REVIEW: Programmed Aging of Mammals: Proof of Concept and Prospects of Biochemical Approaches for Anti-aging Therapy
M. V. Skulachev* and V. P. Skulachev
Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119991 Moscow, Russia; E-mail: skulach@belozersky.msu.ru* To whom correspondence should be addressed.
Received September 4, 2017
(i) In 2015-2017 we compared possible reasons for longevity of two mammalian highly social species, i.e. naked mole rats and humans. We proposed that in both cases longevity is a result of neoteny, prolongation of youth by deceleration of late ontogeny (Skulachev, V. P. (2015) Abst. 11th Conf. on Mitochondrial Physiology (MiP2015), Lucni Bouda, Czech Republic, pp. 64-66; Skulachev, V. P., Holtze, S., Vyssokikh, M. Y., Bakeeva, L. E., Skulachev, M. V., Markov, A. V., Hildebrandt, T. B., and Sadovnichii, V. A. (2017) Physiol. Rev., 97, 699-720). Both naked mole rats and humans strongly decreased the pressure of natural selection, although in two different manners. Naked mole rats preferred an “aristocratic” pathway when reproduction (and, hence, involvement in evolution) is monopolized by the queen and her several husbands. Huge number of subordinates who have no right to take part in reproduction and hence in evolution serves the small queen’s family. Humans used an alternative, “democratic” pathway, namely technical progress facilitating adaptation to the changing environmental conditions. This pathway is open to all humankind. (ii) As a result, aging as a mechanism increasing evolvability by means of facilitation of natural selection became unnecessary for naked mole rats and humans due to strong attenuation of this selection. This is apparently why aging became a counterproductive atavism for these two species and was strongly shifted to late ages. This shift is direct evidence of the hypothesis that aging is programmed, being the last step of late ontogeny. (iii) Further deceleration of aging for humans by means of neoteny is unrealistic since the development of neoteny probably takes million years. (iv) However, if biological aging is a program, an alternative and much simpler way to avoid it seems possible. We mean inhibition of an essential step of this program. (v) At present, the most probable scheme of the aging program assumes that it is a mechanism of slow poisoning of an organism by reactive oxygen species produced by mitochondria. If this is the case, a mitochondria-targeted antioxidant might be an inhibitor of the aging program. During the last 12 years, such an antioxidant, namely SkQ1, was synthesized and studied in detail in our group. It consists of plastoquinone and decyltriphenylphosphonium (a penetrating cation responsible for electrophoretic accumulation of SkQ1 in mitochondria). It was shown that long-term treatment with SkQ1 increased the lifespan of plants, fungi, invertebrates, fish, and mammals. Moreover, SkQ1 is effective in the therapy of various age-related diseases. It was also shown that a single SkQ1 injection could save life in certain models of sudden death of animals. (vi) A tentative scheme is proposed considering aging as a process of chronic phenoptosis, which eventually results in initiation of acute phenoptosis and death. This scheme also suggests that under certain conditions chronic phenoptosis can be neutralized by an anti-aging program that is activated by food restriction regarded by an organism as a signal of starvation. As for acute phenoptosis, it is apparently controlled by receptors responsible for measuring key parameters of homeostasis. The first experimental indications have been already obtained indicating that both chronic and acute phenoptosis are suppressed by SkQ1.
KEY WORDS: phenoptosis, aging program, mitochondria, evolution, Heterocephalus glaberDOI: 10.1134/S000629791712001X
THE MAIN QUESTION OF GERONTOLOGY: IS AGING A PROGRAM USED BY EVOLUTION FOR ITS ACCELERATION, OR A DEFECT OF EVOLUTION THAT COULD NOT COPE WITH ACCUMULATION OF RANDOM ERRORS?
The answer to the question posed in the title of this section is important primarily for medicine. If aging is the inevitable result of deterioration of such a complex system as the organism, its repair is possible only by replacing the worn-out organs with new ones. If it is a program written in our genome, then it can be broken (or, as programmers say, “hacked”). Obviously, it is easier to break the aging program than to build a new organism out of young or artificial organs. Shutting down the program of cell suicide (apoptosis) with the help of inhibitors of the enzymes involved in this process is an example of how to disable a genetic program that is lethal for cells.
Prior to the discovery of the mechanism of apoptosis by Kerr et al. in 1972 [1], it was considered indecent among biologists studying higher animals to argue that aging is a program. In this case, they mistakenly appealed to Darwin, who was credited with the assertion that an aging program, being counterproductive for the individual, would have been surely rejected by natural selection. However, in fact, Darwin recognized the existence of altruism among living organisms, which he believed had been created by natural selection [2]. In Darwin’s books there are no indications suggesting that aging cannot be one of such altruistic programs.
In 1881, A. Weismann issued a statement that death caused by senescence is an adaptation invented by natural selection specifically for alternation of generations, i.e. to accelerate biological evolution [3]. He was immediately accused of anti-Darwinism and in the end had to reject his point of view, which he nevertheless managed to present in his monograph widely cited to this day [3].
In the 126 years that have passed since Weissman’s famous lecture on the non-obligatory nature of death, his opponents have proposed many alternative hypotheses regarding aging as the result of the glaring imperfection of biological evolution. These hypotheses include accumulation of random DNA errors and physical or chemical deterioration of the organism; continuation of the process of ontogenesis after the completion of individual development; lack of vital resources, etc. [4-7]. The authors of such assumptions can be answered with the maxim by F. Crook that any biologist should be guided by a simple rule: evolution is always smarter than her or him.
This does not mean that, for example, wearing out cannot be a direct cause of organ dysfunction or even death of the organism. Teeth in elephants are changed five times during life, and new teeth grow when old ones are disrupted. It seems to be a clear example of an organ functioning till it is completely worn out. Nevertheless, nobody considers the question, what prevents the elephant from changing its teeth for the sixth time?
Or let us take another example: the protein crystallin of the eye lens of the bowhead whale lives as long as the whale, i.e. for more than two centuries [8]. During this time, part of the amino acids of the crystallin spontaneously change from L- to D-isomers. This process is the fastest in case of aspartic acid residues (40% D-isomers after 200 years), which should change the spatial structure of the protein and reduce its transparency for light rays. As a result, cataracts develop [8]. If the case of elephants is an example of the physical aging of an organ, then that of whales is a chemical one. As in the first case, you can ask why whales do not change the old crystallin by a new one at least once in 100 years?
For an unclear reason, vertebrate researchers are particularly uncompromising in their denial of the very possibility of programmed aging, although zoologists of invertebrates, botanists, mycologists, and microbiologists have long recognized this possibility [9]. Numerous cases of programmed death of an organism, which we call “phenoptosis” [10], in the world of invertebrate animals, plants, fungi, and unicellular eukaryotes and prokaryotes clearly show that various phenoptotic programs do exist despite their apparent counterproductivity for individuals [9-14]. Among phenomena of this kind, the phenomenon of “second-order selection”, described by Woods et al. at the University of Michigan, is particularly worth mentioning [15]. Comparing the rate of evolution and the survival of various Escherichia coli clones, they found that it is the bacteria whose offspring have “greater potential for adaptation”, rather than those that better adapt to current circumstances, that eventually become the winners [15].
Study of annual plants, soybean and thale cress, provided vivid examples of senile phenoptosis. Nooden et al. [16] showed in direct experiment that death of soybean plants after seed ripening was due to the transfer in the leaves of some compounds formed by the seeds. Death could be avoided by timely removal of the seeds, which turned soybean from an annual plant to a perennial. Lens et al. [17] and Melzer et al. [18] achieved the same result by knocking out two genes of thale cress (Arabidopsis thaliana) necessary for flowering and subsequent ripening of seeds. In this case, reproduction in the thale cress mutant was changed from sexual (seeds) to vegetative (rhizomes, which are not found in the wild-type plant).
The story of life and death of the female octopus Octopus hummelincki, described by Wodinsky, serves as an example of phenoptosis in invertebrates [19]. This animal loses its ability to feed and dies of hunger after small octopuses hatch from eggs. Up to this point, the female octopus eats normally and zealously protects its eggs from ocean predators. The mother’s death can be prevented by removing the so-called “optical glands”.
Sometimes phenoptosis is caused by the actions of one’s sexual partner, and not the individual per se. The female of one of the mantis species is known to bite off the male’s head at the end of coitus, which has been long seen as an example of the most sophisticated cruelty in the animal kingdom. This fact was explained when it turned out that the male mantis ejaculation occurs only after decapitation [11].
Paradoxically, in stable ecosystems, predator can play the key role in the victim’s phenoptosis. National Geographic has published a photo of a huge herd of buffaloes leaving the watering place. In the rearguard of the herd, one could see four lionesses. The first of the predators is just a few meters away from the last buffalo. These huge animals do not try to gore or trample their enemies, which are obviously intent on pursuing the herd. Such a tactic is understandable, given the known fact that a lioness never attacks a buffalo calf; it tracks adult animals weakened by old age, infections, or wounds, and cleans the herd from rearguard individuals that systematically restrain the herd’s pace and/or may be a source of infection for healthy buffaloes [20].
The situation described above for the lioness/buffalo pair is hardly a special exception. For example, we found in the literature a photograph of a herd of antelopes with a leopard quietly walking among the animals. The predator is carefully examining the antelopes, while they are calmly looking at the leopard and continue to move slowly in the opposite direction [21].
Successful evolution of a species cannot be imagined without “coevolution” of other species of the same ecosystem [22]. Mitteldorf and Sagan [22] discuss the case of the Rocky Mountains locust (Melanopsis spretus) as an example of a situation where the interests of ecosystem partners are violated. In 1874, the area affected by these insects constituted ~500,000 km2 (that is, of the same order as the entire area of California). Throughout this gigantic territory, nothing green was left after the locust invasion, and the soil was covered with a layer of eggs, ready to produce a new generation of voracious insects the next year. However, this generation already had nothing to eat, and it all died out. The authors wrote that this death occurred “not because individual locusts were not sufficiently “adapted”. It happened because these individuals were too aggressive and too prolific” [22]. Examples of moderation and considering the interests of other ecosystem members include long-lived birds (penguins, auklets, condors, griffins, eagles, albatrosses), which lay only one egg. If this single egg gets broken, the bird lays another egg, but never two at once [22]. According to anthropologist Carr-Saunders [23], populations of primitive humans that have maintained their number for many thousands of years used several ways to limit the growth of their population, from abortions to killing the newborns. M. S. Gilpin [24] describes cases when evolution of predators in the ecosystem is organized in such a way as to protect victims from their complete extermination by these predators. As Mitteldorf and Sagan write, “one cannot create a stable ecosystem of species whose individuals tend to grab as much as they can drag, and multiply as quickly as they can” [22].
Mechanisms protecting a population against infections using a simpler form of phenoptosis of infected individuals have been traced from bacteria to mammals. Bacteriophage-infected E. coli “commit suicide”, shutting down one of the vital biochemical systems such as protein biosynthesis or maintaining the insulating properties of the membrane. Suicide occurs long before the phage multiplies to such an extent that it disrupts bacterial vital functions and the infected E. coli becomes dangerous to its fellow bacteria as a source of infection [14, 25]. In mammals, a similar phenomenon is observed with sudden death of an infected individual, resulting from septic shock that occurs in response to appearance of fragments of the bacterial wall in the bloodstream [26-28].
There are mammals that multiply only once in a lifetime. For example, male Australian marsupial mouse dies a few weeks after the rut. It is killed by its own pheromones, which were previously used to attract a female. Both male and female vomeronasal organs have receptors for male pheromones, which are still produced after the completion of the rut. The pheromones somehow block control functions of the hippocampus in relation to the hypothalamus. As a result, the male is struck with severe stress due to a sharp increase in the level of corticosteroids and catecholamines. Castration of males or their maintenance in a vivarium separately from females significantly increases their lifespan: it reaches values characteristic for females [29].
Males of the South American small marsupial Gracilinanus microtarsus die immediately after mating, and females a little later, after the end of lactation (A. Vercesi, 2013, personal communication). Semelparous species have been described among amphibians, reptiles, and fish. All these cases can be seen as a way of accelerating evolution due to increased diversity of offspring. Obviously, diversity increases if an individual can become a father or mother only once [20]. In addition, the very fact of death shortly after childbirth dramatically shortens individual lifespan, thereby increasing the frequency of generation change (which exactly fits the hypothesis of Weismann [3]).
However, what about species that multiply repeatedly? Here, too, we can find cases of acute phenoptosis that suddenly terminates life when the organism reaches a certain age. According to Lecomte et al. [30], older albatrosses (living for over 50 years) nesting on the islands of the Indian Ocean fly in search of fish into waters of the Antarctic, while younger birds never cross the Antarctic Circle. These birds were observed with the aid of a satellite and radio signal sensors mounted on the animal’s body. They looked for some signs of aging of albatrosses, which no one had ever found in these birds, who do not have enemies and grow during their entire lives, thus dying at some point for an unknown reason. The increase in flight range with age Lecomte et al. interpreted as a finally found sign of aging, which, of course, contradicts the very definition of aging as age-related gradual weakening of vital functions, and not their strengthening [25].
WHY BIOLOGICAL AGING IS A LONG PROCESS?
For most classes of higher vertebrates, chronic (rather than acute) phenoptosis is typical, so the very process of aging takes many months, years, or even decades [10, 25]; it is either a result of a long-term program, or because of imperfection of biological evolution, chemical or physical deterioration of organs, which some biologists believe to be inevitable.
If gradual aging is chronic phenoptosis, then what is the biological meaning of such a long process counterproductive for the organism? It seems that accelerated alternation of generations, elimination of infected individuals or those who have lost their reproductive function due to age could be more easily achieved by acute phenoptosis. Weismann did not consider this issue. In 2003, one of the authors of this paper [25] proposed the following interpretation of this phenomenon – how slow aging could accelerate biological evolution. Consider the following situation. Two young hares, a smart and a stupid one, when meeting a fox, have almost equal chances to escape simply because they run much faster than the predator. However, with age, the smart hare will have an advantage over the stupid one, and this advantage may be decisive when the running speed of hares decreases due to aging and becomes similar to that of a fox. Now a smart hare, who, after seeing the fox, will immediately start running, will have much greater chance to survive than the stupid one, who will lose the first moments because of indecision. As a result, the hare population will grow wiser.
