Transgenic Arabidopsis Plants Expressing Grape Glutathione S-Transferase Gene (VvGSTF13) Show Enhanced Tolerance to Abiotic Stress
Jing Xu1, Ai-Qing Zheng2, Xiao-Juan Xing1, Lei Chen1, Xiao-Yan Fu1, Ri-He Peng1, Yong-Sheng Tian1,a*, and Quan-Hong Yao1,b*
1Shanghai Key Laboratory of Agricultural Genetics and Breeding, Biotechnology Research Institute of Shanghai Academy of Agricultural Sciences, Shanghai 201106, China2Institute of Food Crops, Yunnan Academy of Agricultural Science, Kunming 650205, Yunnan, China
* To whom correspondence should be addressed.
Received June 5, 2017; Revision received March 2, 2018
Although glutathione S-transferase (GST, EC 2.5.1.18) is thought to play important roles in abiotic stress, limited information is available regarding the function of its gene in grapes. In this study, a GST gene from grape, VvGSTF13, was cloned and functionally characterized. Transgenic Arabidopsis plants containing this gene were normal in terms of growth and maturity compared with control plants but had enhanced resistance to salt, drought, and methyl viologen stress. The increased tolerance of the transgenic plants correlated with changes in activities of antioxidative enzymes. Our results indicate that the gene from grape plays a positive role in improving tolerance to salinity, drought, and methyl viologen stresses in Arabidopsis.
KEY WORDS: glutathione S-transferase, abiotic stress, stress tolerance, oxidative stress, grapeDOI: 10.1134/S0006297918060135
Abbreviations: CDNB, 1-chloro-2,4-dinitrobenzene; CP, control plants; GST, glutathione S-transferases; MV, methyl viologen; OE, GST-overexpressing lines of Arabidopsis; ROS, reactive oxygen species; T, transgenic plant generation.
Environmental stress is the major limiting factor in plant productivity.
Much of the injury to plants caused by stress exposure is associated
with oxidative damage at the cellular level resulting from increased
production of reactive oxygen species (ROS) [1].
Therefore, plants employ a complex array of detoxification mechanisms
to keep ROS production under control [2].
Glutathione S-transferases (GSTs) are a ubiquitous superfamily of
multifunctional enzymes involved in cellular detoxification of a wide
variety of endobiotic and xenobiotic substrates by conjugating the
tripeptide (γ-Glu-Cys-Gly) glutathione [3, 4]. Based on predicted amino acid sequences, the
soluble GSTs in plants have been grouped into several classes including
Phi, Tau, and Lambda, Dehydroascorbate reductase (DHAR), Theta, Zeta,
Elongation factor 1 gamma (EF1G), and tetrachlorohydroquinone
dehalogenase (TCHQD) [5]. Among these, Phi, Tau,
Lambda, and DHAR classes are specific to plants.
In plants, GSTs were first discovered and are best-known for their ability to detoxify a variety of toxic exogenous compounds, especially herbicides [6]. However, apart from herbicide detoxification, plant GSTs are also known to play an important role in abiotic and biotic stress responses, including salt [7], dehydration [8], hydrogen peroxide [9], metals stress [10], cold [11], UV-light [12], and pathogen attack [13]. Some studies have reported that the expression of GST is induced by plant hormones such as auxin [14], ethylene [15], ABA [16], salicylic acid, and methyl jasmonate [17].
Grape (Vitis vinifera) is one of the world’s largest fruit crops, approximating annual production of 58 million metric tons [18]. Although many GST genes have been cloned and reported, the role of a GST gene from grape is little investigated. In this study, the GST coding sequence VvGSTF13 from grape V. vinifera was cloned by PCR and functionally characterized by heterologous expression in Arabidopsis. Detailed analysis carried out on transgenic lines developed in this study suggests a role of VvGSTF13 in abiotic stress tolerance, particularly in osmotic, salt, and methyl viologen (MV) stress conditions.
MATERIALS AND METHODS
Plant materials and growth conditions. Plants (Arabidopsis thaliana ecotype Columbia L.) were grown in a growth chamber at 22°C on MS medium or in pots filled with vermiculite/peat moss/perlite (9 : 3 : 1) mixture. The plants were kept in a 16 h/8 h day/night cycle at light intensity of ~120 µmol photons·m–2·s–1.
Cloning and sequence analysis of VvGSTF13. The VvGSTF13 (GenBank accession number XM_002263350) open reading frame was cloned from grape V. vinifera leaf cDNA using RT-PCR with specific primers for VvGSTF13 genes (forward: VvGSTF13: 5′-AAGGATCCATGGCGCTCAAGCTCCATG-3′; reverse: VvGSTZ13: 5′-AAGAGCTCTCACTTCTCACCAAAAG-3′). Clones containing the VvGSTF13 coding sequence were further sequenced from both sides to confirm their sequences.
Molecular weight (MW) and isoelectric point (pI) predictions for the deduced protein were estimated using the Compute pI/MW tool (http://web.expasy.org/protparam/). The phylogenetic tree was constructed using the Clustal W2 program.
Vector construction and Arabidopsis transformation. The complete open reading frame of VvGSTF13 was amplified by PCR using gene-specific primers as described, cloned in TA clone vector Simple pMD-18 (Takara, Japan), and sequenced. The fragment was then cloned in the plant expression vector pYK4102 [19] at BamHI and SacI sites under the control of the CaMV35S promoter. The construct was mobilized into Agrobacterium tumefaciens GV3101 and transformed in plants to generate GST-overexpressing lines (OE) of Arabidopsis using a floral dip method [20], and the empty vector without VvGSTF13 was transformed in plants to generate control plants (CP). Both positive transformants in hygromycin (50 µg/ml) plates were selected, and GST overexpressors were confirmed by PCR using the same primers. The T3 generation was selected for further experiments.
