Recovery of Impaired Memory: Expression of c-Fos and Egr-1 Transcription Factors during Restoration of Damaged Engram in the Chick Brain
A. A. Tiunova1, D. V. Bezriadnov1, N. V. Komissarova1, and K. V. Anokhin1,2,3,a,b*
1P. K. Anokhin Research Institute of Normal Physiology, 125315 Moscow, Russia2National Research Center “Kurchatov Institute”, 123182 Moscow, Russia
3Lomonosov Moscow State University, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received May 21, 2018; Revision received May 31, 2018
Impairment of protein synthesis in the brain during learning prevents memory consolidation and results in amnesia, which until recently has been regarded irreversible. However, in some cases impaired memory could be restored by various “reminder” stimuli. The present study is based on the hypothesis that even in behaviorally profound amnesia, some disintegrated fragments of the engram are preserved in the brain and could be re-integrated into the whole system by specific types of stimuli. The aim of the present study was to test this hypothesis in an experimental model of pharmacologically induced memory impairment in young chicks and to reveal the brain areas involved in this process by mapping of reminder-induced expression of transcriptional factors c-Fos and Egr-1. We show that reminder treatment results in the recovery of memory impaired by protein synthesis inhibition during learning and induces c-Fos and Egr-1 expression in the brain regions involved in learning in this behavioral model. The patterns of c-Fos and Egr-1 induced expression in animals with impaired memory differed from the patterns of animals with unimpaired memory and as well as naïve animals with no memory. Thus, analysis of activity-induced c-Fos and Egr-1 expression revealed the brain regions that were specifically activated by the reminder treatment. At the behavioral level, this treatment led to memory recovery. Altogether, these results suggest that the reminder-induced transcriptional activity in the brain of amnestic animals occurs in regions maintaining the engram fragments that reintegrate to recover the impaired memory.
KEY WORDS: memory, amnesia, brain, engram, gene expression, c-Fos, Egr-1DOI: 10.1134/S0006297918090134
Abbreviations: AI, arcopallium intermedium; ANI, anisomycin; HD, hyperpallium densocellulare; Hpc, hippocampus; IMM, intermedial medial mesopallium; MA, methyl anthranilate; MSt, medial striatum; PS, physiological saline.
Understanding the nature of memory is largely based on the data from
studies on memory impairment. In particular, animal models of
pharmacologically induced memory impairment enabled elucidation of the
critical role of gene expression and of new proteins’ synthesis
in the brain for long-term memory formation [1, 2]. The process, mediated by this synthesis, was
designated as synaptic or molecular memory consolidation [3, 4]. It has been traditionally
thought that administration of protein synthesis inhibitors during
training leads to irreversible amnesia [5].
However, alongside with this point of view, there are experimental
data, indicative of the possibility to restore the impaired memory
using various “reminder” procedures [6,
7]. Therefore, at present, there are at least two
alternative hypotheses about the nature of amnesia, experimentally
caused by protein synthesis disruption. According to the first one,
there is no molecular memory consolidation during protein synthesis
blockade, and, consequently, the observed amnesia reflects absence of
the “memory trace”, the engram [8]. The
second hypothesis suggests that in case of such amnesia the engram by
itself remains intact but is not manifested in behavior due to impaired
access to it and/or impaired retrieval [9]. The
facts of restoration of the impaired memory are considered as an
argument in favor of this hypothesis, although there are objections to
this point of view [10].
The present study relies on the third hypothesis suggesting that the brain with impaired memory still contains fragments of the engram, dissociated by impairment, which are unable to be actualized in their current form as an integral memory and to manifest itself in behavior. However, these fragments can be reintegrated into a complete system and the memory can be restored if the reintegration process is initiated by certain stimulation of the remaining engram fragments [11].
Our previous studies of memory impairment in passive avoidance training in chicks and in fear conditioning in mice have shown that memory, disrupted by the blockade of various molecular consolidation mechanisms during training, can indeed be restored by a behavioral reminder. The whole recovery process takes at least 6 h and is protein synthesis-dependent [7, 11]. Based on these data, we suggested that the reminder should induce a reintegrative process, accompanied by gene expression activation in the brain areas maintaining the preserved engram fragments. The aim of this study was to test this hypothesis and to identify such areas in the chick brain by investigation of the reminder-induced expression of the c-Fos and Egr-1 transcription factors (zif268, NGFI-A, ZENK), known for their role in memory consolidation [12, 13].