It is significant that the young (i.e. more numerous and intensively reproducing) part of the population will not participate in such an experiment, being a guarantee of stability of everything that has been already achieved by evolution. However, some small positive or negative signs (which cannot be recognized by natural selection in youth) will be presented to selection in the aging part of the population. As a result, if this property is useful, then it will be transferred to the offspring produced by the “old folks”. In addition, if the new property manifests unfavorable side effects potentially destructive for the species, it will not pass the selection screen, and the experiment itself will not have serious negative consequences for the species because most individuals reproduce at a young age (D. P. Skulachev, not published). Thus, it turns out that young individuals are mainly responsible for conservatism of heredity, and aging organisms for its variability.
Such an evolutionary strategy resembles a well-known feature of sexual dimorphism in many bird species, where the females are gray-haired, voiceless, and not at all inclined to “protrude” (“conservative”), and the males are motley, active, and clamorous, as if looking for dangerous situations (“revolutionary”) [31].
Increased diversity of individuals in a population might be another function of aging as a mechanism contributing to the evolutionary process. As J. Mitteldorf noted, it is impossible to select smaller or larger individuals if they are all the same height [22]. Aging leads to a change in the organism’s characteristics, and this process develops at a slightly different rate in different individuals, which inevitably results in divergence of signs that depend on age.
Mitteldorf points to yet another possible function of aging – creation of a cohort of weaker organisms that play the role of a “demographic buffer” under adverse conditions. Older individuals, being weaker and inferior to the young in their reproductive capacity, will be the first to take the blow of deteriorating conditions (for example, emergence of a new enemy), protecting for a while a healthy, strong, and rapidly multiplying core population [22].
All the listed above-described functions of aging can be useful, like everything in evolution, only for future generations. As for individuals of this generation, aging is certainly harmful for them. This generation, in fact, suffers “for the rattling valor of the coming centuries”, as Osip Mandelstam said.
In this context, it is important to note that such counterproductive for an individual characteristic as aging is essentially optional, facultative. Under critical conditions, the organism can turn it off or, at least, slow it down. Apparently, this is how temporary dietary restriction, perceived by an individual as a signal of danger of hunger, acts. In such a situation, the organism reduces the harm caused by aging and possibly, also some other counterproductive programs, which are inhibited by regulatory systems specialized in finding internal reserves to overcome crisis. Such resources can be mobilized to search for food. This search inevitably involves traveling large distances, i.e. serious muscular load. It is not a coincidence that the heavy work of skeletal muscles serves, along with dietary restriction, as another universal method to slow aging in a wide variety of animal species [20]. The following observation is quite remarkable. Mice kept in cages with a squirrel wheel ran a much longer distance in the wheel (~7 km instead of less than 1 km in the norm) if their diet was restricted. Some animals did not leave the wheel at all and died there [32].
NEOTENY IN NAKED MOLE RATS AND HUMANS
What is neoteny? The previous sections of our review allow us to conclude that aging can be considered as a program that is counterproductive for an individual, but useful for biological evolution due to increasing the pressure of natural selection. A prediction of this conclusion is that for species that have avoid the pressure of selection, aging loses its basic biological function and becomes a harmful atavism and a subject to cancellation. This cancellation could result from neoteny – delay of late stages of ontogenesis, leading to an increase in life expectancy due to prolongation of youth [33, 34].
For most living beings, the contribution of age-dependent mortality to total mortality increases exponentially with age. When the organism is young, input of this parameter is insignificant, but over the years it becomes the main cause of death. If aging is programmed as the last stage of ontogenesis, it can be postponed by slowing the ontogenetic process. Such a slowing is indeed described in some organisms under the name neoteny [33-36]. The most striking, classic example of neoteny is the ontogeny of one of the amphibians, Mexican salamander (Ambystoma mexicanum). Larva of the salamander, axolotl, seems to be stuck in its ontogenesis for many years (up to 32 years). Metamorphosis of the axolotl can be accelerated to days by adding thyroxine to the aquarium or by transferring the axolotl from the aquarium to a moist chamber [37, 38]. Essentially, the axolotl is more a fish than an amphibian. It breathes with external gills, swims with the help of a powerful caudal fin, and its small paws are poorly adapted to travel overland. It is striking that the axolotl is capable of sexual reproduction and is extremely viable due to its unflagging youth and the ability to regenerate any parts of the body except the head. The adult salamander is a typical amphibian, which can live not only in water, but also on land, with strong paws and lungs that develop instead of gills. However, the salamander lives not longer than 5 years [37, 38].
Proteus (Proteus anguinus) looks similar to the axolotl with its external gills, small paws, and some other features. It lives only in the water (in underground lakes in total darkness, which explains its blindness). Sixty-nine years is a record of its lifespan, established experimentally, and mathematical calculation predicts an even larger value – 103 years [39, 40]. It is remarkable that proteus never turns into an imago (even when it is removed for a while from the aquarium or thyroxine is added to the water) [41]. Another axolotl-like salamander, which has no adult form, has been discovered in North America. It is the American proteus (Necturus maculosus), which lives for over 29 years, growing to 40 cm. It reaches sexual maturity at 6 years and survives only in water [42-44].
Like the neoteny phenomenon in animals, it can also be observed in plants (for example, in a number herbs such as Sonchus and Echium). Their tree-like perennial forms grow on isolated islands, separately from small annual continental ancestors [17, 18, 33, 34].
Cases of neoteny have also been described in fish (bullhead [42]1), invertebrates (termites [46, 47]), mayflies, cicadas [20], crustaceans (Stylomesus hexapodus and Haplomesus corniculatus) [48], and jellyfish Turritopsis [49, 50]. The last case is particularly impressive. The juvenile form is a colonial hydroid (polyp), which is then transformed into solitary individuals, small jellyfish, capable of sexual reproduction. It is amazing, but single mature forms can return to the previous, earlier condition (polyp) in response to a physical damage or deterioration of external conditions, i.e. in an unfavorable situation the organism does not die, but “rejuvenates”. In mammals, neoteny was considered, in addition to humans and naked mole rats (see below), with reference to whales [51, 52].
1 Recently Nielsen et al. [45] reported that maximal lifespan of Greenland sharks Somniosus microcephalus is 392 ± 120 years, and the sexual maturation age is 156 ± 22 years. The fish grows during its whole lifespan and reaches more than 5 m in length. It is widespread in the North Atlantic. Its average “cruising” speed is 4 km/h. It eats the corpses of other inhabitants of the ocean sinking to the bottom. The constant growth rate (~1 cm per year) and significantly delayed sexual maturation could be the signs of neoteny, but this speculation requires more detailed study.
Neoteny of naked mole rats. A small African rodent, the naked mole rat (Heterocephalus glaber) is of interest in this context. Age-related diseases such as cancer and diabetes, as well as many cardiovascular and neurological pathologies, were shown to be rare in these mammals or even completely absent from causes of their death. Severe infectious pathologies are also very rare. As a result, mortality of naked mole rats under laboratory conditions is very low and almost independent of age, at least up to 25 years [53, 54].
Why do these mysterious animals die? In a laboratory with no threats of hunger or attacks by either snakes or aggressive neighbors, some naked mole rats can live for over 30 years. Laboratory observations of the maximum lifespan of these rodents have not been ended, but it is likely that this age will be exceeded [53, 55]. Mice are in many respects like naked mole rats: both animals are small rodents of almost the same size and have generally rather close structure. However, mice live for not longer than three years, and by the end of this time they manifest many signs of aging: baldness, spinal curvature, loss of ability to reproduce, etc. Naked mole rats live at least ten times longer than mice, and their fertility only grows with age. In a colony of naked mole rats (up to 300 individuals), only the royal family is involved in reproduction: the queen and her husbands (one to three males).
Note that queens of eusocial insects (bees, wasps, and ants) have a much longer lifespan than their subordinates [56]. As for the naked mole rats living in a vivarium, not only the queen and her husbands, but also subordinates die very rarely. Apparently, their death in captivity is mainly caused by collisions with relatives, which are sometimes fatal [57].
Another difference between naked mole rats and social insects is that in the case of naked mole rats, the queen and her husbands do not initially have morphological differences from their subordinates. A difference develops over the years: the queen becomes larger than her female subordinates. Her vertebrae and some other bones irreversibly elongate during pregnancy. After the queen’s death, her place is usually occupied by the strongest female previously subordinate to the queen. The striking difference between naked mole rats and mice is the monopolization of the reproductive function in the naked mole rats by the queen and her husbands, who are well protected by a large group of subordinates from adverse effects of external factors.
Absence of enemies is also a common feature of many other animals with negligible aging [58]. Sea urchins are protected by poisonous spines; toads by cutaneous glands secreting poisonous substances; large crabs and turtles, as well as oysters and mussels by strong shells; large predators, such as sharks, large birds, and mammals are armed with sharp teeth or powerful beaks and claws; very few natural enemies are a real threat to giant whales. Like those species, naked mole rats in the wild encounter very limited pressure of natural selection. These small highly social animals are divided into two unequal groups: several privileged individuals that participate in reproduction (and, consequently, in natural selection and other evolutionary processes), and the vast majority of other members of the community that do not breed and do not participate directly in natural selection.
When we say that animals with negligible aging do not have enemies that significantly affect their mortality, we do not assume that protection against enemies would cause immediate increase in the animals’ longevity. Any animal in the laboratory does not have problems with predators or food, but it usually continues to age. Probability of death of laboratory mice steadily increases with age, in contrast to naked mole rats observed under the same laboratory conditions. Apparently, transformation of short-lived organisms into long-lived one requires a very long time, like all other events associated with biological evolution [59].
In 1991, Alexander applied the term “neoteny” to naked mole rats [60]. In 2009-2014, studies of neoteny in H. glaber were continued by Larson, Park, et al. in Chicago [61, 62]. They showed that the resistance of neurons in the hippocampus of adult naked mole rats to hypoxia/reoxygenation is much higher than in other studied adult mammals, and it resembles that of neonates (see also [63]). They described the molecular mechanism of this stability, which is at least partially associated with one of the subunits of NMDA receptor (glutamate), namely GluN2D, which is characteristic of adult naked mole rats. In mice, high level of GluN2D is found only in newborns. The number of this subunit gradually decreases with age. GluN2D-mediated membrane permeability for cations is more specific (only Na+ and K+), whereas in GluN2 A, B, and C it also includes Ca2+. As a result of hypoxia, ATP is depleted, since oxidative phosphorylation, the main mechanism of ATP synthesis for brain cells, is turned off. In anaerobiosis, Na+/K+ gradients are not formed because of the deficiency of ATP, leading to long-term depolarization of the neuron after excitation. In the absence of GluN2D, the situation becomes even more acute, because the gradients of not only Na+ and K+, but also Ca2+ are no longer maintained, and intracellular level of Ca2+ greatly increases. In the end, neurons die by apoptosis initiated by Ca2+.
The phenomenon of high degree of resistance of the brain to hypoxia and oxidative stress is an indispensable feature of newborn mammals. In other animals, except for the naked mole rat, this resistance greatly decreases with age (to a level 25 times smaller than that observed in newborns), which was originally described by Boyle in 1725 [64] (see also [65-67]). Larson and Park explained these relations within the framework of the hypothesis of neoteny [61, 62].
In 2015, the groups of T. Harkany and E. Keimpema in Stockholm and Vienna, cooperating with T. Park and D. Larson in Chicago, published an article [68] that describes prolonged delay in the development of brain parameters in naked mole rats. They found that neurogenesis and neuronal migration are inherent in the brain of not only newborns, but also of adult naked mole rats (see also [69]). In addition, prolonged expression of structural plasticity markers and long-term postnatal morphogenesis of neurons, as well as spatial changes in the position of synapses, were found in hippocampus and olfactory zones of this species. Those authors concluded the following: “Naked mole rats show an extremely long period of maturation of the brain, which can provide plasticity and resistance to neurodegenerative processes during their long life. This conclusion is consistent with the hypothesis that naked mole rats are characterized by neoteny with the preservation of the characteristics of adolescences” [68].
Those authors interpreted their findings as being due to the adaptation of naked mole rats to their long underground life under hypoxic conditions. As will be shown below, similar immature brain structures are also present in humans where the hypoxic hypothesis cannot be applied. This important circumstance is not mentioned in Harkany et al. [68]. We explain neoteny of naked mole rats compared to mice, as well as neoteny of humans compared to chimpanzees, by specific social organization of naked mole rats and humans, which made aging as a mechanism for increasing the efficiency of natural selection useless simply because the very significance of such selection was drastically decreased for these two species as a result of their evolution.
In a paper by Harkany et al. [68], the hypothesis of neoteny as the reason for the longevity of naked mole rats is supported by the description of only six signs of ontogenesis, which is slowed during brain development compared to mice and rats. L. E. Bakeeva, M. Yu. Vyssokikh, et al. in our group compared several mitochondrial properties in naked mole rats and other small rodents. They found that: (i) fusion of small mitochondria into a single mitochondrial reticulum developing in skeletal muscles of rats by the third month after birth, is still not formed in three-year-old naked mole rats [33, 34, 70]; (ii) low content of adenine nucleotides in heart mitochondria of newborn mice, which rises to normal value shortly after birth, remains low in five-year-old naked mole rats [33, 34, 70].
Respiration in heart and liver mitochondria in adult naked mole rats, activated by addition of ADP in vitro, only partially decreases after depletion of the ADP [33, 34, 70], which resembles the mitochondria of rat embryos and newborn rats [71]. In adult rats and other mammals, respiration prior to ADP addition and after ADP depletion is almost identical [71].
In the paper in Physiological Reviews [34], we compared these data, published in 2015-2016 [70, 72], with the results of Park, Larson, Harkany, Keimpema et al. on the brain [61, 62, 68] and with many observations of other researchers of naked mole rats accumulated over the past 20 years. More than forty different signs of late stages of ontogenesis have been discovered, the development of which was either strongly delayed or completely blocked in these animals. Among them, along with the well-known absence of pelage we note the absence of external auricles and scrotum, much smaller (than in related rodent species) body weight, prolonged pregnancy (70 versus 21 days in mice), longer lactation period, prolonged puberty (at least 7 versus 1-1.5 months in mice), later opening of the eyes (on the 70th instead of 13th day in mice), severe retardation of age-related morphological changes in the structure of newborn bodies (in naked mole rats they develop only two weeks after birth, while in mice in the first days; during the first 80 days, growth and development of naked mole rats are significantly slowed).