Reverse transcription PCR. Three-week-old Arabidopsis plants were harvested to analyze the GST-overexpressing transformants. Total RNA was isolated using a Multisource Total RNA Miniprep kit (Axygen Scientific, USA) according to the manufacturer’s instructions. First-strand cDNA synthesis was conducted with 5 µg of total RNA using a TransScript Fly First-strand cDNA Synthesis SuperMix (Transgen Biotech, China). Specific primer information for VvGSTF13 was mentioned above. The A. thaliana actin gene (AtActin2, GenBank Accession number U41998) was used as an internal control, and primers were as follows (forward: AtAc2F: 5′-AGTAAGGTCACGTCCAGCAAGG-3′; reverse: AtAc2Z: 5′-GCACCCTGTTCTTCTTACCGAG-3′). The PCR was performed under the following conditions: 10 min at 94°C, followed by 22 cycles of 20 s at 94°C, 20 s at 56°C, 20 s at 72°C, and a final extension at 72°C for 5 min. VvGSTF13 was amplified using PCR conditions as follows: 10 min at 94°C, followed by different cycles of 35 s at 94°C, 30 s at 56°C, 30 s at 72°C, and a final extension at 72°C for 5 min. The PCR products were separated on 2% agarose gel. The DNA intensity ratio of the VvGSTF13 gene to AtAc2 was analyzed with a Shine Tech Gel Analyzer (Shanghai Shine Science of Technology Co., Ltd., China). The same results were obtained in three independent experiments. Therefore, only the results from one experiment are presented.
GST activity measurement. GST activity was determined according to Ji et al. [21] with minor modification. Enzyme activity was tested in the presence of 1 mM glutathione in 100 mM sodium phosphate buffer (pH 6.5). The reaction was started with the addition of 1-chloro-2,4-dinitrobenzene (CDNB) to a final concentration of 1 mM. The changes in optical absorbance at 340 nm were measured. The background levels of spontaneous CDNB decay were subtracted.
Stress tolerance analysis. For germination and seedling growth assay, seeds of both overexpressing and control plants were stratified and planted on MS medium with or without different concentrations of NaCl, mannitol, or MV. The seeds were incubated at 4°C for 2 days before being placed at 22°C under light conditions. Germination rate and cotyledon emergence rate were scored.
For root length assay, seeds were germinated on MS medium for 2 days and then transferred carefully to MS medium containing different concentrations of NaCl or mannitol. Root length was measured after 7 days.
For drought treatment, watering was withheld for 2 weeks for 3-week-old plants grown in pots. The performance of control and transgenic plants was compared when irrigation was resumed.
For salt treatment, 3-week-old plants grown in pots were watered with NaCl (250 mM). Two weeks later, the performance of control and transgenic plants was compared.
For MV treatment, seeds were surface-sterilized and grown on MS solid medium for 15 days. Twenty seedlings were then transferred aseptically into 50-ml conical flasks containing 20 ml of MS liquid medium and grown with rotary shaking (60 rpm) for recovering. After 2 days, the liquid medium was refreshed with new medium that contained MV at different concentrations. The dry weight was scored after 10 days. Total chlorophyll was extracted and assayed as described previously [7].
Measurement of physiological parameters involved in stress tolerance. To measure standardized water content, aerial parts from 3-week-old pot-grown plants were excised and placed on filter paper. The loss of fresh weight was monitored at indicated times. The method was as described by Xu et al. [19].
Furthermore, 3-week-old plants were treated with 250 mM NaCl (salt stress), or 300 mM mannitol (drought stress), or 10 µM MV for 4 days, and then malondialdehyde (MDA) content, electrolyte leakage, superoxide dismutase (SOD; EC 1.15.1.1) and peroxidase (POD; EC 1.11.1.7) activities were measured.
To measure MDA levels and electrolyte leakage, leaves from 3-week-old plants of the same genotype under the same treatment were mixed as sample. MDA content was determined by the reaction of thiobarbituric acid as described previously [22]. Electrolyte leakage was determined using a DDS-307 conductivity meter (Shanghai Reaches Instrument Co., Ltd., China) as described previously [2]. The electrical conductance was measured first after each experiment (ECinitial), then bathing solution containing the leaves was autoclaved at 120°C for 20 min and the electrical conductance was measured again to get the total ion leakage (ECmax). The relative electrolyte leakage was then calculated as a percentage: ECrel (%) = ECinitial/ECmax × 100.
To measure antioxidant enzyme activities, fresh leaf tissue (~0.1 g) from 3-week-old plants of the same genotype under the same treatment was harvested and homogenized in 1.6 ml of chilled 50 mM phosphate buffer and kept on an ice bath. The homogenate was filtered through two layers of muslin cloth and centrifuged at 20,000g for 10 min in a refrigerated centrifuge at 4°C. The supernatant was stored at 4°C and used for enzyme assays within 4 h. SOD activity was determined by monitoring the inhibition of photochemical reduction of nitroblue tetrazolium according to the method of Beyer and Fridovich [23]. POD activity was assayed according to the method of MacAdam [24].
Statistical analysis. The SPSS software version 15.0J (SPSS Inc., USA) was used for statistical analysis. ANOVA was performed followed by Duncan’s multiple comparison tests. Statistically significant differences (p < 0.05) are reported in the text and shown in the figures.
