [13]. The hypothesis on the rotary mechanism of ATP synthase was independently proposed by Australian scientists in 1984 [14]. That time, it was assumed that the β-, γ-, δ-, and ε-subunits of the complex rotate, i.e., both parts of FO and some parts of F1 rotate. It was also suggested that rotation can be reversed and is triggered in a particular direction depending on whether the ATP synthase complex exhibits synthase or hydrolase activity. Probably, the necessity to include the β-subunit (part of F1) in the rotary complex was caused by the fact that the catalytic site is located in the β-subunit, which, as Boyer suggested, undergoes strong conformational rearrangements during transition from the state of tightly bound nucleotides to the state leading to the ATP release from the catalytic site [15]. Later, in 1986, the same Australian group modified their hypothesis by presenting a model according to which all proposed elements rotate in complete dependence on each other [16]. At the same time, in 1985, Nobel laureate Peter Mitchell also published a hypothesis, which he called a “rolling well” [17]. However, all these hypotheses were revised when the Walker’s group obtained a crystal of F1-ATPase [18] and then, based on the X-ray diffraction analysis, reconstituted the structure of the entire ATP synthase complex, including the rotor, whose mechanical energy is harnessed to release ATP from the catalytic site and the stator [19]. In 1996, the Junge’s laboratory demonstrated the physical rotation of the γ-subunit of the complex [20]. The rotary mechanism of the ATP synthase complex was finally proven by an impressive work from the Yoshida’s laboratory, who showed in real time a visually distinguishable spinning of the rotary fragment within the complex after attachment to the γ-subunit of fluorescently labeled biotinylated actin filament [21].
Juhaszova et al. used the following simple idea – if it can be proven that functioning of the ATP synthase machinery is coupled to the cation (proton, K+) transport, it is logical to assume that if the cation transfer (transmembrane current) takes place, it is coupled to the spinning of the rotary fragment (c-subunit ring and γ-subunit), and the direction of this rotation (clockwise or counterclockwise) depends on the direction of transmembrane current (ion transfer toward the matrix or toward the cytoplasm) [5-7]. And vice versa, in a system with the reconstituted ATP synthase complex, the direction of rotation of the rotor can be deduced from the direction of transmembrane current. That is exactly what was done by the authors: after applying pH or K+ gradient to the phospholipid bilayer membrane with the incorporated ATP synthase complex, they observed a current with typical characteristics of a current mediated by the ion channels. This channel activity was activated by diazoxide and suppressed by the ATP synthase inhibitor venturicidin and high ATP concentrations, i.e., demonstrated the properties of both mK/ATP and ATP synthase. Application of voltage of the opposite sign determined the characteristics of the current and indirectly indicated the direction of the rotor spinning, whereas varying the applied transmembrane potential and ATP concentration allowed to estimate the torque created by both factors under different conditions.
It should be admitted that quantitative assessment of ATP synthesis associated with potassium transport in the used model systems (liposomes or a single ATP synthase molecule) is extremely difficult; however, using isolated rat heart mitochondria, the authors obtained reasonable values of approximately 70 nmol phosphorylated ADP per minute per mg protein in a potassium-free medium and about 250 nmol phosphorylated ADP per minute per mg protein in a medium with potassium ions.
The gradient of K+ ions on the inner mitochondrial membrane is unnecessary for the K+ transport associated with the mitochondrial ATP synthesis, because the driving force of this transport is the presence of an electric field through the membrane. It is assumed that potassium ions entering the mitochondrial matrix are further used by the K+/H+ antiporter, which establishes the “status quo” for the K+ ions outside and inside the mitochondria. However, there is some advantage of potassium ion transport over proton transport, which lies in the fact that potassium ions, unlike protons, are osmotically active. Hence, accumulation of K+ in the matrix is accompanied by the water entry into the matrix, i.e., swelling of the mitochondria. It has been shown that a small (regulatory) swelling of mitochondria (not accompanied by a drop in the transmembrane potential) leads to the activation of mitochondrial respiration [12] and, accordingly, to an increase in the coupled ATP synthesis, which is important under increased physiological loads. Thus, the transport of potassium ions into the mitochondria via the FO ATP synthase channel is advantageous not only because of the associated ATP synthesis, but also because activation of the K+ channel provides better response to the increased loads via activation of mitochondrial respiration and additional ATP synthesis caused by a moderate (up to several percent) increase in the mitochondrial volume. As it has become clear, the efficiency of the ATP synthase complex is regulated by the protein inhibitor IF1 [22], which binds to the β- and γ-subunits and, according to the prevailing view, inhibits ATP hydrolysis (in particular, this process takes place in hypoxia, when the generation of membrane potential by the respiratory chain ceases and the membrane potential is maintained by the reversal of the ATP synthase reaction, i.e., by ATP hydrolysis [23]). The absence of IF1 in the mitochondria causes the loss of the ability of mK/ATP activators to enhance the ion transport through the mitochondrial ATP synthase [5-7], leading to the loss of the above-mentioned protective properties.