MATERIALS AND METHODS
Behavior and injections. One-day-old chicks (Gallus gallus domesticus) were trained in a standard one-trial passive avoidance model, based on the innate propensity of chicks to peck new objects within their field of view during the first days of life and to remember their characteristics [13]. Training consisted of presenting twice a neutral (dry) bead to the animals, and then – of a bead of different color, moistened with a substance, aversive for birds, methyl anthranilate (MA; Sigma, USA). After chicks peck an aversive bead, they demonstrate a species-specific aversive behavior (head shaking, eyes closed, wiping the beak). As a result of training, chicks remember the properties of the bead and avoid it upon subsequent presentations [13].
Injections of a protein synthesis inhibitor to chicks. Injections were performed with a micro-syringe, using a head-fixing holder, which directs injections into the region of lateral cerebral ventricles, in a volume of 5 μl per hemisphere [14]. The protein synthesis inhibitor anisomycin (ANI; Sigma) was administered 5 min prior to training in a dose of 80 μg (ANI group). Control animals received physiological saline (PS) (Control group). The reminder procedure was carried out 24 h after training. For this, a part of chicks from the the ANI group was presented with a bead of a different color, also moistened with MA, and allowed to peck it (ANI + Reminder group). The remaining part of animals from the ANI group and animals of the Control group received no reminder. All animals were tested 48 h after training by presenting to them an “aversive” bead and then a neutral bead for 10 s each. During testing, both beads were dry. If the chick did not peck a bead for 10 s, its behavior was classified as avoidance; only those animals that did not avoid the neutral bead, were included in subsequent analysis. Selective avoidance of the “aversive” bead was regarded as presence of long-term memory. Proportions of animals in each group that avoided the “aversive” bead during testing, were compared; differences between the groups were statistically assessed with χ2 test.
Expression of c-Fos and Egr-1 transcription factors. Transcriptional activity in the brain after reminder procedure was assessed in a separate experiment. Animals were divided into following groups (6 animals per group): 1) passive control; 2) normal memory reactivation (reminder treatment after training on the background of PS administration); 3) impaired memory reactivation (reminder treatment after training on the background of ANI administration); 4) new memory formation (“reminder treatment” without prior experience, i.e. training with a new aversive object, a bead of another color, moistened with MA). Animals of experimental groups were decapitated 90 min after the reminder procedure (groups 2, 3, 4); animals of the passive control group (1) were taken from home cages, without any preliminary exposures. Immunohistochemical detection of c-Fos and Egr-1 proteins was carried out on brain cryostat sections (20 μm) with rabbit primary antibodies (Santa Cruz, USA) and horse secondary anti-rabbit antibodies (ImmPress KIT; Vector Labs, USA). Visualization was carried out with diaminobenzidine (Sigma), sections were digitized using Olympus BX50 microscope and TurboScan system (Objective Imaging Ltd, UK). Areas of interest were determined in accordance with the stereotaxic atlas of chick brain [15].
Quantitative analysis of c-Fos and Egr-1 expression was performed using the Image Pro Plus 3.0 program (Media Cybernetics, USA). The density of c-Fos and Egr-1 expression was calculated as a ratio of the number of diaminobenzidine-labeled cells in the selected region to the region area (in mm2). Data were statistically processed using Statistica 6.0 program (Kruskal–Wallis and Mann–Whitney tests).
RESULTS
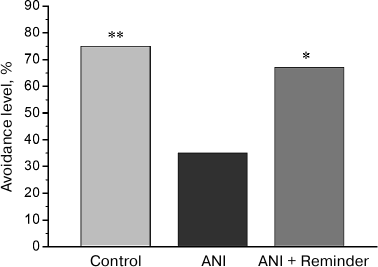
Memory testing in chicks. In the test 48 h after training, avoidance level in the ANI group, trained on the background of protein synthesis blockade, was significantly lower than in the control group, trained on the background of PS administration (Fig. 1). In the group receiving reminder treatment 24 h after training (ANI + Reminder group), avoidance level during testing was significantly higher than in the ANI group and did not differ from that in the control group. Thus, the reminder procedure restored the memory, impaired by protein synthesis blockade during training. This result is in line with the previously obtained data [7, 8, 11, 12] and supports the hypothesis of residual memory trace preservation in the brain upon amnesia.