RESULTS
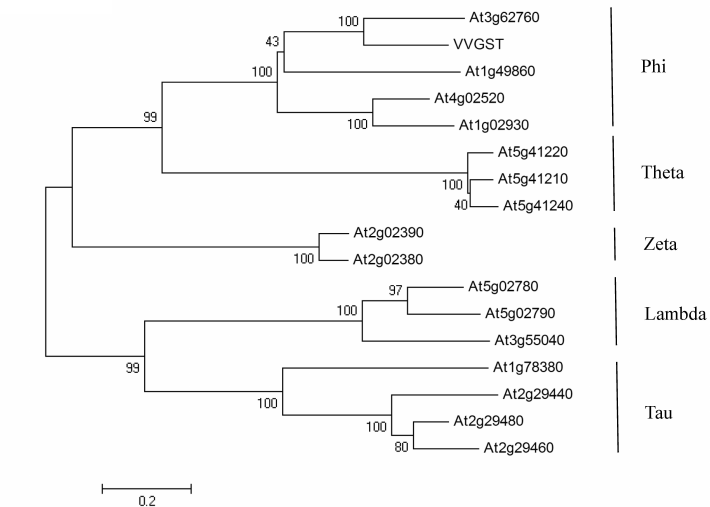
Cloning and characterization of VvGSTF13. The coding region of VvGSTF13 was 654 bp in length and was predicted to encode a 217-amino acid protein with a calculated MW of 24.6 kDa and a pI of 6.16. Multiple sequence alignments showed that VvGSTF13 shares the highest similarity with AtGSTF13 (At3g62760), AtGSTF14 (At1g49860), AtGSTF2 (At4g02520), and AtGSTF6 (At1g02930), which suggests that VvGSTF13 may be a Phi-type GST (Fig. 1).
Fig. 1. Phylogenetic tree for the amino acid sequences of VvGSTF13 with the GST proteins from A. thaliana. The Neighbor-joining (NJ) method was applied using Mega 4 from a tree file produced by Clustal W2.
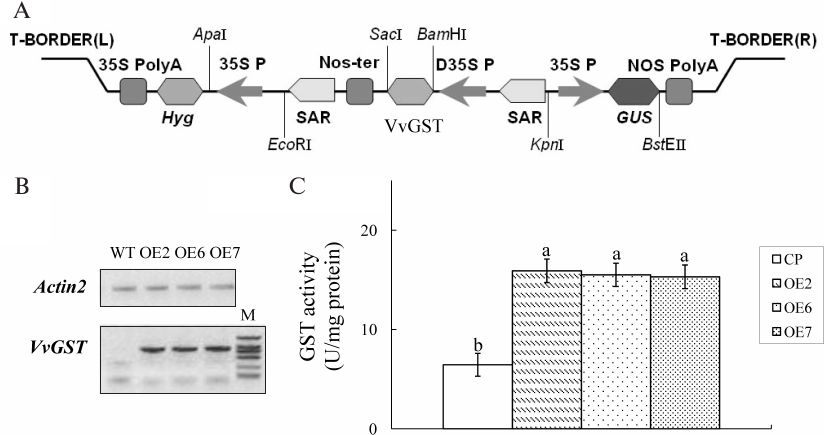
Expression of VvGSTF13 in Arabidopsis. VvGSTF13 cDNA was cloned into plant expression vector pYK4102 (Fig. 2A) and introduced to Arabidopsis cells using the A. tumefaciens-mediated floral dip method. Ten independent lines of transgenic plants (T1 generation) were generated. Among these lines, three homozygous transgenic lines (named OE2, OE6, and OE7) expressing VvGSTF13 of T3 generation were selected for further experiments.
RT-PCR analysis results confirmed that VvGSTF13 was detected in the three transgenic lines in the T3 generation, and no amplification was observed in control (Fig. 2B). GST activity levels were analyzed in both OE plants and control. Compared with control, different OE lines showed a significantly enhanced GST activity when CDNB was used as substrate (Fig. 2C). No obvious effects on growth and development were observed in VvGSTF13 OE plants under normal growth conditions.
Fig. 2. Transgenic Arabidopsis plants with VvGSTF13. A) The VvGSTF13 expression vector for Arabidopsis transformation. Nos-Ter, terminator sequence of the nopaline synthase; SAR, scaffold attachment region. B) Expression of VvGSTF13 in the three transgenic lines and control. Total RNA was isolated from 3-week-old plants grown under normal conditions. M, marker (DL2000; Takara). C) GST activity in Arabidopsis plants expressing VvGSTF13. GST activity was measured in leaves of 3-week-old plants grown under normal condition using CDNB as substrate. Values are means ± SD (n = 3) and different letters above bars indicate significant differences (p < 0.05) among different lines.
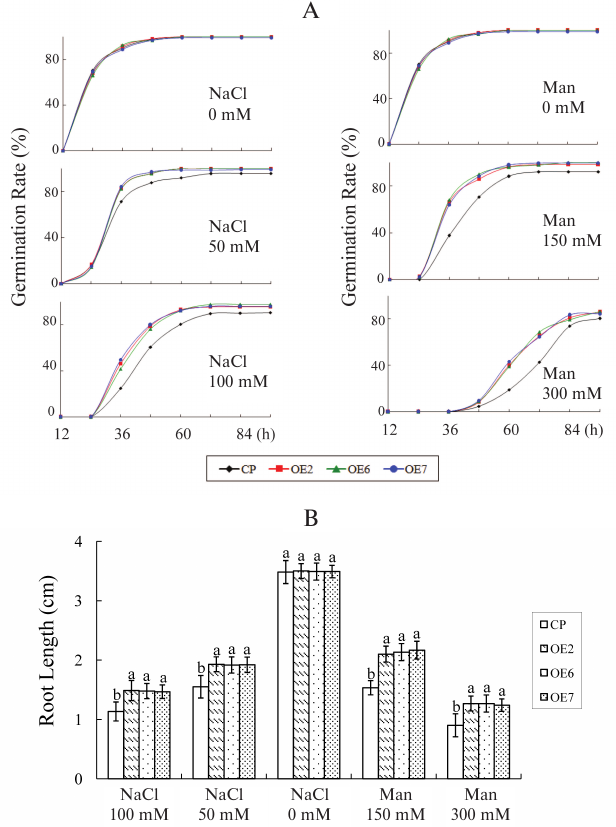
Response of transgenic lines to salt stress and osmotic stress. Seeds were germinated in medium containing different concentrations of NaCl (50 and 100 mM) as salt stress or mannitol (150 and 300 mM) as osmotic stress to analyze the role of VvGSTF13 under stress. There was no significant difference in the germination rates and root length between control and OE plants under normal growth conditions. However, the germination rate and root length were increased in the OE lines compared with control under salt and osmotic stress conditions (Fig. 3). These results suggested that OE plants had enhanced tolerance to salt and osmotic stress.