In addition to delayed brain development, naked mole rats are characterized by delayed formation of the skeleton, thermoregulation system, lungs, and vomeronasal organ. In the last three cases, these systems of the naked mole rats never reach the “adult” morphological and functional level characteristic of other rodents. However, many positive features of youth do not disappear as naked mole rats grow up. We have already mentioned the great resistance of this mammal to diseases characteristic of advanced age. Add to this the total resistance to pain caused by capsaicin or acid, which is due to the lack of substance P in naked mole rats. Many other signs of aging are also absent in these animals: age-related decrease in elasticity of blood vessels, bone mineral density, and overall rate of oxygen uptake by the organism. There is no increase in the lipid peroxidase index reflecting their sensitivity to reactive oxygen species (ROS). ROS level does not increase with age, and there is also no decrease in (i) the activity of the most important antioxidant enzymes (catalase and superoxide dismutase), (ii) number of proteasomes, and (iii) glucose tolerance, and no increase in glycation of hemoglobin [33, 34].
Thus, the available literature and our own data on mitochondria from different naked mole rat tissues seem sufficient for a conclusion about the neoteny of these animals.
Neoteny in humans. High social organization is characteristic for at least one other mammal, which is surely the most interesting for us. This is Homo sapiens. Like naked mole rats, social organization of people results in their escape from the pressure of natural selection. However, human beings, in contrast to naked mole rats, chose not “aristocratic”, but “democratic” way of life. Our society is not divided into a very small multiplying minority and huge, obedient majority, deprived of this right. Instead we invented technological progress, which is accessible to all the members of society. When we needed to fly, we invented the plane, without waiting for wings to grow on our backs. As a result, aging as a way to accelerate natural selection became as unnecessary for people as for naked mole rats. And, similarly to naked mole rats, humans tried to get rid of aging, following the same path of neoteny, i.e. searching for individuals who have slowed late ontogeny and thereby prolonged youth and delayed senescence.
The famous Dutch anthropologist L. Bolk was the first to compile a list of signs of slowing late ontogenesis in humans (in comparison with monkeys). He published two works on this subject in 1926-1927 [73, 74]. Even a shortened list embraced 25 features, including the absence of continuous hair-covering (as in naked mole rats), retention of infantile skull form without superciliary arches or crista until old age, shorter limbs compared to body size, small teeth that crown relatively late in life, very long period of childhood, later puberty, lifespan that is the longest among primates, etc.
Further research of Bolk’s followers greatly expanded his list of signs of human neoteny. However, few cases when certain features of late ontogeny developed in humans faster than in chimpanzees have been found. Much faster growth rate of the human embryo’s brain is the most striking example [75]. As a result, a mosaic pattern has developed that is less harmonious than in the case of the axolotl. As for Bolk with his list of neotenic human features, he began to be perceived as excessively revolutionary. However, if you ponder the essence of the matter, there is nothing surprising in the mosaic nature of neoteny. Slowing of all signs of late ontogeny could lead to failure. Some of these features should have been rather accelerated than slowed, as in the case of the creation of the unique brainpower apparatus of H. sapiens. Such an apparatus could hardly have been constructed if the human brain’s growth had been significantly slowed already at the embryo stage, as in chimpanzees [75].
Let us note that certain mosaic pattern in the development of organs and physiological systems is characteristic also for naked mole rats. Here lungs, brain, skeletal muscles, skeleton, eye formation, and sexual maturation demonstrate inhibition of ontogenetic development, and hair covering, scrotum, and external ear do not develop even by the age of 30 years. However, the heart, liver, and kidneys apparently develop without a strong delay and do not show signs of neoteny.
Returning to human neoteny, it should be emphasized that the common ancestors of humans and chimpanzee probably looked and behaved more like humans than like modern monkeys [76, 77]. Humans have lost some physical strength (for example, a gorilla is physically 15 times stronger than a human [36]). At the same time, embryos and cubs of chimpanzees remind much more of people than of adult primates. This is also true for the structure of the skull [33, 34, 78]. It seems that juvenile anthropoid monkeys become more animal-like with age. This is not the case with people who, as C. Bromhall put it, remain “eternal children” [79, 80].
An important physiological and genetic discovery was made in 2009-2012 in the works of Khaitovich et al. [81, 82]. They analyzed the transcriptome in the prefrontal cortex of humans, chimpanzee, and rhesus macaques. The transcriptome has been shown to undergo significant reconstruction in the postpartum period, and in humans this reconstruction is strongly delayed in comparison to monkeys. Such delays have been found for many genes that are involved in the development of neurons, including genes encoding synaptic proteins (see also [83]). It is noteworthy that delays in the development of the human transcriptome occur even though human pregnancy lasts longer than that of chimpanzees, and the human embryo’s brain grows much faster. It should be emphasized that a significant delay in the development of the human brain compared to its development in monkeys resembles a similar phenomenon in the naked mole rats compared to mice, as described by Penz et al. [68]. However, the brain size of adult naked mole rats is like that of a mouse [77, 84], while the brains of adult humans are much larger than those of chimpanzees [75].
NEOTENY-BASED LONGEVITY OF NAKED MOLE RATS AND HUMANS SHOWS THE
PROGRAMMED CHARACTER OF AGING OF THESE TWO MAMMALIAN SPECIES
This subtitle is explained by the following simple and clear logic: (i) neoteny is a slowing of the development of features programmed in late ontogeny; (ii) aging of naked mole rats and humans is one such feature. Hence, aging, being part of the ontogenetic program, is also programmed.
We predicted this based on the hypothesis of aging as a mechanism invented by biological evolution to accelerate this evolution. Clearly, such a mechanism is effective only if natural selection works. This last requirement is not fulfilled by naked mole rats. Here, the “aristocrats” monopolized the childbearing function, being protected from natural selection by an entire army of non-breeding subordinates, who build at half-meter depth a labyrinth measuring up to two football fields with the apartment for queen and her husbands in its center, carefully guarded by the subordinates. Comparison of the naked mole rat with its two close relatives from the Bathyergidae family, namely, with the Damaraland mole rat (Fukomys damarensis) and silvery mole rat (Heliophobius argenteocinereus) is quite demonstrative. As mentioned above, H. glaber is a highly-social animal that organizes numerous colonies. Silvery mole rats have no social structure; they live a solitary life. Damaraland mole rats occupy an intermediate position. Their social organization resembles a rather large family, which includes a breeding couple, i.e. male (who is the head of the family), and his wife and their children of different ages who have not reached puberty yet. Such families usually include ~15 members (up to a maximum of 40). All three species are underground African rodents. They differ greatly in maximum life expectancy: over 31 years in case of H. glaber, ~16 years for F. damarensis, and ~7 years for H. argenteocinereus. The two most impressive neotenic features of naked mole rats (their small size and lack of hair) are absent in F. damarensis and H. argenteocinereus: both are covered with thick fur and weigh, respectively, ~140 and 240 g (H. glaber < 40 g) [85-88].
We have already emphasized that the number of subordinates in the naked mole rat colony, who serve and protect the few reigning animals, is so large that the pressure of natural selection on the queen and her husbands is greatly reduced. This means that aging as a mechanism contributing to natural selection becomes a harmful atavism for this animal. As a result of evolution, H. glaber was probably able to slow aging by means of neoteny. This did not happen with F. damarensis or H. argenteocinereus, since they both have a much smaller number of subordinates or none at all. These two species do not show neoteny and therefore they are not so long-lived.
We see a similar situation when comparing human beings with anthropoid apes. With the invention of speech, writing, and then technical means of communication, humans have greatly expanded the availability of useful information to all members of society (and today, after the invention of the Internet, to all humankind). All this made the rate of technical progress of humankind incomparable with the snail’s steps of biological evolution. This circumstance practically abolished the essential role of natural selection for H. sapiens. Therefore, spontaneous neoteny as a way of further increasing the life expectancy of a modern person is such a slow process (for the naked mole rats it probably took over 30 million years [89-91]), and human beings can hardly operate with such long periods of time when thinking about future.
When comparing lifespans of humans and monkeys, scientists often appeal to the maximum values of this parameter. But in our case, this criterion has two major drawbacks. First, the sample for humans is many orders of magnitude greater than for monkeys. The Jeanne Calment’s record (122 years old) is the result of measuring lifespans of many millions of people. For chimpanzees, the sample is smaller by many orders of magnitude. Second, the goal of modern gerontology is not so much to improve the Calment’s record (which will concern one or, at best, several people on Earth), but to increase the mean or median lifespan (i.e. the lifespan, when the number of surviving individuals decreased by 50%). The median lifespan of Japanese women who died in 2009 was 82 years, and that of chimpanzees – 18 years [92]. The same values for the naked mole rat and mouse under the laboratory conditions were found to be 22 years [54] and 2 years [93], respectively.
SURVIVAL IS THE RESULT OF TWO PROGRAMS: AGING AND ANTI-AGING. THE
ROLE OF DIETARY RESTRICTION AND MELATONIN
The main regulatory biological programs are characterized by the presence of two opposing systems. For example, the animal blood coagulation system is counterbalanced by the anticoagulation system. Such relationships facilitate regulation of processes controlled by the organism. Aging is apparently not an exception to the general rule.
It is well known that dietary restriction extends the lifespans of a wide variety of organisms from unicellular fungi to humans. In this case, it is not a question of complete starvation, but of a 30-40% decrease in the “caloric value of food”. This term turned out to be erroneous, at least for mammals. It has been shown that a decrease in the amount of one essential amino acid, methionine, causes the same geroprotective effect as a reduction in the intake of all twenty amino acids simultaneously [94-98]. In all likelihood, the organism monitors the intake of protein food by the blood level of methionine. Reduction of this parameter is perceived as a signal of advancing “lean years”, and the organism takes measures against starvation. One of the measures is to disable facultative counterproductive programs such as aging, useful for the evolution of future generations, but harmful to the individual who is currently starving. For a population whose survival is threatened by hunger, the momentary benefit is more important than all the other tasks it faces [20]. To get this benefit, the organism does not require anything other than stimulation of its own anti-aging program that can temporarily (for the period of searching for food) eliminate the harm inflicted by the aging program.
The hormone melatonin, 80% of which is formed in the pineal gland of our brains, is one candidate for the role of the anti-aging program mediator. Melatonin is connected to the circadian rhythm of metabolism [99]. It is due to the circadian rhythm that cells organize their metabolism in a way optimal for a given time of day or night. The peak of melatonin content is observed at midnight. The height of this peak decreases with aging of the organism [99-101], and dietary restriction prevents such a decline [102]. Very recently, it was found that circadian rhythm of the transcriptome of liver, muscle, and epidermis cells changes drastically in aging compared to young mice. In case of young mice, oscillation in the activity of genes that provide homeostasis was observed, while in old mice this phenomenon concerns genes that prevent the development of stress. Dietary restriction in old mice sharply changes this picture making it closer to that of young animals [103, 104]. Apparently, in the case of naked mole rats with their neotenic delay in the aging program, the anti-aging program becomes unnecessary. Indeed, according to data of V. Gladyshev et al., the genes of the two main melatonin receptors are mutated in this animal [105]. W. Quay reported that epiphysis is generally atrophied in naked mole rats [106]. This observation was confirmed in the laboratory of V. Gladyshev and in V. Manskikh’s group in Moscow State University (private communications)2. It has been shown that only one of the melatonin receptor genes is damaged in Damaraland mole rats [105]. Activity of two enzymes necessary for melatonin synthesis from serotonin is extremely low or absent in the brain of naked mole rats [105]. Melatonin and regular circadian rhythm have been discovered in other rodents genetically related to naked mole rats that also live underground but are devoid of eusocial organization [108].
2 For the influence of pre-aging pinealectomy on lifespan of mice, see the work of W. Pierpaoli and D. Bulian [107].
MITOCHONDRIAL REACTIVE OXYGEN SPECIES AS INTERMEDIATES OF THE
AGING PROGRAM
SkQ1 and aging. There are numerous data indicating that mitochondrial reactive oxygen species (mROS) act as mediators of an organism’s aging (see reviews [10, 20, 109, 110]). Therefore, specific mitochondria-targeted antioxidants can be considered as potential medicines to combat aging. Studies of cationic plastoquinone derivatives (SkQ1) [20, 111-114] and the mitochondria-targeted key antioxidant enzyme catalase [94, 115-117] are promising. In mammals, both SkQ1 and targeted catalase cause: 1) prolongation of mean lifespan, and 2) delay in the development of numerous signs of aging, including many age-related pathologies. In particular, age-related diseases of human eyes are successfully treated by SkQ1 [20, 118-120]. Very recently, H. T. Petrie, P. Rabinovitch et al. [121] showed the age-dependent thymus involution to be inhibited by targeted catalase or high doses of non-targeted antioxidants, N-acetylcysteine (NAC) and ascorbate. Previously, a similar effect was observed in our group in response to SkQ1 administration at doses 105 times lower than those of NAC or ascorbate [122]. Quite recently, J. A. Enriquez and colleagues [123] showed that combining the nucleus from one strain of mice with mitochondria from another strain dramatically prolonged the healthy life of the animals. These effects were accompanied by complete suppression of the 4.5-fold age-related increase in mROS found in animals with nuclei and mitochondria from the same strain. The list of age-related features that disappeared in the hybrid mice coincided with those caused with SkQ1 [20, 114].
Blockade of acute phenoptosis by SkQ1. Many cases of acute phenoptosis discussed at the beginning of this review can be seen as “grimaces of sexual reproduction”, when the death of one and especially both parents after mating (males) and birth of offspring (female) stimulates evolution due to increased likelihood of diversity in this progeny. Another example of altruistic suicide is when one of the parents sacrifices himself or herself, saving the offspring. Yet another type of this phenomenon is self-destruction of the infected individual, so as not to become a source of infection in the family, community, or population. In such a case, the organism seems to follow the replica of the unlucky doctor Bahys in Moliere’s comedy “Dr. Cupid”: “It is better to die by all the rules than to recover against the rules!”
In medical practice, there is the concept of sudden death following a crisis. It happens when the critical situation is over, and nothing seems to foreshadow a tragic denouement. It is possible that here we are also dealing with acute phenoptosis. But what could be its evolutionary meaning?
Some time ago one of the authors of this article formulated a principle called the “samurai” law of biology: “It is better to die than to make a mistake” or in the expanded form: “Complex biological systems are equipped with programs of self-elimination that are activated when this system becomes dangerous for any other system that occupies a higher position in the biological hierarchy” [125]. The above-mentioned sudden death is one of the consequences of this law: it can be argued that any critical state of the organism, when it no longer guarantees safety of its genome in case of recovery, can produce offspring with a damaged genome, and this is a signal for suicide [25, 124, 125].