Fig. 1. Effects of anisomycin and reminder procedure on the avoidance level 48 h after training. The Control group (n = 32) was trained on the background of intracranial saline injections and did not receive reminder treatment; the ANI group (n = 31) was trained after anisomycin administration and did not receive reminder treatment; the ANI + Reminder group (n = 33) was trained after anisomycin administration and received reminder treatment 24 h after the training; *p < 0.05, **p < 0.01, compared to the ANI group.
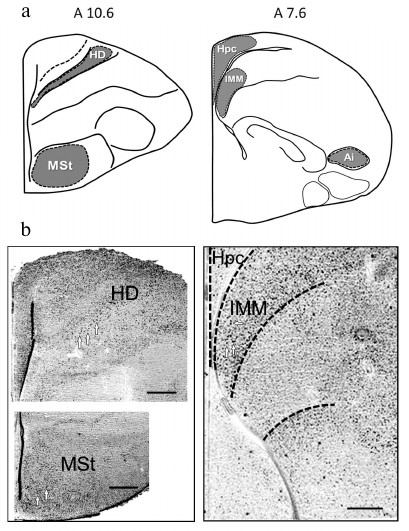
Expression of transcription factors c-Fos and Egr-1. Expression of c-Fos and Egr-1 was analyzed in the brain regions, involved in memory formation in passive avoidance training: intermediate medial mesopallium (IMM), medial striatum (MSt), intermediate arcopallium (AI), hippocampus (Hpc) and densocellular hyperpallium (HD) – the highest structure of the thalamofugal visual projection [13, 16-20]. Brain structures and regions, used for quantitative expression analysis, are shown in Fig. 2.
Fig. 2. a) Brain areas for quantitative analysis of c-Fos and Egr-1 expression (levels A10.6 and A7.6 are defined by the chicken brain atlas [15]); b) microphotographs illustrating c-Fos expression (white arrows designate the labelled cells). Scale, 0.25 mm. HD, densocellular hyperpallium; MSt, medial striatum; Hpc, hippocampus; IMM, intermediate medial mesopallium; AI, intermediate arcopallium.
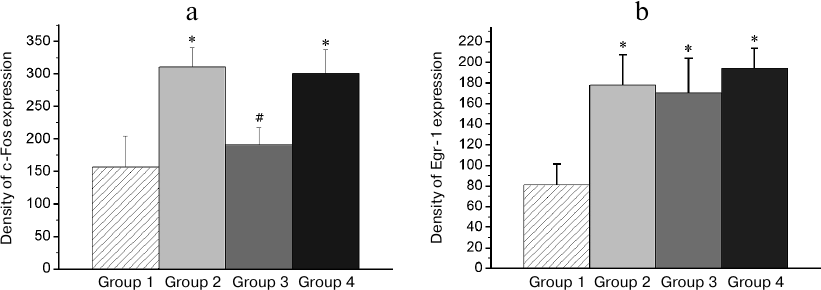
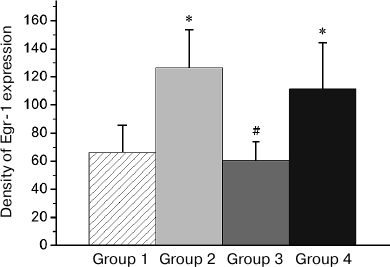
Patterns of transcriptional activity induced by the reminder procedure in the brain of animals with the normal memory (Group 2) differed from those in animals with the impaired memory (Group 3). So, in the HD, MSt, Hpc, and AI regions, the minimum expression level was observed in the passive control group (Group 1; Figs. 3-6). Reactivation of the normal memory (Group 2) caused a significant induction of both transcription factors in these regions compared to the basal expression level in Group 1 (Figs. 3-6). At the same time, reactivation of the impaired memory did not increase significantly the c-Fos expression in HD and MSt compared to the basal level (Group 3; Figs. 3a and 4a) as well as the Egr-1 expression in MSt (Group 3; Fig. 4b). In the hippocampus, the induction of the Egr-1 expression was also detected in case of reactivation of normal but not impaired memory (Groups 2 and 3; Fig. 5). Induction of the c-Fos in this structure was not observed in any of the groups (data not shown).