Moreover, electrophysiological experiments and bioinformatic analysis demonstrated that IF1 belongs to the group of BH3 domain proteins (which, for example, includes Bad). It was suggested that endogenous regulators of IF1 are proteins with a high affinity to the BH3 domain (such as Bcl-xL and Mcl), which ensures their binding to IF1, resulting in the release of the latter from the ATP synthase complex and abolishment of its inhibitory effect.
The idea that excessive potassium ions are pumped out of the mitochondria by the K+/H+ antiporter does not exclude an existence of other mechanisms. The data that the respiratory complex I in mammalian mitochondria can function as a Na+/H+ antiporter [24] are challenging and do not exclude the possibility of the monovalent ion (in particular, K+) transport in the mitochondrial respiratory chain.
It should be emphasized that while confirming the existence of potassium energetics, it was noted that the permeability of the ATP synthase channel for potassium ions is very close to that for sodium ions [5], which suggests that the ATP synthesis associated with the transport of sodium ions is possible if the content of sodium ions is million times higher than the concentration of protons. Normally, the concentration of sodium ions in a eukaryotic cell is several millimoles per liter, but it might significantly increase under certain pathological conditions. Moderate sodium overload due to the opening of sodium channels and weak activity of antiporters pumping Na+ out of the cell has been described for excitable tissues, such as heart and brain. For example, in the cytoplasm of granular neurons, an increase in the Na+ concentration to 60 mM and higher was detected after activation of NMDA or kainate receptors [25, 26]. Sharp increase in the intracellular sodium concentration was observed in the failed heart [27]. There are also recent indications of the presence of mitochondria in the bloodstream, where the content of sodium ions is similar to the intracellular concentration of potassium ions. All the above information allows to suggest a significant contribution of non-protonic, namely sodium, mitochondrial energetics, which can obviously become a subject of future research.
However, it is still impossible to consider changes in the intracellular content of any of these monovalent ions as exclusive conditions for the unidirectional switch of protonic energetics to the alternative one (potassium or sodium), if we take into account that, firstly, as we have already indicated, the permeabilities of the “proton” channel for potassium and sodium ions are very close, and, secondly, the total concentration of potassium and sodium ions is presumably constant, i.e. [K]i + [Na]I ≈ const. Therefore, the non-protonic, i.e., potassium or sodium mitochondrial energetics will be a permanent value making the same contribution to the gross mitochondrial energetics, regardless of the ratio of these ions, which may change, in particular, when the resting potential on the cell membrane changes. It should be noted that without taking into account the role of specific regulators, the proton energetics, in theory, should also be more or less constant, considering fairly stable intracellular pH. Such direct logic does not require to provide a scheme of what will happen to the cell if it transports only potassium ions through the ATP synthase complex. However, this logic requires verification, given that sodium and potassium ions have different effects on the mitochondrial ATPase activity [28]. It is also necessary to take into account the regulatory function of the ATP synthase protein inhibitor during transition to the potassium energetics [5, 6], which requires further research to construct a general scheme of participation of sodium and potassium ions in the energy production and transduction.
CONCLUSIONS
The importance of the works of Juhaszova et al. is determined by the following. Firstly, they significantly contributed to the basic paradigms of bioenergetics. Secondly, the obtained data can be used in pharmacology to reformat the strategy of fighting against ischemic diseases due to the discovery of the nature of mK/ATP, which, as it became clear, is another function of mitochondrial ATP synthase. Since modulators of the ATPase activity are well developed, we can expect an emergence of new anti-ischemic agents based on these compounds, which opens new horizons for the therapy of heart attacks and strokes.
On the other hand, although this discovery represents a significant breakthrough in the fundamental biology and medicine, a number of problems remain unresolved related to the functioning of ATP synthase. First of all, it is unclear which cellular components play a role of endogenous regulators of this enzyme, which has deserved such remarkable epithets, as the “lord of the (c-) ring” [29], “marvelous” [30], “splendid” [31], and “wind-up toy” [32].