It was interesting to learn whether mitochondrial ROS (mROS) are used as an intermediate in the mechanism of acute phenoptosis, the way it happens in the mechanism of slow phenoptosis (aging)3.
3 In this regard, one could note the observation of Matsuzaki et al. [126] that food limitation not only decelerates aging of mice (slow phenoptosis) but also prevents bacterial lipopolysaccharide-induced septic shock (acute phenoptosis). Apart from lipopolysaccharide, acute phenoptosis can also be caused by formylmethionine – an amino acid located at the N-terminus of bacterial proteins. Because many mitochondrial proteins are organized based on the same principle as bacterial proteins, mitochondrial debris, entering the blood flow in case of extensive wounds of soft tissues, causes septic shock [127]. Influenza virus acts in a similar manner, and its inactivation by boiling does not eliminate its lethal activity. In all these cases, the septic shock cascade is triggered by the same mechanism, the key component being the product of peroxidation of polyunsaturated fatty acids in phospholipids [128, 129].
D. Zorov and his colleagues in our group chose rat kidney infarction as a model for checking the possibility of the involvement of mROS in sudden death after crisis. One kidney was removed from the animal, and the other was deprived of blood supply for 90 min. This operation resulted in severe renal failure: blood levels of creatinine and urea were sharply increased, glomerular filtration and resorption of calcium ions were reduced. By the seventh day, only 30% of rats survived. Most animals died, and 50% of all deaths occurred on the 2nd-4th day after the operation, when the markers of kidney failure were already decreasing and, it would seem, the organism was recovering. A single injection of SkQ1 or its rhodamine analog SkQR1 (1 µmol SkQ per kg of weight) on the eve of surgery completely prevented mortality [112, 130-133].
Further experiments conducted in the group of D. Zorov by E. Plotnikov and his colleagues have shown the beneficial effect of SkQR1 on the course of such renal pathologies as pyelonephritis, rhabdomyolysis, and poisoning with the antibiotic gentamycin [132-136]. It was found that under certain conditions SkQR1 can be replaced by C12R1, which does not contain the plastoquinone group. Study of the mechanism of the antioxidant SkQ1 effect revealed that in small amounts it acts as a quencher of free radical intermediates of cardiolipin peroxidation, and in slightly higher concentration as a mild uncoupler that stimulates fatty acid protonophoric cycle at the stage of transmembrane transport of the fatty acid anion [137]. Later it was found that decylrhodamine-19, which is part of SkQR1, and its analog C12R1, serve as cationic protonophores even in the absence of fatty acids [138]. This suggested that in cases of C12R1 therapeutic effect, it can be replaced by a conventional protonophore used in small concentrations. The experiments confirmed this hypothesis (dinitrophenol was taken as the uncoupler) [132]. In this connection, we need to mention the prolongation of life of drosophila [139] and mouse [140] with small doses of dinitrophenol.
The groups of O. Pisarenko and N. Isaev [112, 141, 142] have studied other ischemia/reperfusion models that were not relevant to renal pathology, namely experimental myocardial infarction and stroke in rats. Favorable effect of SkQ1 or SkQR1 was observed in both cases. These phenomena were not related to aging, since they were obtained on young animals. We assume that in these cases SkQ blocked acute phenoptosis induced by the general crisis of the organism.
Short-term hypoxia is known to be dangerous not so much in itself, but because of the subsequent reoxygenation of the tissues. It is at the beginning of reoxygenation that the most dangerous reactive oxygen species, OH˙ radicals, are primarily formed. If there is no oxygen, ROS are not generated, but Fe3+ is reduced to Fe2+ and Cu2+ to Cu+, and this is necessary for nonenzymatic formation of OH˙ from H2O2. O2 is, in turn, the predecessor of H2O2; its content is very low in hypoxia, and it is simply absent in the state of anoxia. With the start of reoxygenation, i.e. with the appearance of O2, the respiratory chain starts to produce superoxide (O˙ˉ), which then turns into H2O2. In the presence of Fe2+ or Cu+, H2O2 immediately produces OH˙. In the new aerobic conditions, the reserves of Fe2+ and Cu+ are soon depleted, and after a few minutes of reoxygenation the danger of oxidative stress drops sharply. It is important to survive during these minutes when O2 has already appeared, and the reduced iron and copper ions are still available [20].
A beautiful experiment was recently reported by Chouchani et al., who studied the transition from hypoxia to reoxygenation [143]. A few minutes before reoxygenation, they administered to the animal the penetrating cation triphenylamylphosphonium, connected to one of the reversible modifiers of protein SH-groups. This procedure sharply reduced the area of myocardial infarction, inhibiting the activity of complex I in the first minutes of reoxygenation. This inhibition of complex I activity disappeared after 30 min of reoxygenation, when the administered dose of the modifier had decomposed. However, it was enough to reduce H2O2 production in the mitochondria at the beginning of the reoxygenation, which led to a favorable therapeutic effect. This experiment directly showed that infarction is caused by mitochondrial oxidative stress, apparently mediated by reverse electron transfer in complex I. This means that mROS are obligatorily involved not only in slow phenoptosis in the course of aging, but also in acute phenoptosis under the conditions of myocardial infarction.
Interesting favorable effect of SkQR1 was found in joint experiments of the group of V. Skrebitsky at the Institute of the Brain (Moscow) and the group of N. Isaev in our Institute [144, 145]. β-Amyloid (peptide involved in brain aging and pathogenesis of Alzheimer’s disease) is known to disrupt the mechanisms of memory at the hippocampus level. This effect can be modeled on hippocampus slices. Excitation of hippocampus cells by a series of electrical impulses leads to a steady increase in ΔΨ on the membrane of these cells. ΔΨ remains increased for tens of minutes (the phenomenon of “long-term potentiation”). Incubation of hippocampus slices with β-amyloid for 15 min prevents long-term potentiation. A single SkQR1 injection one day prior to the experiment was shown to completely remove the adverse β-amyloid effect. SkQ1 [144] or MitoQ [146] added directly to hippocampal slices have similar effect. We are now studying the mechanisms of this new effect of SkQ and the possibility of its application in the treatment of Alzheimer’s disease4.
4 Some experimental results suggest an important role of mitochondrial ROS in progression of this still incurable disease (see review [147]). Du et al. [148] showed that β-amyloid facilitates opening of the mROS-induced mitochondrial pore via interaction with cyclophilin D (remember that mitochondrial ROS are known to induce this pore, and its opening causes rupture of the outer mitochondrial membrane and release of cytochrome c to cytosol, which triggers a cascade of events resulting in cell death). It appeared that brain mitochondria devoid of cyclophilin D generate less ROS and do not swell after addition of β-amyloid. Mice devoid of cyclophilin D in brain suffered less memory loss and did not lose the ability for long-term cell potentiation in hippocampal slices under in vivo increase in β-amyloid level.
Hypothetical scheme of aging program of mammals. We have already discussed the cases of fast programmed death of the organism as the final event of ontogenesis, when death occurs so rapidly that its meaning cannot be explained by the stimulation of Darwin’s dynamic selection within the fable of the fox and hares (this framework requires the inclusion of prolonged chronic phenoptosis). However, acute phenoptosis combined with chronic phenoptosis could play a positive role as one of the mechanisms of Schmalhausen’s stabilizing selection [149], designed not to look for something new and useful, but to preserve what has already been achieved. The organism tries to put under its control as many as possible of processes occurring in its body. Therefore, the most crucial question of life or death could not remain uncontrolled (see above “the Bahys rule”). Incidentally, the elderly, with their reduced level of the immune system, are much more frequent objects of infection than young individuals and can therefore become its distributors. The prospect of mutations among the children of elderly parents is not less terrible. In this case, fathers are more dangerous than mothers, protected by menopause from childbearing in old age. Perhaps that is why men who, do not have this protection, do not live as long as women. Finally, individuals weakened by old age restrain the movement of their flock or herd and can therefore be subject to liquidation.
All these considerations point to the possibility that the program of slow aging (chronic phenoptosis) eventually leads to an acute phenoptosis. But who gives the sinister command and how is it carried out? Apparently, the organism should possess some system that controls the key parameters of homeostasis. When these parameters continuously cross the limits of some “epsilon-neighborhood” of the norm, it should be the signal enabling acute phenoptosis. Based on SkQ1 experiments described in the previous section, we suggest that the mechanism of acute phenoptosis in mammals can be qualitatively similar to that of chronic phenoptosis, namely, mediated by mROS which in this case are formed suddenly, at a high rate, when the antioxidant defense system is significantly suppressed. The fact that SkQ can save rats from experimental acute phenoptosis opens an important prospect of saving people from sudden death not only in old age, but also in catastrophes, heavy operations, etc. It is important that rats subjected to a short-term cessation of kidney blood supply were not only rescued by a single SkQ injection, but “lived happily ever after”, i.e. without visible deterioration of their health. Apparently, SkQ allows to avoid a crisis that causes programmed death of the organism in response to a negative signal of the system that controls homeostasis.
The well-known Australian veterinarian P. Britten has been using SkQ1 for 5 years as a remedy against the sudden death of dogs and cats. In his opinion, timely administration of SkQ1 helps to avoid the death of old animals that have already lost the ability to move (about 70 such cases have been described already). As in the experiments of D. Zorov and colleagues, overcoming the momentary crisis can prolong the animal life by many months and even years.
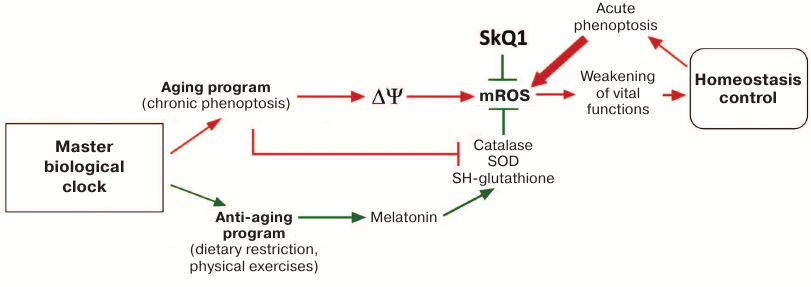
The above hypothesis can be illustrated by the following figure. The figure shows a Master biological clock [75a, 150]. Its nature is unknown, but it is clear that the process of ontogenesis, in some cases requiring years, cannot operate without some year-counting device. It has already been shown that the clock estimating the time of day in mammals is located in the suprachiasmatic nucleus of the hypothalamus, and it is even quite clear how it works. The clock counting groups of months (seasons) is probably also located there [151].
Aging program and some other regulatory systems that determine lifespan and its quality in humans and higher animals. Abbreviations: ΔΨ, the difference of electrical potentials on the inner mitochondrial membrane; mROS, mitochondrial reactive oxygen species; SOD, superoxide dismutase; SkQ1, regenerable mitochondria-targeted antioxidant; →, activation; ┴, inhibition. Biological aging is assumed to include chronic and acute phenoptosis caused by mitochondrial ROS (red arrows). It is countered by an anti-aging program or an artificial geroprotector (green arrows)
Once neoteny had been shown to cause longevity in naked mole rats and humans, it became clear that aging is one of the programs of late ontogenesis [33, 34]. It is based on the slow poisoning of the organism with the self-formed poison. There are good reasons to believe that it is mROS that play the role of this poison. The highest rates of mROS production are observed during hyperpolarization of the inner mitochondrial membrane, when the difference of mitochondrial electrical potentials (ΔΨ) reaches its maximum [110, 120]. It remains unclear how exactly the aging program raises ΔΨ and whether some other mitochondrial processes that increase ΔΨ or form mROS independently of ΔΨ are involved in this stage. For example, it has been shown that the protein p66shc interacts with cytochrome c in such a way that the complex of two proteins starts to reduce O2 to superoxide. Knockout of the gene encoding p66shc dramatically increases the lifespan of mice [152-155].
The aging program causes a decrease in activity of catalase, mitochondrial superoxide dismutase (SOD), as well as enzymes that provide the reduction of mitochondrial glutathione, the main low molecular weight antioxidant of the mitochondrial matrix. This effect is mediated by the age-related decrease in the level of SIRT3 protein deacetylase, which stimulates mitochondrial antioxidant enzymes [156].
The prooxidant aging program is counterbalanced by the antioxidant anti-aging program. The latter program is designed to slow (and maybe even cancel) signals of the aging program. It is activated under conditions critical for the survival of the individual, to interrupt the process of organism self-poisoning by reactive oxygen species formed in its mitochondria.
Thus, the interests of evolution, i.e. future generations, are sacrificed for the sake of the immediate interests of the individual. However, this is a transitory measure that, as any temporary tactical maneuver is, does not change overall strategy. For example, dietary restriction, perceived by the organism as a sign of impending hunger, may act as a signal to activate the anti-aging program. The epiphysis hormone melatonin can serve as a mediator in this signal transfer. In “obese years”, the anti-aging program can serve as a guarantee against progeria, when the aging program for some reason becomes hyperactive. ROS are poisonous and can cause a chain reaction of lipid oxidation. It is reasonable to have a system capable of quick “extinguishing the fire”.
The figure provides an explanation of the paradox of the naked mole rats, who have very high resistance to ROS. At the same time, ROS level, being the same in young and old naked mole rats, is significantly higher than in other rodents in youth. Due to neoteny, naked mole rats significantly slowed the late stages of ontogenesis, so that the activation of aging program is strongly retarded. Under such conditions, the anti-aging program became unnecessary and atrophied, the epiphysis disappeared, genes of melatonin receptors were damaged, and the transcription of melatonin synthesis enzymes slowed or stopped [105]. The level of ROS in young naked mole rats has somewhat increased due to the shutdown of the antioxidant anti-aging system, but it does not increase with age, as the aging program is retarded.