Fig. 3. Effect of reminder treatment on the c-Fos (a) and Egr-1 (b) expression in the densocellular hyperpallium (HD). Density of c-Fos- and Egr-1-positive cells/mm2 is shown as mean ± SE. Group 1, passive control; Group 2, normal memory reactivation; Group 3, impaired memory reactivation; Group 4, new memory formation (6 animals in each group); *p < 0.05 compared to Group 1; #p < 0.05 compared to Group 2.
Fig. 4. Effect of reminder treatment on the c-Fos (a) and Egr-1 (b) expression in the medial striatum (MSt). Density of c-Fos- and Egr-1-positive cells/mm2 is shown as mean ± SE. Group 1, passive control; Group 2, normal memory reactivation; Group 3, impaired memory reactivation; Group 4, new memory formation (6 animals in each group); *p < 0.05 compared to Group 1; #p < 0.05 compared to Group 2.
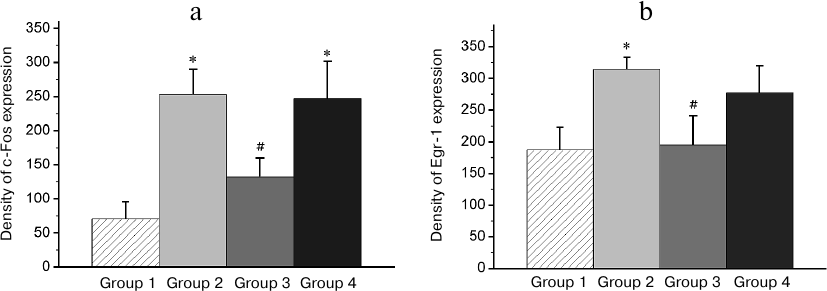
Fig. 5. Effect of reminder treatment on the Egr-1 expression in the hippocampus (Hpc). Density of Egr-1-positive cells/mm2 is shown as mean ± SE. Group 1, passive control; Group 2, normal memory reactivation; Group 3, impaired memory reactivation; Group 4, new memory formation (6 animals in each group); *p < 0.05 compared to Group 1; #p < 0.05 compared to Group 2.
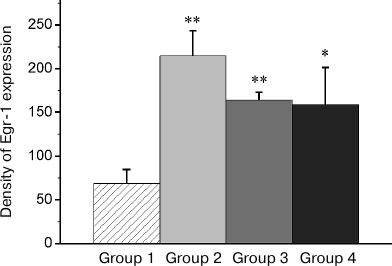
Fig. 6. Effect of reminder on Egr-1 expression in the intermediate arcopallium (AI). Density of Egr-1-positive cells/mm2 is shown as mean ± SE. Group 1, passive control; Group 2, normal memory reactivation; Group 3, impaired memory reactivation; Group 4, new memory formation (6 animals in each group); *p < 0.05, **p < 0.01 compared to Group 1.
Thus, in animals with amnesia, in contrast to animals with normal memory, the reminder procedure did not induce transcriptional activity in a number of brain regions, which seems to reflect the absence of a memory trace, to which the reminder was addressed. On the other hand, patterns of the reminder treatment-induced transcriptional activity in amnestic animals differed from those in naive (not trained previously) animals, which formed a new memory as a result of this procedure (Group 4). Thus, the level of the induced c-Fos expression in HD and MSt as well as Egr-1 expression in MSt and hippocampus in naive animals was significantly elevated compared to baseline and did not differ from that in animals with normal memory (Group 4; Figs. 3a, 4 and 5).
In regions HD and AI, the Egr-1 expression in all the three groups (Groups 2-4) significantly exceeded the basal level (Figs. 3b and 6). In these regions, the reactivation of both normal and impaired memory caused an increase in the Egr-1 expression. The Egr-1 level in Group 4, lacking any prior experience, was also significantly higher than baseline and did not differ from the expression level in the groups with reactivation of normal or impaired memory (Figs. 3b and 6). These data, indicative of involvement of HD and AI in reactivation of the impaired memory in animals with amnesia, suggest that these regions can support long-term plastic changes associated with previous training.
