Effect of SkQ1 on Activity of the Glutathione System and NADPH-Generating Enzymes in an Experimental Model of Hyperglycemia
Ya. G. Voronkova1*, T. N. Popova1, A. A. Agarkov1, and R. A. Zinovkin2
1Voronezh State University, 394006 Voronezh, Russia; fax: +7 (473) 220-8755; E-mail: janavoronkova@yandex.ru2Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received July 21, 2015; Revision received August 28, 2015
We studied the effect of mitochondria-targeted antioxidant 10-(6′-plastoquinonyl)decyltriphenylphosphonium (SkQ1) on the antioxidant activity of the glutathione system and NADPH-generating enzymes in liver and blood serum of rats with hyperglycemia induced by protamine sulfate. It was found that intraperitoneal injection of SkQ1 prevented both decrease in reduced glutathione level and increase in activity of glutathione system enzymes – glutathione peroxidase, glutathione reductase, and glutathione transferase. Activity of NADPH-generating enzymes – glucose-6-phosphate dehydrogenase and NADP-isocitrate dehydrogenase – was also attenuated by SkQ1. Probably, in this model of hyperglycemia, decreased level of reactive oxygen species in mitochondria led to the decreased burden on the glutathione antioxidant system and NADPH-generating enzymes. Thus, SkQ1 appears to be a promising compound for the treatment and/or prevention of the adverse effects of hyperglycemia.
KEY WORDS: hyperglycemia, SkQ1, glutathione, glutathione peroxidase, glutathione reductase, glutathione transferaseDOI: 10.1134/S000629791512010X
Abbreviations: DM, diabetes mellitus; DM2, type 2 diabetes mellitus; G6PDH, glucose-6-phosphate dehydrogenase; GP, glutathione peroxidase; GR, glutathione reductase; GSH, reduced glutathione; GT, glutathione transferase; NADP-IDH, NADP-isocitrate dehydrogenase; SkQ1, 10-(6′-plastoquinonyl)decyltriphenylphosphonium.
The last two decades of research on diabetes mellitus (DM) have shown a
sharp increase in the prevalence of this endocrinopathy [1]. According to the International Diabetes
Federation, almost 250 million people currently suffer from this
disease, and by 2030 this figure could exceed 366 million [1]. Among patients with DM, 70% have type 2 diabetes
mellitus (DM2), which is the fifth leading cause of death in the world
[2]. DM2 decompensation can lead to the development
of such severe liver malfunctions as fatty liver infiltration and
subsequent ketoacidosis [3].
It is known that chronic hyperglycemia, being the main objective indication of DM2, is closely connected to the development of oxidative stress [4, 5] accompanied by excessive generation of free radicals [6]. These highly reactive compounds directly damage blood vessels, tissues, and organs. In cells, free radicals contribute to the activation of transcription factor NF-κB, cause apoptosis, impaired insulin secretion, and thrombogenic vascular wall transformation [7].
The glutathione peroxidase/glutathione reductase (GP/GR) antioxidative system, preventing adverse effects of oxidative stress, plays an important role in protection of the organism against damaging effects of free radicals. The antioxidant properties of reduced glutathione (GSH) are determined by its direct interaction with free radicals and hydroperoxides [8]. It should be noted that GSH formed in the glutathione reductase reaction acts as the acceptor of hydroxyl radical; it also inhibits lipid peroxidation at the stage of chain branching. Glutathione peroxidase is the key enzyme neutralizing reactive oxygen species and peroxidation products [9]. Glutathione reductase increases GSH level without increasing its synthesis [9]. Due to the conversion of reduced glutathione to the oxidized form, the glutathione system is centrally involved in maintaining thiol–disulfide balance in tissues, which is essential for such vital processes as the activity of membrane structures, cytoskeleton, cell division, and the regulation of the activity of peptide hormones [10].
Glutathione transferase (GT) is an important component of antioxidant protection. It detoxifies products of lipid peroxidation generated in endoplasmic reticulum during metabolism of xenobiotics [11].
Activity of the GP/GR system is limited by the concentration of NADPH used for reduction of glutathione by GR. The pentose phosphate pathway is one of the core suppliers of reducing equivalents for the GP/GR system. The key enzyme of this pathway, glucose-6-phosphate dehydrogenase (G6PDH), catalyzes the conversion of glucose-6-phosphate to gluconolactone-6-phosphate. Certain data suggest that there can be an alternative source of NADPH – the reaction of oxidative decarboxylation of isocitrate to 2-oxoglutarate catalyzed by NADP-isocitrate dehydrogenase (NADP-IDH) [6].