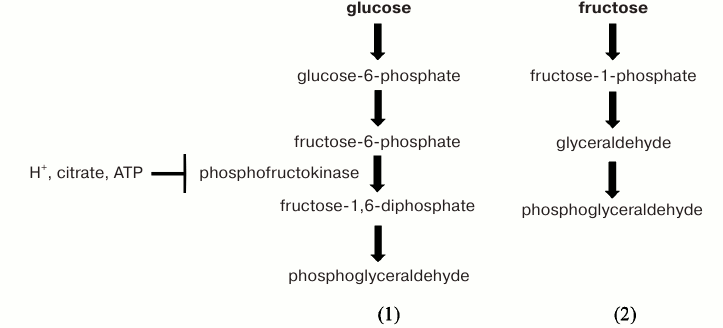
There is yet another reason for the elevated ROS level in naked mole rats: these animals cleave a significant portion of carbohydrates using not the glucose (reaction 1) but rather the fructose (reaction 2) pathway [157]. This second pathway, widely represented in embryos (especially in ungulate mammals [158, 159]), allows bypass of the phosphofructokinase stage, which is obligatory in glucose splitting (see Scheme).
Scheme
Phosphofructokinase is the enzyme responsible for the Pasteur effect, i.e. inhibition of aerobic glycolysis by respiration. Glucose cleavage at the phosphofructokinase stage (formation of fructose-1,6-diphosphate from fructose-6-phosphate and ATP) is “locked” simultaneously by three mechanisms, i.e. the enzyme is allosterically inhibited by (i) H+ formed at the final stage of glycolysis due to the dissociation of lactic acid, (ii) citrate synthesized already at the first stage of the Krebs respiratory cycle, and (iii) ATP, product of respiratory phosphorylation (the latter “lock” should be particularly effective, since it is a rare case of a decrease in the enzyme activity by one of its own substrates) [157, 160]. Shutdown of glucose transformation to lactate and hydrogen ion blocks the acidification of the medium, which causes protonation of superoxide anion (O2–·) leading to the formation of a much more aggressive HO2˙ radical. Thus, oxidative stress is prevented.
The fact that naked mole rats use fructose instead of glucose as one of the main carbohydrate substrates of oxidative metabolism makes metabolism more versatile in terms of substrate choice (carbohydrates can be aerobically oxidized along with fats). At the same time, there is a danger some carbohydrates will turn into lactate and H+ and acidify the medium.
This solution is fraught with two more consequences that exacerbate oxidative stress. One is increased intracellular fructose concentration (apparently due to cleavage by alkaline phosphatase of fructose-6-phosphate obtained from glucose in reaction 1). Fructose is present in the form of a ketone in a much higher percentage than glucose in the form of an aldehyde [161], which leads to a sevenfold acceleration of protein glycation [162]. Another circumstance that needs to be considered is that one of the intermediates of the fructose (but not glucose) pathway is glyceraldehyde (reaction 2). The “aldehyde danger” of glyceraldehyde is significantly higher than that of phosphoglyceraldehyde, since phosphoglyceraldehyde dehydrogenase (the enzyme that oxidizes phosphoglyceric aldehyde to acid) is present in a record high concentration in the cytosol: the amount of enzyme is more than 1000 times higher than that of its substrate (phosphoglyceraldehyde). This means that phosphoglyceraldehyde can be immediately attacked by its dehydrogenase in any place inside the cell. The intracellular concentration of glyceraldehyde kinase is smaller than that of phosphoglyceraldehyde dehydrogenase, and its direct product is still an aldehyde.
The fact that the naked mole rats took such a risky step as activation of the fructose pathway in addition to the glucose pathway is another indication of the strong antioxidant defense in these animals, which is expressed as the highest resistance to anoxia/reoxygenation among mammals [61-63]. As mentioned above, a number of important parameters do not change during aging in naked mole rats: ROS concentration and the amount of unsaturated fatty acids are not increased; the level of the main antioxidant enzymes, catalase and superoxide dismutase, are not reduced; the amount of reduced glutathione is not decreased; these animals have a special mechanism of ΔΨ reduction in mitochondria following ADP depletion [33, 34]; the most easily oxidizable cysteine (Cys272) in β-actin is replaced by a non-oxidizable serine, etc. [105]. That is why ROS level, being somewhat higher in naked mole rats than in mice, but constant throughout life, cannot serve as an argument against the role of ROS in aging [163].
In addition to aging and anti-aging programs, the figure shows acute phenoptosis, which is activated by the Master biological clock (in non-aging organisms) or by the system that monitors the key parameters of homeostasis (in both aging and non-aging organisms). In non-aging species (e.g. albatross or marsupial mouse), acute phenoptosis causes sudden death, thereby accelerating the change of generations and increasing the variety of offspring. In addition, acute phenoptosis is used for self-elimination of individuals with a persistent deviation from the norm of some parameters of homeostasis that endangers the very existence of the population (for example, the appearance of lipopolysaccharide fragments of the bacterial wall in blood or a sharp increase in mutagenesis). If chronic phenoptosis leads to a slow but steady age-related increase of mROS level, then acute phenoptosis should cause a sudden, rapid outburst of mROS, which results, for example, in the mass death of cells by apoptosis or necrosis, which causes dysfunction of vital organs and rapid death of the organism (“biochemical suicide”).
Today the scheme presented in the figure is nothing more than a working hypothesis. The mechanism of the Master clock and the place of its localization in the mammalian brain remains a mystery. It is clear only that it functions as the driver of ontogeny and of such ontogenetic program as aging. It is also obvious that this program should be attributed to the phenomena of chronic phenoptosis. At least in mammals, it involves mROS formed in large quantities during electrical hyperpolarization of the internal mitochondrial membrane. It is very likely that age-related increase in mROS level is connected both to the acceleration of ROS formation in complexes I and III of the respiratory chain and to the slowing of mROS detoxification due to the age-related decline in the activity of catalase, mitochondrial superoxide dismutase, and glutathione-reducing systems. Moreover, it is possible that mROS are formed by a complex of cytochrome c and p66shc in the intermembrane space, by lipoate dehydrogenase in mitochondrial matrix, and by monoamino oxidase of the outer mitochondrial membrane.
It should be mentioned that, although such a mitochondria-targeted antioxidant as SkQ1 prolongs lifespan of many animals (crustaceans, insects, fish, and mammals), as well as plants and fungi [93, 112, 142, 164], we could not obtain SkQ1 geroprotective effect on the nematode C. elegans (E. I. Rogaev and A. P. Grigorenko, unpublished data). This very short-lived creature (lifespan of only 15 days) has no ROS-induced apoptosis. Moreover, low ROS doses, instead of stimulating, inhibit apoptosis [165]. Apparently, this invertebrate has some other mechanism of programmed aging (if such a program exists in this nematode with a two-week life expectancy) [166].
It is possible that also in higher animals mROS-mediated aging is not the only program of aging, which might be stimulated by some other poisons formed in their organism.
The mechanism of acute phenoptosis and its role as the final stage of aging are even less clear. The fact that not only chronic but also acute phenoptosis is suppressed by SkQ1 indicates that both mechanisms are mediated by mROS in mammals. However, it remains to be clarified whether all cases of acute phenoptosis require mROS and are sensitive to SkQ1. As for the mediators operating between the Master clock (or the control mechanisms of homeostasis) and the burst in mROS that kills the organism, this is entirely the subject of future research. This also true to the system of receptors that control the state of homeostasis.
Many questions still need to be solved for deciphering the mechanism of the anti-aging program that insures the body against too fast death. The fact of geroprotective effect of dietary restriction has been firmly established. Presumably, a serious muscle load that always accompanies search for food, is also part of this effect [20], but the role of melatonin in this second geroprotective action has not been studied yet, as well as other possible ways to stop chronic phenoptosis without the use of specific inhibitors such as SkQ1. However, undoubtedly, the success of using new geroprotectors as means of fighting sudden death should be developed.
Experimental evidence of the most important hypothesis that aging is one of the programs of late ontogeny, which is retarded by neoteny in the naked mole rats and humans, makes us reconsider our understanding of the mechanisms ensuring functioning of our organism. Apparently, a series of our most terrible ailments result not from some external causes, but from our own counterproductive programs inherited from animals, and these programs are already unnecessary for humans, being harmful atavisms subjected to cancellation. In this connection, we would like to conclude our review with the words of I. I. Mechnikov, written over a hundred years ago: “Humans, being the result of a long cycle of development, carry obvious traces of animal origin. Having acquired a degree of mental development unknown in the animal world, we kept many animal features that proved to be not only unnecessary, but also directly harmful” [167].
Acknowledgments
The authors are grateful to M. Vyssokikh, V. Gladyshev, A. Markov, D. Skulachev, B. Chernyak and G. Shilovsky for useful advices.
The work was financially supported by the Russian Science Foundation (project No. 14-50-00029).
REFERENCES
1.Kerr, J. F., Wyllie, A. H., and Currie, A. R.
(1972) Apoptosis: a basic biological phenomenon with wide-ranging
implications in tissue kinetics, Br. J. Cancer, 26,
239-257.
2.Darwin, C. (1871) The Descent of Man, and
Selection in Relation to Sex, J. Murray, London.
3.Weismann, A. (1889) Essays upon Heredity and
Kindred Biological Problems, 2nd Edn., Clarendon Press, Oxford.
4.Kirkwood, T. B., and Melov, S. (2011) On the
programmed/non-programmed nature of ageing within the life history,
Curr. Biol., 21, 701-707.
5.Williams, G. C. (1957) Pleiotropy, natural
selection, and the evolution of senescence, Evolution,
11, 398-411.
6.Blagosklonny, M. V. (2013) Aging is not programmed:
genetic pseudo-program is a shadow of developmental growth, Cell
Cycle, 12, 3736-3742.
7.Walker, R. F. (2017) On the cause and mechanism of
phenoptosis, Biochemistry (Moscow), 82, 1462-1479.
8.George, J. C., Bada, J., Zeh, J., Scott, L., Brown,
S. E., O’Hara, T., and Suydam, R. (1999) Age and growth estimates
of bowhead whales (Balaena mysticetus) via aspartic acid
racemization, Can. J. Zool., 77, 571-580.
9.Guiamet, J. J., John, I., Pichersky, E., and
Nooden, L. D. (1997) Expression of a soybean thiol protease during leaf
senescence and nitrogen starvation, Plant Physiol., 114,
1220-1220.
10.Skulachev, V. P. (1997) Aging is a specific
biological function rather than the result of a disorder in complex
living systems: biochemical evidence in support of Weismann’s
hypothesis, Biochemistry (Moscow), 62, 1191-1195.
11.Skulachev, V. P., Skulachev, M. V., and Feniuk,
B. A. (2013) Life without Aging [in Russian], EKSMO, Moscow.
12.Libertini, G. (2012) Classification of
phenoptotic phenomena, Biochemistry (Moscow), 77,
707-715.
13.Libertini, G. (2012) Phenoptosis, another
specialized neologism, or the mark of a widespread revolution?
Biochemistry (Moscow), 77, 795-798.
14.Lewis, K. (2000) Programmed death in bacteria,
Microbiol. Mol. Biol. Rev., 64, 503-514.
15.Woods, R. J., Barrick, J. E., Cooper, T. F.,
Shrestha, U., Kauth, M. R., and Lenski, R. E. (2011) Second-order
selection for evolvability in a large Escherichia coli
population, Science, 331, 1433-1436.
16.Nooden, L. D., and Murray, B. J. (1982)
Transmission of the monocarpic senescence signal via the xylem in
soybean, Plant Physiol., 69, 754-756.
17.Lens, F., Smets, E., and Melzer, S. (2012) Stem
anatomy supports Arabidopsis thaliana as a model for insular
woodiness, New Phytol., 193, 12-17.
18.Melzer, S., Lens, F., Gennen, J., Vanneste, S.,
Rohde, A., and Beeckman, T. (2008) Flowering-time genes modulate
meristem determinacy and growth form in Arabidopsis thaliana,
Nat. Genet., 40, 1489-1492.
19.Wodinsky, J. (1977) Hormonal inhibition of
feeding and death in octopus – control by optic gland secretion,
Science, 198, 948-951.
20.Skulachev, V. P., Skulachev, M. V., and Feniuk,
B. A. (2018) Life without Aging [in Russian], MSU, Moscow, in
press.
21.Severtsev, A., and Shubkina, A. (2014) Predator
as a universal breeder, Nauka v Rossii, 5, 11-19.
22.Mitteldorf, J., and Sagan, D. (2017) Suicide
Genes, MacMillan Press, in press.
23.Carr-Saunders, A. M. (1922) The Population
Problem; A Study in Human Evolution, Clarendon Press, Oxford.
24.Gilpin, M. E. (1975) Group Selection in
Predator–Prey Communities, Princeton University Press,
Princeton, N. J.
25.Skulachev, V. P. (2003) Aging and the programmed
death phenomena, in Topics of Current Genetics, Model Systems
in Aging (Nystrom, T., and Osiewacz, H. D., eds.) Springer-Verlag,
Berlin-Heidelberg, pp. 192-237.
26.Fenton, M. J., and Golenbock, D. T. (1998)
LPS-binding proteins and receptors, J. Leukoc. Biol., 64,
25-32.
27.Klosterhalfen, B., and Bhardwaj, R. S. (1998)
Septic shock, Gen. Pharmacol., 31, 25-32.
28.Skulachev, V. P. (2002) Programmed death
phenomena: from organelle to organism, Ann. N. Y. Acad. Sci.,
959, 214-237.
29.Bradley, A. J., Mcdonald, I. R., and Lee, A. K.
(1980) Stress and mortality in a small marsupial (Antechinus
stuartii, Macleay), Gen. Comp. Endocr., 40,
188-200.
30.Lecomte, V. J., Sorci, G., Cornet, S., Jaeger,
A., Faivre, B., Arnoux, E., Gaillard, M., Trouve, C., Besson, D.,
Chastel, O., and Weimerskirch, H. (2010) Patterns of aging in the
long-lived wandering albatross, Proc. Natl. Acad. Sci. USA,
107, 6370-6375.
31.Zahavi, A. (1975) Mate selection – a
selection for a handicap, J. Theor. Biol., 53,
205-214.
32.Hopkin, K. (2003) Dietary drawbacks, Sci.
Aging Knowledge Environ., 2003, NS4.
33.Skulachev, V. P., Holtze, S., Vyssokikh, M. Y.,
Bakeeva, L. E., Skulachev, M. V., Markov, A. V., Hildebrandt, T. B.,
and Sadovnichii, V. A. (2017) Neoteny, prolongation of youth: from
naked mole rats to “naked apes” (humans),
Biokhimiya, 82, 1389-1416.
34.Skulachev, V. P., Holtze, S., Vyssokikh, M. Y.,
Bakeeva, L. E., Skulachev, M. V., Markov, A. V., Hildebrandt, T. B.,
and Sadovnichii, V. A. (2017) Neoteny, prolongation of youth: from
naked mole rats to “naked apes” (humans), Physiol.