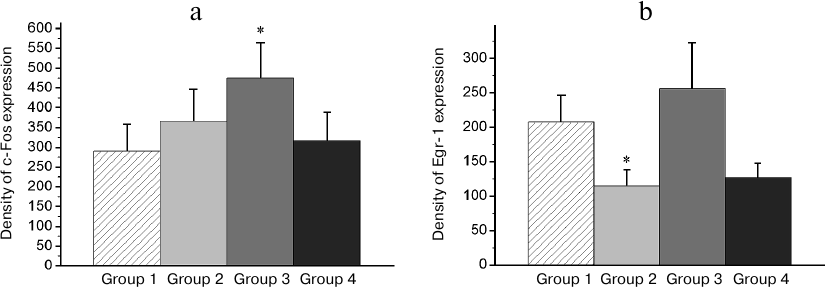
In IMM region, a high basal expression of both transcription factors was observed (Group 1; Fig. 7). The reminder treatment in the group with normal memory (Group 2) as well as training of naïve chicks (Group 4) did not alter the c-Fos expression level (Fig. 7a) and decreased the Egr-1 expression (Fig. 7b). At the same time, in amnestic animals, the induction of the c-Fos expression was observed in this area, but the Egr-1 expression was not changed (Figs. 7a and 7b).
Fig. 7. Effect of the reminder procedure on the c-Fos (a) and Egr-1 (b) expression in the intermediate medial mesopallium (IMM). Density of the c-Fos- and Egr-1-positive cells/mm2 is shown as mean ± SE. Group 1, passive control; Group 2, normal memory reactivation; Group 3, impaired memory reactivation; Group 4, new memory formation (6 animals in each group); *p < 0.05 compared to Group 1.
DISCUSSION
We demonstrated that administration of the protein synthesis inhibitor anisomycin prior to passive avoidance training disrupts long-term memory formation in chicks, which agrees with the existing data [13]. Previously, it has been shown that the memory disrupted by anisomycin can be restored by a reminder treatment [7, 11]. Anisomycin not only inhibits protein synthesis but also affects other processes in neural cells, for example, by altering neurotransmitter release [21] and by suppressing neuronal activity [22]. However, using passive avoidance training in chicks it was demonstrated that in the case of intracranial anisomycin administration, memory impairment is manifested only 40-60 min after the training or the reminder procedure, while the short-term protein-independent memory is not impaired [13]. But, if anisomycin caused amnesia through the altered neurotransmitter release or the neuronal activity suppression, the short-term memory would inevitably be disrupted. In addition, other protein synthesis inhibitors cause disruption of the long-term but not short-term memory that is identical to the anisomycin-induced amnesia, without the described side effects [13]. Collectively, these data support the assumption that it is the protein synthesis blockade that causes the anisomycin-induced amnesia.
The main objective of this study was to investigate expression of the c-Fos and Egr-1 transcription factors (zif268, NGFI-A, ZENK), known for their role in the experience-dependent plasticity in avian brain [12, 13, 23, 24], in the reminder-induced restoration of pharmacologically disrupted memory in chicks. In general, our results show that presentation of a reminder stimulus induces expression of transcription factors c-Fos and Egr-1 in the brain of animals with impaired memory; at that patterns of the expression differ from those in animals with normal memory and in previously untrained animals. The activation patterns of both transcription factors in the studied brain regions coincided in animals trained for the first time and in animals with normal memory, which was reactivated by a reminder treatment. Such a coincidence of patterns indicates that these brain regions seem to participate both in long-term memory formation and in its reactivation and/or reconsolidation. The difference in the expression patterns between the amnestic (Group 3) and previously untrained animals (Group 4), on the other hand, is indicative of preservation in the animals with impaired memory of some long-term, learning-induced changes in the brain, i.e. the fragments of the incomplete engram. In particular, in the two investigated regions, AI and HD, the level of the induced Egr-1 expression was similarly high in all groups (Figs. 3b and 6), which may reflect participation of these brain areas in maintaining the fragments of the impaired memory engram.