Chronic diseases are often accompanied by a lengthy oxidative stress, which leads to the depletion of the antioxidant system. This explains increased interest in research on compounds capable of regulating free radical homeostasis in the organism. It is known that increased level of mitochondrial reactive oxygen species plays an important role in the pathogenesis of DM2 [12]. According to some evidence, mitochondrial functions are impaired, and mitochondria are damaged and produce less ATP in patients with DM2 [13, 14]. Thus, attempts have been made to treat DM2 and hyperglycemia by suppressing the generation of excessive amounts of free radicals in mitochondria [15].
10-(6′-Plastoquinonyl)decyltriphenylphosphonium (SkQ1) is a plastoquinone connected by a hydrocarbon linker to a lipophilic cation [16]. This compound is a highly effective mitochondria-targeted antioxidant [17]. In vitro, SkQ1 prevents apoptosis at nanomolar concentrations [18], and in vivo it effectively prevents and treats a number of diseases associated with oxidative stress and aging [19, 20]. SkQ1 effectively prevented oxidative stress, production of reactive oxygen species, and decrease in amount of mitochondrial respiratory chain proteins in a murine model of DM2 caused by high fat diet [21].
The purpose of this study was to evaluate the contents of reduced glutathione, activity of glutathione system enzymes (GR, GP, GT), as well as enzymes supplying NADPH (G6PDH and NADP-IDH) in rat liver and blood serum in hyperglycemia induced by administration of protamine sulfate, and to study SkQ1 effect on these processes.
MATERIALS AND METHODS
Induction of hyperglycemia in rats. Male white laboratory rats (Rattus rattus L.) weighing 150-200 g were used in the studies. All experimental procedures were consistent with the requirements of international norms for humane treatment of animals reflected in the sanitary rules for the selection and maintenance of experimental-biological clinics (vivaria).
Hyperglycemia was induced by intramuscular injection of protamine sulfate (10 mg/kg of body weight) for 3 weeks in 0.5 ml of 0.9% NaCl, three times per day [22]. Three weeks after the beginning of hyperglycemia induction, narcotized animals were euthanized and used for further studies. In experiments, the animals were divided into three groups: group 1 (n = 12) – control animals; group 2 (n = 12) – rats with hyperglycemia induced by protamine sulfate administration; group 3 (n = 12) – animals with hyperglycemia treated by SkQ1 (SkQ1 solution was administered intraperitoneally in a dose of 1250 nmol/kg per day, one time per day, starting from the second week).
Biochemical tests. Blood samples were taken from the tail vein on the 15th, 17th, and 19th days of the experiment. The content of glucose in rat blood serum was measured by the glucose oxidase method using the Glucose-12-vital reagent kit (Vital-Diagnostics, Russia). The basic principle of the method is that glucose oxidase-mediated β-glucose oxidation by air oxygen leads to the formation of an equimolar amount of hydrogen peroxide. When affected by peroxidase, hydrogen peroxide oxidizes chromogenic substrates forming a colored product. The color intensity of this product is proportional to glucose concentration in the sample.
To obtain tissue homogenate, samples of liver tissue were homogenized in a 4-fold volume of a cold extraction buffer (0.1 M Tris-HCl, pH 7.8, 1 mM EDTA, 1% β-mercaptoethanol). The resulting extract was filtered through a layer of kapron with square cells (0.1 mm) and then centrifuged at 10,000g for 10 min to separate unbroken tissue elements. The supernatant was used for further studies.
Venous blood was collected into a clean glass test tube without anticoagulant and placed for 0.5 h into a thermostat at 37°C; after phase separation, the supernatant was collected and centrifuged at 4000g for 10 min. The resulting serum was used for further study.
GSH concentration was determined spectrophotometrically at 412 nm using Ellman’s reagent [23]. The sulfhydryl group of reduced glutathione reacts with 5,5-dithio-bis-(2-nitrobenzoic) acid (Ellman’s reagent) with the formation of equimolar amounts of yellow-colored thionitrophenyl anion with absorption maximum at 412 nm.