Rev., 97, 699-720.
35.Kollman, J. (1905) Neue gedanken uber das alter
problem von der abstammung des menschen, Bl. Dtsch. Ges. Anthropol.
Ethnol. Urges, pp. 9-20.
36.Bufill, E., Agusti, J., and Blesa, R. (2011)
Human neoteny revisited: the case of synaptic plasticity, Am. J.
Hum. Biol., 23, 729-739.
37.Rosenkilde, P., and Ussing, A. P. (1996) What
mechanisms control neoteny and regulate induced metamorphosis in
urodeles? Int. J. Dev. Biol., 40, 665-673.
38.Safi, R., Bertrand, S., Marchand, O., Duffraisse,
M., de Luze, A., Vanacker, J. M., Maraninchi, M., Margotat, A.,
Demeneix, B., and Laudet, V. (2004) The axolotl (Ambystoma
mexicanum), a neotenic amphibian, expresses functional thyroid
hormone receptors, Endocrinology, 145, 760-772.
39.Snider, A. T., and Bowler, J. K. (1992)
Longevity of Reptiles and Amphibians in North American
Collections, 2nd Edn., Society for the Study of Amphibians and
Reptiles, Oxford, Ohio.
40.Voituron, Y., De Fraipont, M., Issartel, J.,
Guillaume, O., and Clobert, J. (2011) Extreme lifespan of the human
fish (Proteus anguinus): a challenge for ageing mechanisms,
Biol. Lett., 7, 105-107.
41.Huynh, T. D. H., Gallien, C. L., Durand, J. P.,
and Chanoine, C. (1996) Cloning and expression of a thyroid hormone
receptor alpha 1 in the perennibranchiate amphibian Proteus
anguinus, Int. J. Dev. Biol., 40, 537-543.
42.Pfingsten, R. A., and White, A. M. (1989)
Necturus maculosus (Rafinesque), mudpuppy, in Salamanders of
Ohio. Ohio Biological Survey (Pfingsten, R. A., and Downs, F. L.,
eds.) Columbus, Ohio, pp. 72-78.
43.McDaniel, T. V., Martin, P. A., Barrett, G. C.,
Hughes, K., Gendron, A. D., Shirose, L., and Bishop, C. A. (2009)
Relative abundance, age structure, and body size in mudpuppy
populations in southwestern Ontario, J. Great Lakes Res.,
35, 182-189.
44.Bonin, J., Desgranges, J. L., Bishop, C. A.,
Rodrigue, J., Gendron, A., and Elliott, J. E. (1995) Comparative-study
of contaminants in the mudpuppy (Amphibia) and the common snapping
turtle (Reptilia), St-Lawrence-River, Canada, Arch. Environ. Con.
Tox., 28, 184-194.
45.Nielsen, J., Hedeholm, R. B., Heinemeier, J.,
Bushnell, P. G., Christiansen, J. S., Olsen, J., Ramsey, C. B., Brill,
R. W., Simon, M., Steffensen, K. F., and Steffensen, J. F. (2016) Eye
lens radiocarbon reveals centuries of longevity in the Greenland shark
(Somniosus microcephalus), Science, 353,
702-704.
46.Soltanimazouni, N., and Bordereau, C. (1987)
Changes in the cuticle, ovaries and colleterial glands during the
pseudergate and neotenic molt in Kalotermes flavicollis (Fabr)
(Isoptera, Kalotermitidae), Int. J. Insect. Morphol., 16,
221-235.
47.Perdereau, E., Bagneres, A. G., Vargo, E. L.,
Baudouin, G., Xu, Y., Labadie, P., Dupont, S., and Dedeine, F. (2015)
Relationship between invasion success and colony breeding structure in
a subterranean termite, Mol. Ecol., 24, 2125-2142.
48.Brokeland, W., and Brandt, A. (2004) Two new
species of Ischnomesidae (Crustacea: Isopoda) from the Southern Ocean,
displaying neoteny, Deep Sea Res. Pt. II, 51,
1769-1785.
49.Piraino, S., Boero, F., Aeschbach, B., and
Schmid, V. (1996) Reversing the life cycle: medusae transforming into
polyps and cell transdifferentiation in Turritopsis nutricula
(Cnidaria, Hydrozoa), Biol. Bull., 190, 302-312.
50.Devarapalli, P., Kumavath, R. N., Barh, D., and
Azevedo, V. (2014) The conserved mitochondrial gene distribution in
relatives of Turritopsis nutricula, an immortal jellyfish,
Bioinformation, 10, 586-591.
51.Tsai, C. H., and Fordyce, R. E. (2014) Juvenile
morphology in baleen whale phylogeny, Naturwissenschaften,
101, 765-769.
52.Tsai, C. H., and Fordyce, R. E. (2014) Disparate
heterochronic processes in baleen whale evolution, Evol. Biol.,
41, 299-307.
53.Buffenstein, R. (2008) Negligible senescence in
the longest living rodent, the naked mole-rat: insights from a
successfully aging species, J. Comp. Physiol. B, 178,
439-445.
54.Buffenstein, R. (2005) The naked mole-rat? A new
long-living model for human aging research, J. Gerontol.,
60, 1369-1377.
55.Orr, M. E., Garbarino, V. R., Salinas, A., and
Buffenstein, R. (2015) Sustained high levels of neuroprotective, high
molecular weight, phosphorylated tau in the longest-lived rodent,
Neurobiol. Aging, 36, 1496-1504.
56.Edrey, Y. H., Hanes, M., Pinto, M., Mele, J., and
Buffenstein, R. (2011) Successful aging and sustained good health in
the naked mole rat: a long-lived mammalian model for biogerontology and
biomedical research, ILAR J., 52, 41-53.
57.Delaney, M. A., Nagy, L., Kinsel, M. J., and
Treuting, P. M. (2013) Spontaneous histologic lesions of the adult
naked mole rat (Heterocephalus glaber): a retrospective survey
of lesions in a zoo population, Vet. Pathol., 50,
607-621.
58.Finch, C. (1990) Longevity, Senescence, and
the Genome, University of Chicago Press, Chicago.
59.Skulachev, M. V., and Skulachev, V. P. (2017)
(accepted) Phenoptosis – programmed death of an organism, in
Apoptosis and Beyond: The Many Ways Cells Die (Radosevich, J.,
ed.) Springer-Verlag, Berlin-Heidelberg.
60.Alexander, R. D. (1991) Some unanswered questions
about naked mole-rats, in The Biology of the Naked Mole-Rat:
Monographs in Behavior and Ecology (Sherman, P. W., Jarvis, J. U.
M., and Alexander, R. D., eds.) Princeton University Press, Princeton,
NJ, pp. 446-465.
61.Larson, J., Drew, K. L., Folkow, L. P., Milton,
S. L., and Park, T. J. (2014) No oxygen? No problem! Intrinsic brain
tolerance to hypoxia in vertebrates, J. Exp. Biol., 217,
1024-1039.
62.Larson, J., and Park, T. J. (2009) Extreme
hypoxia tolerance of naked mole-rat brain, Neuroreport,
20, 1634-1637.
63.Nathaniel, T., Umesiric, F., Saras, A., and
Olajuyigbe, F. (2009) Tolerance to oxygen nutrient deprivation in the
hippocampal slices of the naked mole rat, J. Cerebr. Blood. F
Met., 29, S451-S451.
64.Boyle, R. (1725) Philosophical Works,
Innys, London.
65.Duffy, T. E., Kohle, S. J., and Vannucci, R. C.
(1975) Carbohydrate and energy metabolism in perinatal rat-brain
– relation to survival in anoxia, J. Neurochem.,
24, 271-276.
66.Bickler, P. E., Fahlram, C. S., and Taylor, D. M.
(2003) Oxygen sensitivity of NMDA receptors: relationship to NR2
subunit composition and hypoxia tolerance of neonatal neurons,
Neuroscience, 118, 25-35.
67.Cherubini, E., Benari, Y., and Krnjevic, K.
(1989) Anoxia produces smaller changes in synaptic transmission,
membrane-potential, and input resistance in immature rat hippocampus,
J. Neurophysiol., 62, 882-895.
68.Penz, O. K., Fuzik, J., Kurek, A. B., Romanov,
R., Larson, J., Park, T. J., Harkany, T., and Keimpema, E. (2015)
Protracted brain development in a rodent model of extreme longevity,
Sci. Rep., 5, 11592.
69.Triplett, J. C., Swomley, A., Kirk, J., Lewis,
K., Orr, M., Rodriguez, K., Cai, J., Klein, J. B., Buffenstein, R., and
Butterfield, D. A. (2015) Metabolic clues to salubrious longevity in
the brain of the longest-lived rodent: the naked mole-rat, J.
Neurochem., 134, 538-550.
70.Holtze, S., Eldarov, C. M., Vays, V. B., Vangeli,
I. M., Vyssokikh, M. Y., Bakeeva, L. E., Skulachev, V. P., and
Hildebrandt, T. B. (2016) Study of age-dependent structural and
functional changes of mitochondria in skeletal muscles and heart of
naked mole rats (Heterocephalus glaber), Biochemistry
(Moscow), 81, 1429-1437.
71.Aprille, J. R., and Asimakis, G. K. (1980)
Postnatal-development of rat-liver mitochondria – state-3
respiration, adenine-nucleotide translocase activity, and the net
accumulation of adeninenucleotides, Arch. Biochem. Biophys.,
201, 564-575.
72.Skulachev, V. P. (2015) Moscow News: Two More
Representatives of Sodium Motive Force Generators
(Na+-cbb3 Oxidase and
Na+-Bacteriorhodopsin); Natural Delay of the Aging Program
(Neoteny) in Mammals, Namely in Naked Mole Rat and “Naked
Ape” Human), MIP-2015, 11th Conf. Mitochondrial Physiology,
Lucni Bouda, Czech Republic, Abstracts, pp. 64-66.
73.Bolk, L. (1926) The Problem of Human
Development, Gustav Fischer, Jena.
74.Bolk, L. (1927) On the origin of human races,
P K Akad. Wet-Amsterd., 30, 320-328.
75.Sakai, T., Hirata, S., Fuwa, K., Sugama, K.,
Kusunoki, K., Makishima, H., Eguchi, T., Yamada, S., Ogihara, N., and
Takeshita, H. (2012) Fetal brain development in chimpanzees versus
humans, Curr. Biol., 22, R791-792.
75a.Dilman, V. M. (1978) Ageing, metabolic immunodepression and
carcinogenesis, Mech. Ageing Dev., 8, 153-173.
76.Kreger, C. D., Modern Human Origins
(www.modernhumanorigins.net).
77.Lovejoy, C. O. (2009) Reexamining human origins
in light of Ardipithecus ramidus, Science, 326,
74e1-8.
78.Schwartz, J. H. (1999) Sudden Origins:
Fossils, Genes, and the Emergence of Species, John Wiley &
Sons, New York.
79.Bromhall, C. (2003) The Eternal Child: an
Explosive New Theory of Human Origins and Behaviour, Ebury,
London.
80.Moxon, S. (2003) The eternal child: an explosive
new theory of human origins and behaviour by Clive Bromhall, Ebury
Press, Hum. Nat. Rev., 3, 402-405.
81.Liu, X. L., Somel, M., Tang, L., Yan, Z., Jiang,
X., Guo, S., Yuan, Y., He, L., Oleksiak, A., Zhang, Y., Li, N., Hu, Y.
H., Chen, W., Qiu, Z. L., Paabo, S., and Khaitovich, P. (2012)
Extension of cortical synaptic development distinguishes humans from
chimpanzees and macaques, Genome Res., 22, 611-622.
82.Somel, M., Franz, H., Yan, Z., Lorenc, A., Guo,
S., Giger, T., Kelso, J., Nickel, B., Dannemann, M., Bahn, S., Webster,
M. J., Weickert, C. S., Lachmann, M., Paabo, S., and Khaitovich, P.
(2009) Transcriptional neoteny in the human brain, Proc. Natl. Acad.
Sci. USA, 106, 5743-5748.
83.Petanjek, Z., Judas, M., Simic, G., Rasin, M. R.,
Uylings, H. B. M., Rakic, P., and Kostovic, I. (2011) Extraordinary
neoteny of synaptic spines in the human prefrontal cortex, Proc.
Nat. Acad. Sci. USA, 108, 13281-13286.
84.Xiao, J. (2007) A new coordinate system for
rodent brain and variability in the brain weights and dimensions of
different ages in the naked mole-rat, J. Neurosci. Meth.,
162, 162-170.
85.Davies, K. T. J., Bennett, N. C., Tsagkogeorga,
G., Rossiter, S. J., and Faulkes, C. G. (2015) Family wide molecular
adaptations to underground life in african mole-rats revealed by
phylogenomic analysis, Mol. Biol. Evol., 32,
3089-3107.
86.Faulkes, C. G., Bennett, N. C., Cotterill, F. P.
D., Stanley, W., Mgode, G. F., and Verheyen, E. (2011) Phylogeography
and cryptic diversity of the solitary-dwelling silvery mole-rat, genus
Heliophobius (family: Bathyergidae), J. Zool.,
285, 324-338.
87.Okrouhlik, J., Burda, H., Kunc, P., Knizkova, I.,
and Sumbera, R. (2015) Surprisingly low risk of overheating during
digging in two subterranean rodents, Physiol. Behav.,
138, 236-241.
88.Edrey, Y. H., Casper, D., Huchon, D., Mele, J.,
Gelfond, J. A., Kristan, D. M., Nevo, E., and Buffenstein, R. (2012)
Sustained high levels of neuregulin-1 in the longest-lived rodents; a
key determinant of rodent longevity, Aging Cell, 11,
213-222.
89.Ingram, C. M., Burda, H., and Honeycutt, R. L.
(2004) Molecular phylogenetics and taxonomy of the African mole-rats,
genus Cryptomys and the new genus Coetomys Gray, 1864,
Mol. Phylogenet. Evol., 31, 997-1014.
90.Patterson, B. D., and Upham, N. S. (2014) A newly
recognized family from the Horn of Africa, the Heterocephalidae
(Rodentia: Ctenohystrica), Zool. J. Linn. Soc. Lond.,
172, 942-963.
91.Dammann, P. (2017) Slow aging in mammals –
lessons from African mole-rats and bats, Semin. Cell. Dev.
Biol., S1084-9521, 30313-30315.