Fig. 3. Response of VvGSTF13 transgenic plants to salt and osmotic stress. A) Seed germination. The germination rates were recorded in MS medium supplemented with different concentration of NaCl (as salt stress) and mannitol (as osmotic stress) during a period from 12 to 96 h after stratification. Each value is the mean ± SD (n = 3) of at least 50 seeds. B) Primary root length of different genotypes under salt stress and osmotic stress. Seeds were germinated on MS medium for 2 days and then transferred carefully to MS medium containing different concentrations of NaCl or mannitol. Root length was measured after 7 days. Each value is the mean ± SD of at least 50 seedlings.
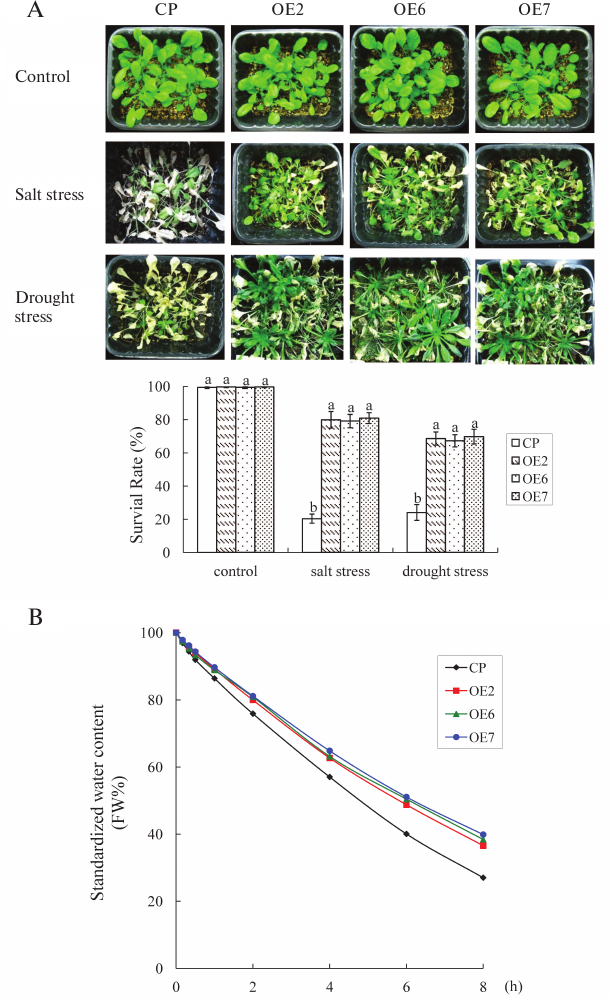
Salt and drought stress tolerance. Three-week-old seedlings were watered with NaCl (250 mM) for salt stress or with withheld watering for water deficit stress to further analyze salt/drought tolerance. After 2 weeks, some plants exhibited lethal effects, and then the plants were irrigated again. The plants were photographed on the 15th day after the plants were exposed to stress, and the survival rate was then measured (Fig. 4A). Under salt stress condition, >80% of the OE plants survived, whereas only 20% of the control plants survived. Under water deficit stress for 2 weeks, 68% of the OE plants survived, whereas approximately 20% of control plants survived.
Fig. 4. Salt and drought stress tolerance of transgenic plants. A) Three-week-old plants were given salt or drought stress treatment. Control, 3-week-old plants growing under normal conditions. Salt stress, plants watered with NaCl (250 mM). Drought stress, plants with withheld watering for 2 weeks, then irrigated again. Survival rate was analyzed. Values are mean ± SD of three individual experiments. The statistical significance was determined by Duncan’s multiple comparison tests. Different letters above bars indicate significant differences (p < 0.05) among different genotypes with the same treatment. B) Standardized water content of different lines. Each data point is the mean ± SD of three replicates, each from ≥10 plants.
The water content was analyzed further. When the leaves of control and OE plants were exposed to air for 2 h, the average water content rate of the OE plants was 80.6%, whereas it was 75.8% in control. At 8 h, the average water content rate of OE plants was 38.2%, which was 1.4-fold higher than that of the control. These results indicated that OE plants had a higher capacity to conserve water than the control plants (Fig. 4B).