Area AI (intermediate arcopallium) is a part of the arcopallio-amygdaloid complex of avian forebrain and belongs to its sensorimotor section [25]. AI sends descending projections to the sensory, somatosensory and motor regions of telencephalon, to the thalamus and brain stem and performs premotor functions, thus forming the motor response program [26]. Lesion of the complex leads to incapability of passive avoidance learning [27]. Involvement of arcopallium in this training is confirmed by morphological data, indicating that it can serve as a relay area between IMM and MSt, the two key structures involved in learning and maintaining memory in passive avoidance model [15, 16]. Totally, all these data suggest that the residual engram fragments can be stored in AI in amnesia.
HD (densocellular hyperpallium) is a part of the visual Wulst – the highest projection of one of the two parallel visual pathways in avian brain. HD receives inputs from the thalamus nuclei and sends afferents to other Wulst components and to dorso-caudal pallial regions, including AI [17, 27]. Projections of HD to the pyriform cortex, associated with smell sense, have been shown in pigeons [28]. In chicks, HD has projections to IMM and plays a key role in visual imprinting [29]. The reminder-induced expression of transcription factor Egr-1 in amnestic animals can be associated with reactivation of the dissociated memory trace fragments. At the same time, it cannot be ruled out that this induced expression, similar in all groups, reflects activity of the systems associated with perception of the reminder stimulus, its visual, olfactory and other properties.
Thus, analysis of the c-Fos and Egr-1 expression in the brain of animals with impaired memory revealed areas, specifically activated upon the reminder treatment (HD, AI, IMM). Applying to the animals in reminder treatment the same aversive stimulus that was used during primary training results in restoration of the impaired memory at the behavioral level. The results obtained suggest that the specific transcriptional activity upon reminder procedure in amnestic animals occurs in brain structures supporting the fragments of the dissociated engram, which, as a result of the reminder procedure, are reintegrated into the complete engram – a process which provides memory recovery.
Funding
The study was supported by the Russian Foundation for Basic Research grant No. 16-04-01848.
Conflict of Interests
Authors declare no conflict of interests neither in financial nor in any other area.
Ethical Approval
All procedures conducted in this study with participation of laboratory animals were in accordance with the ethical standards of the P. K. Anokhin Research Institute of Normal Physiology (Rules for Using Animals in Experiments, Protocol no. 1, September 3, 2005) and legal requirements adopted by the Russian Federation and international organizations
(Directive 2010/63/EU of the European Parliament and of the Council of the European Union issued September 22, 2010, on the protection of animals, used for scientific purposes (Section 27)).
REFERENCES
1.McGaugh, J. L. (2000) Memory – a century of
consolidation, Science, 287, 248-251.
2.Kandel, E. R. (2001) The molecular biology of
memory storage: a dialogue between genes and synapses, Science,
294, 1030-1038.
3.Dudai, Y. (2004) The neurobiology of
consolidations, or, how stable is the engram? Annu. Rev.
Psychol., 55, 51-86.
4.Anokhin, K. V. (2005) Memory consolidation:
narrowing the gap between systems and molecular genetics neurosciences,
in Complex Brain Functions: Conceptual Advances in Russian
Neuroscience (Miller, R., Ivanitsky, A. M., and Balaban, P. M.,
eds.), CRC Press, Amsterdam, pp. 51-71.
5.Sweatt, J. D. (2016) Neural plasticity and behavior
– sixty years of conceptual advances, J. Neurochem.,
139, 179-199.
6.Mactutus, C. F., Ferek, J. M., and Riccio, D. C.
(1980) Amnesia induced by hyperthermia: an unusually profound, yet
reversible, memory loss, Behav. Neural Biol., 30,
260-277.
7.Radyushkin, K. A., and Anokhin, K. V. (1999)
Recovery of memory in chicks after disruption during learning: the
reversibility of amnesia induced by protein synthesis inhibitors,
Neurosci. Behav. Physiol., 29, 31-36.
8.Hardt, O., Wang, S. H., and Nader, K. (2009)
Storage or retrieval deficit: the yin and yang of amnesia, Learn.
Mem., 16, 224-230.
9.Squire, L. R. (2006) Lost forever or temporarily
misplaced? The long debate about the nature of memory impairment,
Learn. Mem., 13, 522-529.
10.Nader, K., and Wang, S. H. (2006) Fading in,
Learn. Mem., 13, 530-535.