92.Jones, O. R., Scheuerlein, A., Salguero-Gomez,
R., Camarda, C. G., Schaible, R., Casper, B. B., Dahlgren, J. P.,
Ehrlen, J., Garcia, M. B., Menges, E. S., Quintana-Ascencio, P. F.,
Caswell, H., Baudisch, A., and Vaupel, J. W. (2014) Diversity of ageing
across the tree of life, Nature, 505, 169-173.
93.Anisimov, V. N., Egorov, M. V., Krasilshchikova,
M. S., Lyamzaev, K. G., Manskikh, V. N., Moshkin, M. P., Novikov, E.
A., Popovich, I. G., Rogovin, K. A., Shabalina, I. G., Shekarova, O.
N., Skulachev, M. V., Titova, T. V., Vygodin, V. A., Vyssokikh, M. Y.,
Yurova, M. N., Zabezhinsky, M. A., and Skulachev, V. P. (2011) Effects
of the mitochondria-targeted antioxidant SkQ1 on lifespan of rodents,
Aging (Albany, NY), 3, 1110-1119.
94.Schriner, S. E., Linford, N. J., Martin, G. M.,
Treuting, P., Ogburn, C. E., Emond, M., Coskun, P. E., Ladiges, W.,
Wolf, N., Van Remmen, H., Wallace, D. C., and Rabinovitch, P. S. (2005)
Extension of murine life span by overexpression of catalase targeted to
mitochondria, Science, 308, 1909-1911.
95.Miller, R. A., Buehner, G., Chang, Y., Harper, J.
M., Sigler, R., and Smith-Wheelock, M. (2005) Methionine-deficient diet
extends mouse lifespan, slows immune and lens aging, alters glucose,
T4, IGF-I and insulin levels, and increases hepatocyte MIF levels and
stress resistance, Aging Cell, 4, 119-125.
96.Sanz, A., Caro, P., Ayala, V., Portero-Otin, M.,
Pamplona, R., and Barja, G. (2006) Methionine restriction decreases
mitochondrial oxygen radical generation and leak as well as oxidative
damage to mitochondrial DNA and proteins, FASEB J., 20,
1064-1073.
97.Caro, P., Gomez, J., Sanchez, I., Garcia, R.,
Lopez-Torres, M., Naudi, A., Portero-Otin, M., Pamplona, R., and Barja,
G. (2009) Effect of 40% restriction of dietary amino acids (except
methionine) on mitochondrial oxidative stress and biogenesis, AIF and
SIRT1 in rat liver, Biogerontology, 10, 579-592.
98.Sanchez-Roman, I., Gomez, A., Perez, I., Sanchez,
C., Suarez, H., Naudi, A., Jove, M., Lopez-Torres, M., Pamplona, R.,
and Barja, G. (2012) Effects of aging and methionine restriction
applied at old age on ROS generation and oxidative damage in rat liver
mitochondria, Biogerontology, 13, 399-411.
99.Stehle, J. H., Saade, A., Rawashdeh, O.,
Ackermann, K., Jilg, A., Sebesteny, T., and Maronde, E. (2011) A survey
of molecular details in the human pineal gland in the light of
phylogeny, structure, function and chronobiological diseases, J.
Pineal. Res., 51, 17-43.
100.Wetterberg, L., Eberhard, G., and von Knorring,
L. (1993) The Influence of Age, Sex, Height, Weight, Urine Volume
and Latitude on Melatonin Concentrations in Urine from Normal Subjects:
a Multinational Study. Light and Biological Rhythms in Man,
Pergamon Press Ltd., Oxford, pp. 275-286.
101.Sack, R. L., Lewy, A. J., Erb, D. L., Vollmer,
W. M., and Singer, C. M. (1986) Human melatonin production decreases
with age, J. Pineal. Res., 3, 379-388.
102.Roth, G. S., Lesnikov, V., Lesnikov, M.,
Ingram, D. K., and Lane, M. A. (2001) Dietary caloric restriction
prevents the age-related decline in plasma melatonin levels of rhesus
monkeys, J. Clin. Endocrinol. Metab., 86, 3292-3295.
103.Solanas, G., Peixoto, F. O., Perdiguero, E.,
Jardi, M., Ruiz-Bonilla, V., Datta, D., Symeonidi, A., Castellanos, A.,
Welz, P. S., Caballero, J. M., Sassone-Corsi, P., Munoz-Canoves, P.,
and Benitah, S. A. (2017) Aged stem cells reprogram their daily
rhythmic functions to adapt to stress, Cell, 170,
678-692.
104.Sato, S., Solanas, G., Peixoto, F. O., Bee, L.,
Symeonidi, A., Schmidt, M. S., Brenner, C., Masri, S., Benitah, S. A.,
and Sassone-Corsi, P. (2017) Circadian reprogramming in the liver
identifies metabolic pathways of aging, Cell, 170,
664-677.
105.Kim, E. B., Fang, X., Fushan, A. A., Huang, Z.,
Lobanov, A. V., Han, L., Marino, S. M., Sun, X., Turanov, A. A., Yang,
P., Yim, S. H., Zhao, X., Kasaikina, M. V., Stoletzki, N., Peng, C.,
Polak, P., Xiong, Z., Kiezun, A., Zhu, Y., Chen, Y., Kryukov, G. V.,
Zhang, Q., Peshkin, L., Yang, L., Bronson, R. T., Buffenstein, R.,
Wang, B., Han, C., Li, Q., Chen, L., Zhao, W., Sunyaev, S. R., Park, T.
J., Zhang, G., Wang, J., and Gladyshev, V. N. (2011) Genome sequencing
reveals insights into physiology and longevity of the naked mole rat,
Nature, 479, 223-227.
106.Quay, W. B. (1981) Pineal atrophy and other
neuroendocrine and circumventricular features of the naked mole-rat,
Heterocephalus glaber (Ruppell), a fossorial, equatorial rodent,
J. Neural. Transm., 52, 107-115.
107.Pierpaoli, W., and Bulian, D. (2005) The pineal
aging and death program: life prolongation in pre-aging pinealectomized
mice, Ann. N. Y. Acad. Sci., 1057, 133-144.
108.Hart, L., Bennett, N. C., Malpaux, B.,
Chimimba, C. T., and Oosthuizen, M. K. (2004) The chronobiology of the
Natal mole-rat, Cryptomys hottentotus natalensis,
Physiol. Behav., 82, 563-569.
109.Skulachev, V. P. (1996) Role of uncoupled and
non-coupled oxidations in maintenance of safely low levels of oxygen
and its one-electron reductants, Q Rev. Biophys., 29,
169-202.
110.Korshunov, S. S., Skulachev, V. P., and
Starkov, A. A. (1997) High protonic potential actuates a mechanism of
production of reactive oxygen species in mitochondria, FEBS
Lett., 416, 15-18.
111.Skulachev, V. P. (2013) Cationic antioxidants
as a powerful tool against mitochondrial oxidative stress, Biochem.
Biophys. Res. Commun., 441, 275-279.
112.Skulachev, V. P., Anisimov, V. N., Antonenko,
Y. N., Bakeeva, L. E., Chernyak, B. V., Erichev, V. P., Filenko, O. F.,
Kalinina, N. I., Kapelko, V. I., Kolosova, N. G., Kopnin, B. P.,
Korshunova, G. A., Lichinitser, M. R., Obukhova, L. A., Pasyukova, E.
G., Pisarenko, O. I., Roginsky, V. A., Ruuge, E. K., Senin, I. I.,
Severina, I. I., Skulachev, M. V., Spivak, I. M., Tashlitsky, V. N.,
Tkachuk, V. A., Vyssokikh, M. Y., Yaguzhinsky, L. S., and Zorov, D. B.
(2009) An attempt to prevent senescence: a mitochondrial approach,
Biochim. Biophys. Acta, 1787, 437-461.
113.Skulachev, V. P., Antonenko, Y. N., Cherepanov,
D. A., Chernyak, B. V., Izyumov, D. S., Khailova, L. S., Klishin, S.
S., Korshunova, G. A., Lyamzaev, K. G., Pletjushkina, O. Y., Roginsky,
V. A., Rokitskaya, T. I., Severin, F. F., Severina, I. I., Simonyan, R.
A., Skulachev, M. V., Sumbatyan, N. V., Sukhanova, E. I., Tashlitsky,
V. N., Trendeleva, T. A., Vyssokikh, M. Y., and Zvyagilskaya, R. A.
(2010) Prevention of cardiolipin oxidation and fatty acid cycling as
two antioxidant mechanisms of cationic derivatives of plastoquinone
(SkQs), Biochim. Biophys. Acta, 1797, 878-889.
114.Shabalina, I. G., Vyssokikh, M. Y., Gibanova,
N., Csikasz, R. I., Edgar, D., Hallden-Waldemarson, A.,
Rozhdestvenskaya, Z., Bakeeva, L. E., Vays, V. B., Pustovidko, A. V.,
Skulachev, M. V., Cannon, B., Skulachev, V. P., and Nedergaard, J.
(2017) Improved health-span and lifespan in mtDNA mutator mice treated
with the mitochondrially targeted antioxidant SkQ1, Aging (Albany,
NY), 9, 315-339.
115.Dai, D. F., Chen, T., Wanagat, J., Laflamme,
M., Marcinek, D. J., Emond, M. J., Ngo, C. P., Prolla, T. A., and
Rabinovitch, P. S. (2010) Age-dependent cardiomyopathy in mitochondrial
mutator mice is attenuated by overexpression of catalase targeted to
mitochondria, Aging Cell, 9, 536-544.
116.Dai, D. F., and Rabinovitch, P. S. (2009)
Cardiac aging in mice and humans: the role of mitochondrial oxidative
stress, Trends Cardiovas. Med., 19, 213-220.
117.Lee, H. Y., Choi, C. S., Birkenfeld, A. L.,
Alves, T. C., Jornayvaz, F. R., Jurczak, M. J., Zhang, D., Woo, D. K.,
Shadel, G. S., Ladiges, W., Rabinovitch, P. S., Santos, J. H.,
Petersen, K. F., Samuel, V. T., and Shulman, G. I. (2010) Targeted
expression of catalase to mitochondria prevents age-associated
reductions in mitochondrial function and insulin resistance, Cell.
Metab., 12, 668-674.
118.Brzheskiy, V. V., Efimova, E. L., Vorontsova,
T. N., Alekseev, V. N., Gusarevich, O. G., Shaidurova, K. N.,
Ryabtseva, A. A., Andryukhina, O. M., Kamenskikh, T. G., Sumarokova, E.
S., Miljudin, E. S., Egorov, E. A., Lebedev, O. I., Surov, A. V.,
Korol, A. R., Nasinnyk, I. O., Bezditko, P. A., Muzhychuk, O. P.,
Vygodin, V. A., Yani, E. V., Savchenko, A. Y., Karger, E. M., Fedorkin,
O. N., Mironov, A. N., Ostapenko, V., Popeko, N. A., Skulachev, V. P.,
and Skulachev, M. V. (2015) Results of a multicenter, randomized,
double-masked, placebo-controlled clinical study of the efficacy and
safety of visomitin eye drops in patients with dry eye syndrome,
Adv. Ther., 32, 1263-1279.
119.Petrov, A., Perekhvatova, N., Skulachev, M.,
Stein, L., and Ousler, G. (2016) SkQ1 ophthalmic solution for dry eye
treatment: results of a phase 2 safety and efficacy clinical study in
the environment and during challenge in the controlled adverse
environment model, Adv. Ther., 33, 96-115.
120.Skulachev, V. P., Bogachev, A. V., and
Kasparinsky, F. O. (2013) Principles of Bioenergetics, Springer,
Berlin-Heidelberg.
121.Griffith, A. V., Venables, T., Shi, J., Farr,
A., van Remmen, H., Szweda, L., Fallahi, M., Rabinovitch, P., and
Petrie, H. T. (2015) Metabolic damage and premature thymus aging caused
by stromal catalase deficiency, Cell. Rep., 12,
1071-1079.
122.Obukhova, L. A., Skulachev, V. P., and
Kolosova, N. G. (2009) Mitochondria-targeted antioxidant SkQ1 inhibits
age-dependent involution of the thymus in normal and senescence-prone
rats, Aging (Albany, NY), 1, 389-401.
123.Latorre-Pellicer, A., Moreno-Loshuertos, R.,
Lechuga-Vieco, A. V., Sanchez-Cabo, F., Torroja, C., Acin-Perez, R.,
Calvo, E., Aix, E., Gonzalez-Guerra, A., Logan, A., Bernad-Miana, M.
L., Romanos, E., Cruz, R., Cogliati, S., Sobrino, B., Carracedo, A.,
Perez-Martos, A., Fernandez-Silva, P., Ruiz-Cabello, J., Murphy, M. P.,
Flores, I., Vazquez, J., and Enriquez, J. A. (2016) Mitochondrial and
nuclear DNA matching shapes metabolism and healthy ageing,
Nature, 535, 561-565.
124.Skulachev, V. P. (2000) Mitochondria in the
programmed death phenomena; a principle of biology: “it is better
to die than to be wrong”, IUBMB Life, 49,
365-373.
125.Skulachev, V. P. (2001) The programmed death
phenomena, aging, and the Samurai law of biology, Exp.
Gerontol., 36, 995-1024.
126.Matsuzaki, J., Kuwamura, M., Yamaji, R., Inui,
H., and Nakano, Y. (2001) Inflammatory responses to lipopolysaccharide
are suppressed in 40% energy-restricted mice, J. Nutr.,
131, 2139-2144.
127.Zhang, Q., Raoof, M., Chen, Y., Sumi, Y.,
Sursal, T., Junger, W., Brohi, K., Itagaki, K., and Hauser, C. J.
(2010) Circulating mitochondrial DAMPs cause inflammatory responses to
injury, Nature, 464, 104-107.
128.Imai, Y., Kuba, K., Neely, G. G.,
Yaghubian-Malhami, R., Perkmann, T., Van Loo, G., Ermolaeva, M.,
Veldhuizen, R., Leung, Y. H., Wang, H., Liu, H., Sun, Y., Pasparakis,
M., Kopf, M., Mech, C., Bavari, S., Peiris, J. S., Slutsky, A. S.,
Akira, S., Hultqvist, M., Holmdahl, R., Nicholls, J., Jiang, C.,
Binder, C. J., and Penninger, J. M. (2008) Identification of oxidative
stress and Toll-like receptor 4 signaling as a key pathway of acute
lung injury, Cell, 133, 235-249.