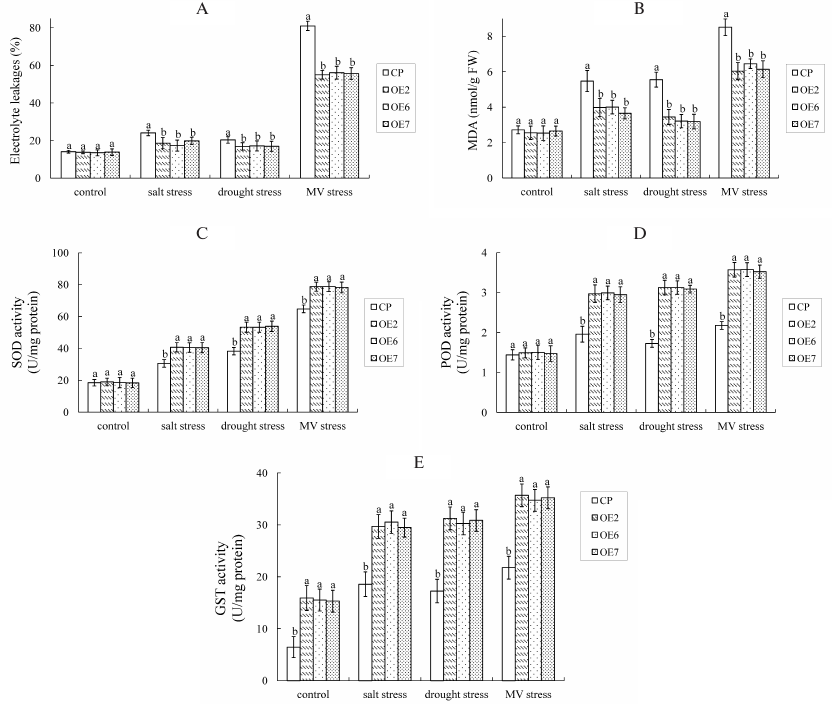
MDA and electrolyte leakages were analyzed (Fig. 5, A and B), and the results showed that there were no differences between OE plants and control under normal conditions. Under stress conditions, MDA and electrolyte leakages increased in both OE plants and control. However, the contents of MDA and electrolyte leakages were significantly higher in control than in OE plants during NaCl and mannitol stresses, which means control plants exhibited higher rates of cell damage than OE lines under stress conditions.
The activities of SOD, POD, and GST were also measured (Fig. 5, C-E), and the results showed that SOD and POD activities had no significant difference between OE and control plants under normal conditions. Under stress conditions, the SOD, POD, and GST activities increased in both the OE and control plants. However, the SOD, POD, and GST activities in OE lines were significantly higher than in the control plants during NaCl and mannitol stresses. These results suggest that VvGSTF13 strengthens cellular ROS scavenging ability and maintains ROS homeostasis by increasing the activities of antioxidant enzymes.
Fig. 5. Changes in the electrolyte leakage, content of MDA, and activities of antioxidant enzymes with different lines treated with NaCl (250 mM) as salt stress, mannitol (300 mM) as drought stress, and MV (10 µM) stress for 4 days: A) electrolyte leakage; B) MDA content; C) SOD activity; D) POD activity; E) GST activity. Values are mean ± SD (n = 3), each from 10 plants.
Statistical significance was determined by Duncan’s multiple comparison tests. Different letters above bars indicate significant differences (p < 0.05) among different lines in the same treatment.
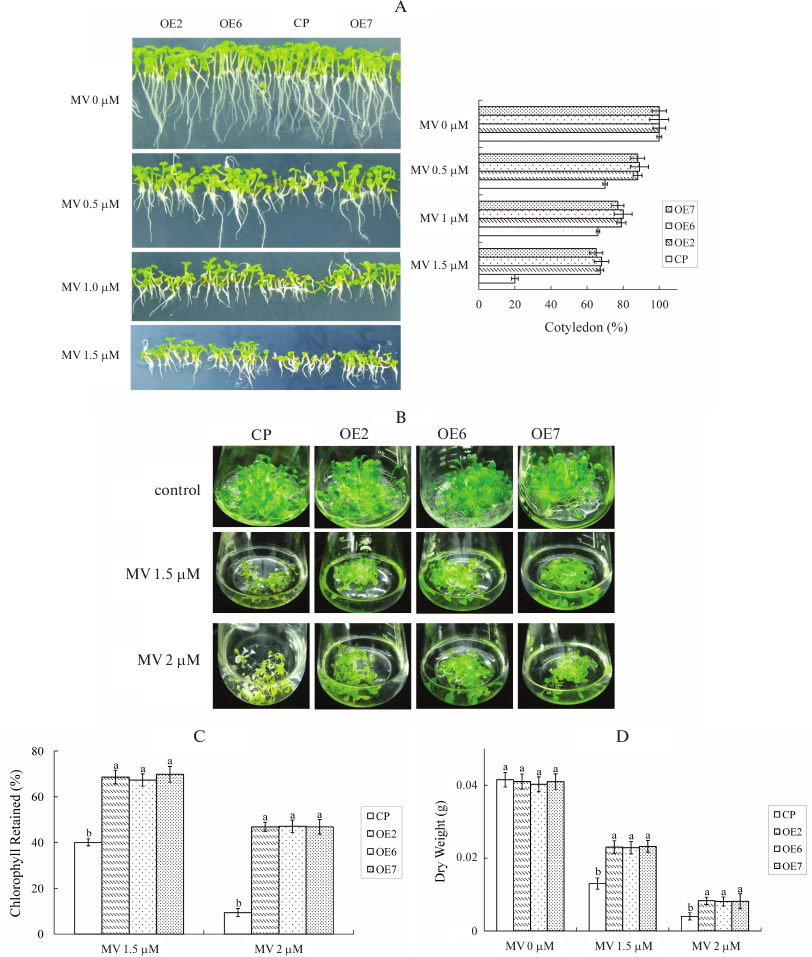
MV stress tolerance of transgenic plants. Given that GST is involved in anti-oxidation processes in plants and animals, we analyzed the anti-oxidation capacity of transgenic plants. Seeds were germinated on the MS medium containing different MV concentrations, and the percentage of cotyledon emergence was compared between OE and control plants. Photographs were taken on the 7th day after stratification. In the medium containing 0.5 µM MV, the cotyledon emergence rate of control was 70%, and the mean rate of OE lines was approximately 88%. When the MV concentration reached 1.5 µM, the cotyledon emergence rate decreased to 20% in control, and the mean rate of OE lines was approximately 66% (Fig. 6A).