Funding. The work was supported by the Russian Science Foundation (project no. 19-14-00173-p).
Ethics declarations. The author declares no conflicts of interest. This article does not describe any studies involving humans or animals.
REFERENCES
1.Judah, J. D., and Nicholls, M. R. (1971) The
separation of intracellular serum albumin from rat liver, Biochem.
J., 123, 643-648, doi: 10.1016/j.bbabio.2009.12.020.
2.Verkhovsky, M. I., and Bogachev, A. V. (2010)
Sodium-translocating NADH:quinone oxidoreductase as a redox-driven ion
pump, Biochim. Biophys. Acta, 1797, 738-746, doi:
10.1016/j.bbabio.2009.12.020.
3.Muntyan, M. S., Cherepanov, D. A., Malinen, A. M.,
Bloch, D. A., Sorokin, D. Y., et al. (2015) Cytochrome cbb3 of
Thioalkalivibrio is a Na+-pumping cytochrome oxidase,
Proc. Natl. Acad. Sci. USA, 112, 7695-7700, doi:
10.1073/pnas.1417071112.
4.Dimroth, P., von Ballmoos, C., Meier, T., and Kaim,
G. (2003) Electrical power fuels rotary ATP synthase, Structure,
11, 1469-1473, doi: 10.1016/j.str.2003.11.011.
5.Juhaszova, M., Kobrinsky, E., Zorov, D. B., Yaniv,
Y., Fishbein, K. W., et al. (2022) ATP synthase K+- and
H+-flux drive ATP synthesis and enable mitochondrial
K+-uniporter function. I. Characterization of ion fluxes,
Function, 3, zqab065, doi: 10.1093/function/zqab065.
6.Juhaszova, M., Kobrinsky, E., Zorov, D. B., Yaniv,
Y., Fishbein, K. W., et al. (2022) ATP synthase K+- and
H+-flux drive ATP synthesis and enable mitochondrial
K+-uniporter function. II. Ion and ATP synthase flux
regulation, Function, 3, zqac001, doi:
10.1093/function/zqac001.
7.Juhaszova, M., Kobrinsky, E., Zorov, D. B., Aon, M.
A., Cortassa, S., et al. (2022) Setting the record straight: a new
twist on the chemiosmotic mechanism of oxidative phosphorylation,
Function, 3, zqac018, doi:
10.1093/function/zqac018.
8.Bajgar, R., Seetharaman, S., Kowaltowski, A. J.,
Garlid, K. D., and Paucek, P. (2001) Identification and properties of a
novel intracellular (mitochondrial) ATP-sensitive potassium channel in
brain, J. Biol. Chem., 1276, 33369-33374, doi:
10.1074/jbc.M103320200.
9.Foster, D. B., Ho, A. S., Rucker, J., Garlid, A.
O., Chen, L., et al. (2012) Mitochondrial ROMK channel is a
molecular component of mitoK(ATP), Circ. Res., 111,
446-454, doi: 10.1161/CIRCRESAHA.112.266445.
10.Wind, T., Prehn, J. H., Peruche, B., and
Krieglstein, J. (1997) Activation of ATP-sensitive potassium channels
decreases neuronal injury caused by chemical hypoxia, Brain
Res., 751, 295-299, doi: 10.1016/s0006-8993(96)01419-9.
11.Garlid, K. D., Paucek, P., Yarov-Yarovoy, V.,
Murray, H. N., Darbenzio, R. B., et al. (1997) Cardioprotective
effect of diazoxide and its interaction with mitochondrial
ATP-sensitive K+ channels. Possible mechanism of
cardioprotection, Circ. Res., 81, 1072-1082, doi:
10.1161/01.res.81.6.1072.
12.Juhaszova, M., Zorov, D. B., Kim, S. H., Pepe,
S., Fu, Q., et al. (2004) Glycogen synthase kinase-3beta
mediates convergence of protection signaling to inhibit the
mitochondrial permeability transition pore, J. Clin. Invest.,
113, 1535-1549, doi: 10.1172/JCI19906.
13.Boyer, P., and Kohlbrenner, W. E. (1981) The
present status of the binding-change mechanism and its relation to ATP
formation by chloroplasts, in Energy coupling in Photosynthesis,
Selman, Selman-Reimer, pp. 231-240.
14.Cox, G. B., Jans, D. A., Fimmel, A. L., Gibson,
F., and Hatch, L. (1984) Hypothesis. The mechanism of ATP synthase.
Conformational change by rotation of the beta-subunit, Biochim.