129.Shirey, K. A., Lai, W., Scott, A. J., Lipsky,
M., Mistry, P., Pletneva, L. M., Karp, C. L., McAlees, J., Gioannini,
T. L., Weiss, J., Chen, W. H., Ernst, R. K., Rossignol, D. P.,
Gusovsky, F., Blanco, J. C., and Vogel, S. N. (2013) The TLR4
antagonist Eritoran protects mice from lethal influenza infection,
Nature, 497, 498-502.
130.Plotnikov, E. Y., Kazachenko, A. V., Vyssokikh,
M. Y., Vasileva, A. K., Tcvirkun, D. V., Isaev, N. K., Kirpatovsky, V.
I., and Zorov, D. B. (2007) The role of mitochondria in oxidative and
nitrosative stress during ischemia/reperfusion in the rat kidney,
Kidney Int., 72, 1493-1502.
131.Bakeeva, L. E., Barskov, I. V., Egorov, M. V.,
Isaev, N. K., Kapelko, V. I., Kazachenko, A. V., Kirpatovsky, V. I.,
Kozlovsky, S. V., Lakomkin, V. L., Levina, S. B., Pisarenko, O. I.,
Plotnikov, E. Y., Saprunova, V. B., Serebryakova, L. I., Skulachev, M.
V., Stelmashook, E. V., Studneva, I. M., Tskitishvili, O. V.,
Vasilyeva, A. K., Victorov, I. V., Zorov, D. B., and Skulachev, V. P.
(2008) Mitochondria-targeted plastoquinone derivatives as tools to
interrupt execution of the aging program. 2. Treatment of some ROS- and
age-related diseases (heart arrhythmia, heart infarctions, kidney
ischemia, and stroke), Biochemistry (Moscow), 73,
1288-1299.
132.Plotnikov, E. Y., Chupyrkina, A. A.,
Jankauskas, S. S., Pevzner, I. B., Silachev, D. N., Skulachev, V. P.,
and Zorov, D. B. (2011) Mechanisms of nephroprotective effect of
mitochondria-targeted antioxidants under rhabdomyolysis and
ischemia/reperfusion, Biochim. Biophys. Acta, 1812,
77-86.
133.Zorov, D. B., Plotnikov, E. Y., Jankauskas, S.
S., Isaev, N. K., Silachev, D. N., Zorova, L. D., Pevzner, I. B.,
Pulkova, N. V., Zorov, S. D., and Morosanova, M. A. (2012) The
phenoptosis problem: what is causing the death of an organism? Lessons
from acute kidney injury, Biochemistry (Moscow), 77,
742-753.
134.Plotnikov, E. Y., Silachev, D. N., Jankauskas,
S. S., Rokitskaya, T. I., Chupyrkina, A. A., Pevzner, I. B., Zorova, L.
D., Isaev, N. K., Antonenko, Y. N., Skulachev, V. P., and Zorov, D. B.
(2012) Mild uncoupling of respiration and phosphorylation as a
mechanism providing nephro- and neuroprotective effects of penetrating
cations of the SkQ family, Biochemistry (Moscow), 77,
1029-1037.
135.Jankauskas, S. S., Plotnikov, E. Y.,
Morosanova, M. A., Pevzner, I. B., Zorova, L. D., Skulachev, V. P., and
Zorov, D. B. (2012) Mitochondria-targeted antioxidant SkQR1 ameliorates
gentamycin-induced renal failure and hearing loss, Biochemistry
(Moscow), 77, 666-670.
136.Plotnikov, E. Y., Morosanova, M. A., Pevzner,
I. B., Zorova, L. D., Manskikh, V. N., Pulkova, N. V., Galkina, S. I.,
Skulachev, V. P., and Zorov, D. B. (2013) Protective effect of
mitochondria-targeted antioxidants in an acute bacterial infection,
Proc. Natl. Acad. Sci. USA, 110, 3100-3108.
137.Severin, F. F., Severina, I. I., Antonenko, Y.
N., Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M.
Y., Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova,
G. A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
Penetrating cation/fatty acid anion pair as a mitochondria-targeted
protonophore, Proc. Natl. Acad. Sci. USA, 107,
663-668.
138.Antonenko, Y. N., Avetisyan, A. V., Cherepanov,
D. A., Knorre, D. A., Korshunova, G. A., Markova, O. V., Ojovan, S. M.,
Perevoshchikova, I. V., Pustovidko, A. V., Rokitskaya, T. I., Severina,
I. I., Simonyan, R. A., Smirnova, E. A., Sobko, A. A., Sumbatyan, N.
V., Severin, F. F., and Skulachev, V. P. (2011) Derivatives of
rhodamine 19 as mild mitochondria-targeted cationic uncouplers, J.
Biol. Chem., 286, 17831-17840.
139.Padalko, V. I. (2005) Uncoupler of oxidative
phosphorylation prolongs the lifespan of Drosophila,
Biochemistry (Moscow), 70, 986-989.
140.Caldeira da Silva, C. C., Cerqueira, F. M.,
Barbosa, L. F., Medeiros, M. H., and Kowaltowski, A. J. (2008) Mild
mitochondrial uncoupling in mice affects energy metabolism, redox
balance and longevity, Aging Cell, 7, 552-560.
141.Plotnikov, E. Y., Silachev, D. N., Chupyrkina,
A. A., Danshina, M. I., Jankauskas, S. S., Morosanova, M. A.,
Stelmashook, E. V., Vasileva, A. K., Goryacheva, E. S., Pirogov, Y. A.,
Isaev, N. K., and Zorov, D. B. (2010) New-generation Skulachev’
ions exhibiting nephroprotective and neuroprotective propertie,
Biochemistry (Moscow), 75, 145-150.
142.Skulachev, M. V., Antonenko, Y. N., Anisimov,
V. N., Chernyak, B. V., Cherepanov, D. A., Chistyakov, V. A., Egorov,
M. V., Kolosova, N. G., Korshunova, G. A., Lyamzaev, K. G., Plotnikov,
E. Y., Roginsky, V. A., Savchenko, A. Y., Severina, I. I., Severin, F.
F., Shkurat, T. P., Tashlitsky, V. N., Shidlovsky, K. M., Vyssokikh, M.
Y., Zamyatnin, A. A., Zorov, D. B., and Skulachev, V. P. (2011)
Mitochondrial-targeted plastoquinone derivatives. Effect on senescence
and acute age-related pathologies, Curr. Drug Targets,
12, 800-826.
143.Chouchani, E. T., Methner, C., Nadtochiy, S.
M., Logan, A., Pell, V. R., Ding, S., James, A. M., Cocheme, H. M.,
Reinhold, J., Lilley, K. S., Partridge, L., Fearnley, I. M., Robinson,
A. J., Hartley, R. C., Smith, R. A., Krieg, T., Brookes, P. S., and
Murphy, M. P. (2013) Cardioprotection by S-nitrosation of a cysteine
switch on mitochondrial complex I, Nat. Med., 19,
753-759.
144.Kapay, N. A., Isaev, N. K., Stelmashook, E. V.,
Popova, O. V., Zorov, D. B., Skrebitsky, V. G., and Skulachev, V. P.
(2011) In vivo injected mitochondria-targeted plastoquinone
antioxidant SkQR1 prevents β-amyloid-induced decay of long-term
potentiation in rat hippocampal slices, Biochemistry (Moscow),
76, 1367-1370.
145.Kapay, N. A., Popova, O. V., Isaev, N. K.,
Stelmashook, E. V., Kondratenko, R. V., Zorov, D. B., Skrebitsky, V.
G., and Skulachev, V. P. (2013) Mitochondria-targeted plastoquinone
antioxidant SkQ1 prevents amyloid-beta-induced impairment of long-term
potentiation in rat hippocampal slices, J. Alzheimer’s
Dis., 36, 377-383.
146.Ma, T., Hoeffer, C. A., Wong, H., Massaad, C.
A., Zhou, P., Iadecola, C., Murphy, M. P., Pautler, R. G., and Klann,
E. (2011) Amyloid beta-induced impairments in hippocampal synaptic
plasticity are rescued by decreasing mitochondrial superoxide, J.
Neurosci., 31, 5589-5595.
147.Skulachev, V. P. (2012) Mitochondria-targeted
antioxidants as promising drugs for treatment of age-related brain
diseases, J. Alzheimer’s Dis., 28, 283-289.
148.Du, H., Guo, L., Fang, F., Chen, D., Sosunov,
A. A., McKhann, G. M., Yan, Y., Wang, C., Zhang, H., Molkentin, J. D.,
Gunn-Moore, F. J., Vonsattel, J. P., Arancio, O., Chen, J. X., and Yan,
S. D. (2008) Cyclophilin D deficiency attenuates mitochondrial and
neuronal perturbation and ameliorates learning and memory in
Alzheimer’s disease, Nat. Med., 14, 1097-1105.
149.Schmalhausen, I. I. (1949) Factors of
Evolution: the Theory Stabilizing Selection, Blakiston,
Philadelphia.
150.Comfort, A. (1979) The Biology of
Senescence, 3rd Edn., Elsevier, N. Y.
151.Wood, S. H., Christian, H. C., Miedzinska, K.,
Saer, B. R. C., Johnson, M., Paton, B., Yu, L., McNeilly, J., Davis, J.
R. E., McNeilly, A. S., Burt, D. W., and Loudon, A. S. I. (2015) Binary
switching of calendar cells in the pituitary defines the phase of the
circannual cycle in mammals, Curr. Biol., 25,
2651-2662.
152.Migliaccio, E., Giorgio, M., Mele, S., Pelicci,
G., Reboldi, P., Pandolfi, P. P., Lanfrancone, L., and Pelicci, P. G.
(1999) The p66shc adaptor protein controls oxidative stress response
and life span in mammals, Nature, 402, 309-313.
153.Trinei, M., Giorgio, M., Cicalese, A., Barozzi,
S., Ventura, A., Migliaccio, E., Milia, E., Padura, I. M., Raker, V.
A., Maccarana, M., Petronilli, V., Minucci, S., Bernardi, P.,
Lanfrancone, L., and Pelicci, P. G. (2002) A p53-p66Shc signalling
pathway controls intracellular redox status, levels of
oxidation-damaged DNA and oxidative stress-induced apoptosis,
Oncogene, 21, 3872-3878.
154.Napoli, C., Martin-Padura, I., De Nigris, F.,
Giorgio, M., Mansueto, G., Somma, P., Condorelli, M., Sica, G., De
Rosa, G., and Pelicci, P. (2003) Deletion of the p66Shc longevity gene
reduces systemic and tissue oxidative stress, vascular cell apoptosis,
and early atherogenesis in mice fed a high-fat diet, Proc. Natl.
Acad. Sci. USA, 100, 2112-2116.
155.Giorgio, M., Migliaccio, E., Paolucci, D.,
Orsini, F., Contursi, C., Moroni, M., Marcaccio, A., Paolucci, F., and
Pelicci, P. G. (2004) p66Shc Is a Signal Transduction Redox
Enzyme, 13th EBEC Meeting Abstr., p. 27.
156.Ku, H. H., Brunk, U. T., and Sohal, R. S.
(1993) Relationship between mitochondrial superoxide and hydrogen
peroxide production and longevity of mammalian species, Free Radic.
Biol. Med., 15, 621-627.
157.Park, T. J., Reznick, J., Peterson, B. L.,
Blass, G., Omerbasic, D., Bennett, N. C., Kuich, P., Zasada, C., Browe,
B. M., Hamann, W., Applegate, D. T., Radke, M. H., Kosten, T.,
Lutermann, H., Gavaghan, V., Eigenbrod, O., Begay, V., Amoroso, V. G.,
Govind, V., Minshall, R. D., Smith, E. S. J., Larson, J., Gotthardt,
M., Kempa, S., and Lewin, G. R. (2017) Fructose-driven glycolysis
supports anoxia resistance in the naked mole-rat, Science,
356, 307-311.
158.Kim, J., Song, G., Wu, G., and Bazer, F. W.
(2012) Functional roles of fructose, Proc. Natl. Acad. Sci. USA,
109, 1619-1628.
159.Randall, G. C., and L’Ecuyer, C. (1976)
Tissue glycogen and blood glucose and fructose levels in the pig fetus
during the second half of gestation, Biol. Neonate, 28,
74-82.
160.Alva, N., Alva, R., and Carbonell, T. (2016)
Fructose 1,6-bisphosphate: a summary of its cytoprotective mechanism,
Curr. Med. Chem., 23, 4396-4417.
161.Dills, W. L. (1993) Protein fructosylation:
fructose and the maillard reaction, Am. J. Clin. Nutr.,
58, 779-787.
162.Lustig, R. H. (2013) Fructose: it’s
“alcohol without the buzz”, Adv. Nutr., 4,
226-235.
163.Andziak, B., O’Connor, T. P., and
Buffenstein, R. (2005) Antioxidants do not explain the disparate
longevity between mice and the longest-living rodent, the naked
mole-rat, Mech. Ageing Dev., 126, 1206-1212.
164.Anisimov, V. N., Bakeeva, L. E., Egormin, P.
A., Filenko, O. F., Isakova, E. F., Manskikh, V. N., Mikhelson, V. M.,
Panteleeva, A. A., Pasyukova, E. G., Pilipenko, D. I., Piskunova, T.
S., Popovich, I. G., Roshchina, N. V., Rybina, O. Y., Saprunova, V. B.,
Samoylova, T. A., Semenchenko, A. V., Skulachev, M. V., Spivak, I. M.,
Tsybul’ko, E. A., Tyndyk, M. L., Vyssokikh, M. Y., Yurova, M. N.,
Zabezhinsky, M. A., and Skulachev, V. P. (2008) Mitochondria-targeted
plastoquinone derivatives as tools to interrupt execution of the aging
program. 5. SkQ1 prolongs lifespan and prevents development of traits
of senescence, Biochemistry (Moscow), 73, 1655-1670.
165.Yee, C., Yang, W., and Hekimi, S. (2014) The
intrinsic apoptosis pathway mediates the pro-longevity response to
mitochondrial ROS in C. elegans, Cell, 157,
897-909.
166.Skulachev, M. V., and Skulachev, V. P. (2014)
New data on programmed aging – slow phenoptosis, Biochemistry
(Moscow), 79, 977-993.
167.Mechnikov, I. I. (1964) Sketches of
Optimism [in Russian], AN SSSR, Moscow.