Fig. 6. MV stress tolerance of transgenic plants. A) Phenotypes and cotyledon emergency rate of different genotypes under MV stress treatment. Seeds were germinated on MS containing different concentrations of MV. Cotyledon emergency rate was measured after 7 days. Each value is the mean ± SD of at least 50 seedlings. B) Twenty seedlings grown in MS solid medium for 2 weeks were transferred to MS liquid medium in flasks containing different concentrations of MV. C) Chlorophyll content of seedlings in the individual flasks with treatment as mentioned in (B). The percentage of chlorophyll retained was calculated as follows: the chlorophyll content of each genotype was measured after MV treatment and compared to the chlorophyll content of plants grown on MS liquid medium only. The experiments were repeated three times. Values are means ± SD (n = 3) and different letters above bars indicate significant differences (p < 0.05) among different genotypes in the same treatment. D) Dry weights of seedlings in the individual flasks with treatment as mentioned in (B). Values are means ± SD (n = 3) and different letters above bars indicate significant differences (p < 0.05) among different genotypes with the same treatment.
To analyze MV tolerance, 2-week-old T3 seedlings grown on MS solid medium were transferred aseptically into MS liquid medium and grown with rotary shaking (60 rpm) for recovery. After 2 days, the liquid medium was refreshed with new medium supplemented with different MV concentrations. Photographs were taken on the 10th day after exposure to stress (Fig. 6B). The seedlings of control and OE lines grown in the liquid medium without MV appeared normal. This result indicated that toxic effects were caused by MV stress and not the result of submersion in the liquid medium. The dry weights of control and OE lines were nearly the same without MV stress. However, the growth of control plants was inhibited when MV stress was induced. When control plants were treated with 1.5 µM MV, the growth of plants was significantly inhibited, the chlorophyll content decreased, and the leaves turned chlorotic. The retained chlorophyll content compared with normal condition was only 40% in control, while the mean content in OE lines was 68.6% (Fig. 6C). The relative dry weight rates of control and OE lines were 32 and 52%, respectively (Fig. 6D). When MV concentration reached 2 µM, the growth of both control and OE plants was inhibited, and the leaves turned chlorotic. The retained chlorophyll contents were 9.3 and 46% in control and OE lines, respectively. The relative dry weight rates were 9 and 20% in control and OE lines, respectively. When MV concentration reached 3 µM, both control and OE plants died (data not shown). These results showed that OE plants exhibit higher tolerance to MV stress than control plants.
MDA and electrolyte leakages were analyzed (Fig. 5, A and B). Under stress conditions, MDA and electrolyte leakages increased in both OE and control plants. However, the contents of MDA and electrolyte leakages were significantly higher in control than in OE plants during MV stress, which means control plants exhibited higher rates of cell damage than OE lines under stress conditions.
The antioxidant enzymes were also detected under MV stress conditions (Fig. 5, C-E). Under stress conditions, the SOD, POD, and GST activities increased in both OE and control plants. However, SOD, POD, and GST activities in OE lines were significantly higher than in the control plants under MV stress. The enzyme profiles under MV stress were similar to those observed under salt/drought stress.
Together, the overexpression of VvGSTF13 enhanced the tolerance to salt, drought, and MV stresses in Arabidopsis. VvGSTF13 is suggested to play an important role in adaptation to at least three distinct stress agents (salt, drought, and MV).
DISCUSSION
Environment stress, such as pathogen attack, wounding, drought, high heavy metal content, and extreme high or low temperatures lead to the generation of ROS in plant cells, which then produce membrane lipid peroxidation and form base propenals, highly cytotoxic products of oxidative DNA damage [25]. GSTs were proposed to afford protection under various stress conditions by detoxifying endogenous plant toxins that accumulate due to increased oxidative stress [8]. Although many GST genes have been cloned and analyzed, the role of a GST gene from grape has been little investigated. In this study, VvGSTF13, a GST gene from grape, was cloned and introduced into Arabidopsis to evaluate gene function during abiotic stresses. Our results revealed that transgenic Arabidopsis plant lines overexpressing the VvGSTF13 gene have improved drought, salinity, and MV tolerance.
Functional analysis on the role of VvGSTF13 in abiotic stress tolerance of Arabidopsis was carried out using transgenic OE lines. The phenomena of increased germination, root growth, and percentage of cotyledon emergence were observed under stress treatment (Figs. 3 and 6A), which suggests that VvGSTF13 play a role in tolerance phenotype. Recent studies have shown that expression of a rice Lambda class of GST, OsGSTL2, in Arabidopsis provides tolerance through increasing root growth, seed germination, and cotyledon emergence in heavy metal stress, and increasing root growth in salt and osmotic stress [10]. Similar phenotypes were also observed in the functional study of ThGSTZ1 from Tamarix hispida in drought and salinity stresses tolerance [26]. Furthermore, OE plants displayed tolerance to salt/drought stress and conserved water to a higher extent than control plants (Fig. 4); these results suggest that VvGSTF13 plays a positive role in tolerance to salt/drought stress.