11.Amelchenko, E. M., Zvorykina, S. W., Bezriadnov,
D. V., Chekhov, S. A., and Anokhin, K. V. (2013) Possibility of memory
reparation: conditioned freezing disrupted by protein synthesis
inhibition can be restored by reminder stimulation in mice, Zh.
Vyssh. Nerv. Deyat. im. I. P. Pavlova, 63, 227-234.
12.Clayton, D. F. (2013) The genomics of memory and
learning in songbirds, Annu. Rev. Genom. Hum. Genet., 14,
45-65.
13.Rose, S. P. (2000) God’s organism? The
chick as a model system for memory studies, Learn. Mem.,
7, 1-17.
14.Davis, J. L., Masuoka, D. T., Gerbrandt, L. K.,
and Cherkin, A. (1979) Autoradiographic distribution of L-proline in
chicks after intracerebral injection, Physiol. Behav.,
22, 693-695.
15.Kuenzel, W. J., and Masson, M. (1988) A
Stereotaxic Atlas of the Brian of the Chick (Gallus domesticus),
The Johns Hopkins University Press, Baltimore.
16.Gilbert, D. B., Patterson, T. A., and Rose, S. P.
(1991) Dissociation of brain sites necessary for registration and
storage of memory for a one-trial passive avoidance task in the chick,
Behav. Neurosci., 105, 553-561.
17.Csillag, A. (1999) Striato-telencephalic and
striato-tegmental circuits: relevance to learning in domestic chicks,
Behav. Brain Res., 98, 227-236.
18.Deng, C., and Rogers, L. J. (2000) Organization
of intratelencephalic projections to the visual Wulst of the chick,
Brain Res., 856, 152-162.
19.Shimizu, T., Cox, K., and Karten, H. J. (1995)
Intratelencephalic projections of the visual Wulst in pigeons
(Columba livia), J. Comp. Neurol., 359,
551-572.
20.Canal, C. E., Chang, Q., and Gold, P. E. (2007)
Amnesia produced by altered release of neurotransmitters after
intraamygdala injections of a protein synthesis inhibitor, Proc.
Natl. Acad. Sci. USA, 104, 12500-12505.
21.Sharma, A. V., Nargang, F. E., and Dickson, C. T.
(2012) Neurosilence: profound suppression of neural activity following
intracerebral administration of the protein synthesis inhibitor
anisomycin, J. Neurosci., 32, 2377-2387.
22.Nikolakopoulou, A. M., Davies, H. A., and
Stewart, M. G. (2006) Passive avoidance training decreases synapse
density in the hippocampus of the domestic chick, Eur. J.
Neurosci., 23, 1054-1062.
23.Knapska, E., and Kaczmarek, L. (2004) A gene for
neuronal plasticity in the mammalian brain:
Zif268/Egr-1/NGFI-A/Krox-24/TIS8/ZENK? Prog. Neurobiol.,
74, 183-211.
24.Solomonia, R. O., and McCabe, B. J. (2015)
Molecular mechanisms of memory in imprinting, Neurosci. Biobehav.
Rev., 50, 56-69.
25.Herold, C., Paulitschek, C., Palomero-Gallagher,
N., Gunturkun, O., and Zilles, K. (2018) Transmitter receptors reveal
segregation of the arcopallium/amygdala complex in pigeons (Columba
livia), J. Comp. Neurol., 526, 439-466.
26.Matsushima, T., Izawa, E., Aoki, N., and
Yanagihara, S. (2003) The mind through chick eyes: memory, cognition
and anticipation, Zool. Sci., 20, 395-408.
27.Lowndes, M., and Davies, D. C. (1994) The effects
of archistriatal lesions on one-trial passive avoidance learning in the
chick, Eur. J. Neurosci., 6, 525-530.
28.Patzke, N., Manns, M., and Gunturkun, O. (2011)
Telencephalic organization of the olfactory system in homing pigeons
(Columba livia), Neuroscience, 194, 53-61.
29.Nakamori, T., Maekawa, F., Sato, K., Tanaka, K.,
and Ohki-Hamazaki, H. (2013) Neural basis of imprinting behavior in
chicks, Dev. Growth Differ., 55, 198-206.