Biophys. Acta, 768, 201-208, doi:
10.1016/0304-4173(84)90016-8.
15.Boyer, P. D., Cross, R. L., and Momsen, W. (1973)
A new concept for energy coupling in oxidative phosphorylation based on
a molecular explanation of the oxygen exchange reactions, Proc.
Natl. Acad. Sci. USA, 70, 2837-2839, doi:
10.1073/pnas.70.10.2837.
16.Cox, G. B., Fimmel, A. L., Gibson, F., and Hatch,
L. (1986) The mechanism of ATP synthase: a reassessment of the
functions of the b and a subunits, Biochim. Biophys. Acta,
849, 62-69, doi: 10.1016/0005-2728(86)90096-4.
17.Mitchell, P. (1985) Molecular mechanics of
protonmotive F0F1 ATPases. Rolling well and turnstile hypothesis,
FEBS Lett., 182, 1-7, doi:
10.1016/0014-5793(85)81142-x.
18.Abrahams, J. P., Leslie, A. G., Lutter, R., and
Walker, J. E. (1994) Structure at 2.8 Å resolution of F1-ATPase
from bovine heart mitochondria, Nature, 370, 621-628,
doi: 10.1038/370621a0.
19.Walker, J. E. (2013) The ATP synthase: the
understood, the uncertain and the unknown, Biochem. Soc. Trans.,
41, 1-16, doi: 10.1042/BST20110773.
20.Sabbert, D., Engelbrecht, S., and Junge, W.
(1996) Intersubunit rotation in active F-ATPase, Nature,
381, 623-625, doi: 10.1038/381623a0.
21.Noji, H., Yasuda, R., Yoshida, M., and Kinosita,
K. Jr. (1997) Direct observation of the rotation of F1-ATPase,
Nature, 386, 299-302, doi: 10.1038/386299a0.
22.Pullman, M. E., and Monroy, G. C. (1963) A
naturally occurring inhibitor of mitochondrial adenosine
triphosphatase, J. Biol. Chem., 238, 3762-3769.
23.Di Lisa, F., Blank, P. S., Colonna, R., Gambassi,
G., Silverman, H. S., et al. (1995) Mitochondrial membrane
potential in single living adult rat cardiac myocytes exposed to anoxia
or metabolic inhibition, J. Physiol., 486, 1-13, doi:
10.1113/jphysiol.1995.sp020786.
24.Roberts, P. G., and Hirst, J. (2012) The deactive
form of respiratory complex I from mammalian mitochondria is a
Na+/H+ antiporter, J. Biol. Chem.,
287, 34743-34751, doi: 10.1074/jbc.M112.384560.
25.Kiedrowski, L., Brooker, G., Costa, E., and
Wroblewski, J. T. (1994) Glutamate impairs neuronal calcium extrusion
while reducing sodium gradient, Neuron, 12, 295-300, doi:
10.1016/0896-6273(94)90272-0.
26.Kiedrowski, L., Wroblewski, J. T., and Costa, E.
(1994) Intracellular sodium concentration in cultured cerebellar
granule cells challenged with glutamate, Mol. Pharmacol.,
45, 1050-1054.
27.Liu, T., and O’Rourke, B. (2008) Enhancing
mitochondrial Ca2+ uptake in myocytes from failing hearts
restores energy supply and demand matching, Circ. Res.,
103, 279-288, doi: 10.1161/CIRCRESAHA.108.175919.
28.Beattie, D. S., and Basford, R. E. (1968)
Sodium-stimulated adenosine triphosphatase activity of rat brain
mitochondria, J. Neurochem., 15, 325-353, doi:
10.1111/j.1471-4159.1968.tb11617.x.
29.Nesci, S., Trombetti, F., Ventrella, V., and
Pagliarani, A. (2016) The c-ring of the F1FO-ATP
synthase: facts and perspectives, J. Membr. Biol., 249,
11-21, doi: 10.1007/s00232-015-9860-3.
30.Yoshia, M., Muneyuki, E., and Hisabori, T. (2001)
ATP synthase – a marvellous rotary engine of the cell, Nat.
Rev. Mol. Cell. Biol., 2, 669-677, doi:
10.1038/35089509.
31.Boyer, P. D. (1997) The ATP synthase-a splendid
molecular machine, Annu. Rev. Biochem., 66, 717-749, doi:
10.1146/annurev.biochem.66.1.717.
32.Berry, R. M. (2005) ATP synthesis: the
world’s smallest wind-up toy, Curr. Biol., 15,
R385-R387.