Exposure to drought and salt stress caused increased formation of reactive oxygen species and thus oxidative stress [8]. Therefore, a complex array of detoxification mechanisms has evolved to maintain ROS homeostasis, including not only non-enzymatic components such as antioxidant ascorbate and glutathione, but also enzymatic scavengers. The major antioxidant enzymes include SOD, POD, catalase (CAT), GST, and guaiacol peroxidase (GPx) [27]. In fact, overexpression of a specific antioxidant gene has been shown to influence the expression of other antioxidant genes. For instance, transgenic GST expressing seedlings were shown to have a higher ascorbate peroxidase (APX) activity and monodehydroascorbate reductase (MDHAR) activity compared to non-transformed seedlings [28]. Tobacco plants expressing a human DHAR (dehydroascorbate reductase) gene showed an increased glutathione reductase (GR) activity [29]. Tobacco chloroplast transformants expressing DHAR and GST genes had increased GR activity compared to the wild type plants [2]. In this study, overexpression of VvGSTF13 resulted in increased activities of SOD and POD and decreased MDA and electrolyte leakages under stress conditions compared to the control. This suggests that VvGSTF13 acts as a stress regulator through increasing the activity of antioxidant enzymes to strengthen ROS scavenging ability or maintain ROS homeostasis.
Methyl viologen (paraquat) also causes oxidative stress by increasing the production of O2– in both animal and plant systems [30]. The effect of VvGSTF13 on resistance to oxidative stress caused by MV was also explored through the analysis of chlorophyll retention, electrolyte leakage, lipid peroxidation, and antioxidant enzymes. The results were similar to those in experiments when plants were exposed to salt and drought stresses. Yu et al. transformed a cotton GST gene into tobacco and observed an enhanced resistance to oxidative stress induced by MV in the transgenic tobacco plants [9]. Overexpression of VvGSTF13 resulted in increased tolerance to MV stress treatment through enhancing activities of antioxidant enzymes. Lee et al. demonstrated that overexpression of Cu,Zn-SOD, APX, and DHAR genes in tobacco chloroplasts elevated protection against MV and salt stress [31]. In a recent study, transplastomic tobacco plants expressing GST and GR genes showed enhanced tolerance to MV induced oxidative stresses [2].
In this work, improved protection against MV and salt/drought stress could be explained by synergistic effects of GST with SOD and POD. The expression of these antioxidant enzymes would participate in a more rapid scavenging of superoxide radicals and hydrogen peroxide before their interaction with target molecules.
In conclusion, VvGSTF13 plays a positive role in tolerance to salt/drought/MV stress in Arabidopsis. Further analyses, particularly on the role of phytohormones, are needed to elucidate the function and the regulatory mechanism of VvGSTF13 in plant stress responses.
Acknowledgments
This study was sponsored by the Shanghai Rising-Star Program (Grant No. 14QB1403400), Shanghai Natural Science Foundation (Grant No. 13ZR1460600), National Natural Science Foundation (Grant Nos. 31300237, 31401458), and Youth Talents Growth Plan of Shanghai Academy of Agricultural Sciences (Grant No. 2015-1-26).
REFERENCES
1.Bowler, C., Montagu, M. V., and Inze, D. (1992)
Superoxide dismutase and stress tolerance, Annu. Rev. Plant Physiol.
Plant Mol. Biol., 43, 83-116.
2.Le Martret, B., Poage, M., Shiel, K., Nugent, G.
D., and Dix, P. J. (2011) Tobacco chloroplast transformants expressing
genes encoding dehydroascorbate reductase, glutathione reductase, and
glutathione-S-transferase, exhibit altered antioxidant metabolism and
improved abiotic stress tolerance, Plant Biotechnol. J.,
9, 661-673.
3.Cummins, I., Dixon, D. P., Freitag-Pohl, S.,
Skipsey, M., and Edwards, R. (2011) Multiple roles for plant
glutathione transferases in xenobiotic detoxification, Drug Metab.
Rev., 43, 266-280.
4.Dixon, D. P., Lapthorn, A., and Edwards, R. (2002)
Plant glutathione transferases, Genome Biol., 3,
REVIEWS3004.
5.Edwards, R., and Dixon, D. P. (2005) Plant
glutathione transferases, Methods Enzymol., 401,
169-186.
6.Edwards, R., Del Buono, D., Fordham, M., Skipsey,
M., Brazier, M., Dixon, D. P., and Cummings, I. (2005) Differential
induction of glutathione transferases and glucosyltransferases in
wheat, maize and Arabidopsis thaliana by herbicide safeners, Z.
Naturforsch. C, 60, 307-316.
7.Yang, G., Wang, Y., Xia, D., Gao, C., Wang, C., and
Yang, C. (2014) Overexpression of a GST gene (ThGSTZ1) from Tamarix
hispida improves drought and salinity tolerance by enhancing the ablity
to scavenge reacive oxygen species, Plant Cell Tiss. Organ.
Cult., 117, 99-112.
8.Chen, J. H., Jiang, H. W., Hsieh, E. J., Chen, H.
Y., Chien, C. T., Hsieh, H. L., and Lin, T. P. (2012) Drought and salt
stress tolerance of an Arabidopsis glutathione S-transferase U17
knockout mutant are attributed to the combined effect of glutathione
and abscisic acid, Plant Physiol., 158, 340-351.
9.Yu, T., Li, Y. S., Chen, X. F., Hu, J., Chang, X.,
and Zhu, Y. G. (2003) Transgenic tobacco plants overexpressing cotton
glutathione S-transferase (GST) show enhanced resistance to methyl
viologen, J. Plant Physiol., 160, 1305-1311.
10.Kumar, S., Asif, M. H., Chakrabarty, D.,
Tripathi, R. D., Dubey, R. S., and Trivedi, P. K. (2013) Expression of
a rice Lambda class of glutathione S-transferase, OsGSTL2, in
Arabidopsis provides tolerance to heavy metal and other abiotic
stresses, J. Hazard. Mater., 248-249, 228-237.
11.Seppanen, M. M., Cardi, T., Borg Hyokki, M., and
Pehu, E. (2000) Characterization and expression of cold-induced
glutathione S-transferase in freezing tolerant Solanum commersonii,
sensitive S. tuberosum and their interspecific somatic hybrids,
Plant Sci., 153, 125-133.
12.Loyall, L., Uchida, K., Braun, S., Furuya, M.,
and Frohnmeyer, H. (2000) Glutathione and a UV light-induced
glutathione S-transferase are involved in signaling to chalcone
synthase in cell cultures, Plant Cell, 12, 1939-1950.
13.Mauch, F., and Dudler, R. (1993) Differential
induction of distinct glutathione-S-transferases of wheat by
xenobiotics and by pathogen attack, Plant Physiol., 102,
1193-1201.
14.Takahashi, Y., and Nagata, T. (1992) parB: an
auxin-regulated gene encoding glutathione S-transferase, Proc. Natl.
Acad. Sci. USA, 89, 56-59.
15.Zhou, J., and Goldsbrough, P. B. (1993) An
Arabidopsis gene with homology to glutathione S-transferases is
regulated by ethylene, Plant Mol. Biol., 22, 517-523.
16.Xu, F., Lagudah, E. S., Moose, S. P., and
Riechers, D. E. (2002) Tandemly duplicated Safener-induced glutathione
S-transferase genes from Triticum tauschii contribute to genome- and
organ-specific expression in hexaploid wheat, Plant Physiol.,
130, 362-373.
17.Wagner, U., Edwards, R., Dixon, D. P., and Mauch,
F. (2002) Probing the diversity of the Arabidopsis glutathione
S-transferase gene family, Plant Mol. Biol., 49,
515-532.
18.Jayaprakasha, G. K., Singh, R. P., and Sakariah,
K. K. (2001) Antioxidant activity of grape seed (Vitis vinifera)
extracts on peroxidation models in vitro, Food Chemistry,
73, 285-290.
19.Xu, J., Tian, Y. S., Peng, R. H., Xiong, A. S.,
Zhu, B., Jin, X. F., Gao, F., Fu, X. Y., Hou, X. L., and Yao, Q. H.
(2010) AtCPK6, a functionally redundant and positive regulator involved
in salt/drought stress tolerance in Arabidopsis, Planta,
231, 1251-1260.
20.Zhang, X., Henriques, R., Lin, S. S., Niu, Q. W.,
and Chua, N. H. (2006) Agrobacterium-mediated transformation of
Arabidopsis thaliana using the floral dip method, Nat. Protoc.,
1, 641-646.
21.Ji, W., Zhu, Y., Li, Y., Yang, L., Zhao, X., Cai,
H., and Bai, X. (2010) Over-expression of a glutathione S-transferase
gene, GsGST, from wild soybean (Glycine soja) enhances drought and salt
tolerance in transgenic tobacco, Biotechnol. Lett., 32,
1173-1179.
22.Cakmak, I., and Horst, W. J. (1991) Effect of
aluminium on lipid peroxidation, superoxide dismutase, catalase, and
peroxidase activities in root tips of soybean (Glycine max),
Physiol. Plant., 83, 463-468.
23.Beyer, W. F., Jr., and Fridovich, I. (1987)
Assaying for superoxide dismutase activity: some large consequences of
minor changes in conditions, Anal. Biochem., 161,
559-566.
24.Macadam, J. W., Sharp, R. E., and Nelson, C. J.
(1992) Peroxidase activity in the leaf elongation zone of tall fescue.
II. Spatial distribution of apoplastic peroxidase activity in genotypes
differing in length of the elongation zone, Plant Physiol.,
99, 879-885.
25.Marrs, K. A. (1996) The functions and regulation
of glutathione S-transferases in plants, Annu. Rev. Plant Physiol.
Plant Mol. Biol., 47, 127-158.
26.Yang, G., Wang, Y., Xia, D., Gao, C., Wang, C.,
and Yang, C. (2014) Overexpression of a GST gene (ThGSTZ1) from Tamarix
hispida improves drought and salinity tolerance by enhancing the
ability to scavenge reactive oxygen species, Plant Cell Tiss. Organ.
Cult., 117, 99-112.
27.Jiang, Y., Yang, B., Harris, N. S., and Deyholos,
M. R. (2007) Comparative proteomic analysis of NaCl stress-responsive
proteins in Arabidopsis roots, J. Exp. Bot., 58,
3591-3607.
28.Roxas, V. P., Lodhi, S. A., Garett, D. K., Mahan,
J. R., anad Allen, R. D. (2000) Stress tolerance in transgenic tobacco
seedlings that overexpress glutathione S-transferase/glutathione
peroxidase, Plant Cell Physiol., 41, 1229-1234.
29.Kwon, S. Y., Ahn, Y. O., Lee, H. S., and Kwak, S.
S. (2001) Biochemical characterization of transgenic tobacco plant
expressing a human dehydroascorbate reductase, J. Biochem. Mol.
Biol., 34, 316-321.
30.Hassan, H. M., and Fridovich, I. (1977)
Regulation of the synthesis of superoxide dismutase in Escherichia
coli. Induction by methyl viologen, J. Biol. Chem., 252,
7667-7672.
31.Lee, Y. P., Kim, S. H., Bang, J. W., Lee, H. S.,
Kwak, S. S., and Kwon, S. Y. (2007) Enhanced tolerance to oxidative
stress in transgenic tobacco plants expressing three antioxidant
enzymes in chloroplasts, Plant Cell Rep., 26,
591-598.