GP activity was measured using the coupled enzymatic reaction in the following spectrophotometric medium: 50 mM potassium phosphate buffer, pH 7.4, 1 mM EDTA, 0.12 mM NADPH, 0.85 mM GSH, 0.37 mM H2O2, 1 U/ml GR. The control sample contained no GSH.
GR activity was measured in spectrophotometric medium containing 50 mM potassium phosphate buffer, pH 7.4, 1 mM EDTA, 0.16 mM NADPH, 0.8 mM oxidized glutathione.
GT activity was measured in the following medium: 0.1 M potassium phosphate buffer, pH 7.4, 1 mM EDTA, 1 mM 1-Cl-2,4-dinitrobenzene, 5 mM GSH.
G6PDH activity was measured using 50 mM Tris-HCl buffer, pH 7.8, containing 3 mM glucose-6-phosphate, 0.25 mM NADP, 1 mM MnCl2.
NADP-IDH activity was measured in the following medium: 50 mM Tris-HCl buffer (pH 7.6-7.8), 1.5 mM isocitrate, 2 mM MnCl2, 0.25 mM NADP, 0.1 mM EDTA. The reaction was initiated by the addition of the enzyme preparation.
Activities of the enzymes GP, GR, GT, G6PDH, and NADP-IDH were determined spectrophotometrically at 340 nm using a Hitachi U-1900 instrument (Hitachi High-Technologies, Japan).
One unit of enzymatic activity (U) was taken as the amount of enzyme catalyzing the formation of 1 µmol of reaction product or transformation of 1 µmol of substrate per min at 25°C. Total protein was determined by the biuret method.
Analysis of results. Experiments were repeated at least 12 times (biological repetitiveness) and two times (analytical repetitiveness). Experimental results were compared to control. The data were processed using the Student’s t-test calculating the mean and standard deviation; the differences were considered significant at p ≤ 0.05.
RESULTS AND DISCUSSION
Previously, we have shown that SkQ1 prevents increase in blood glucose level caused by administration of protamine sulfate [24]. In the current study, we analyzed biochemical changes in liver and blood serum occurring in this model.
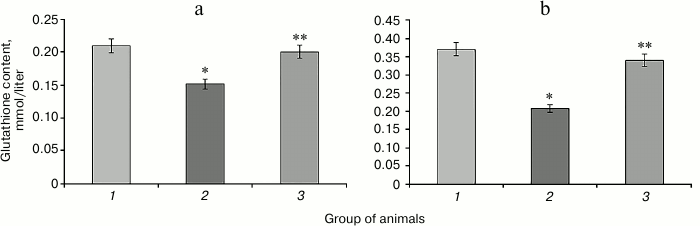
Injection of protamine sulfate caused 1.4-fold reduction in GSH content in liver and 1.8-fold reduction in blood serum relative to control values (Fig. 1). This might be related to a significant increase in expenditure of this metabolite under intensified free radical oxidation in DM. In addition, there could be another cause of the decrease in glutathione concentration: reduction in the activity of the pentose phosphate pathway in DM, resulting in decreased availability of NADPH needed for the regeneration of reduced glutathione [25]. Violation of glucose metabolism triggers the development of free radical oxidation in DM. Hyperglycemia activates numerous cellular signaling mechanisms: enhancement of the polyol pathway of glucose metabolism, which depletes cytosolic NADPH level and subsequently GSH, increase in glucose autoxidation accompanied by the formation of advanced glycation end products (AGEs), which damage proteins, violate their functions, and activate AGE-receptors using reactive oxygen species as secondary messengers leading to protein kinase C activation, followed by the enhancement of hyperglycemia and tissue hypoxia [26].
Fig. 1. Glutathione content in liver (a) and blood serum (b) of rats of the control group (1), animals with hyperglycemia induced by administration of protamine sulfate (2), and animals with the pathology treated by SkQ1 (3). The figure shows mean values and standard deviations. Differences at p ≤ 0.05: * relative to the control group; ** relative to the group with experimental hyperglycemia.
Administration of SkQ1 prevented the reduction in glutathione content in rat liver and blood serum that is observed in hyperglycemia caused by protamine sulfate injection (Fig. 1). This can be explained by the decrease in the intensity of free radical oxidation in animals treated with this compound. It is known that mitochondria-targeted antioxidants of the SkQ family can “break” the chain reactions of free radical formation, preventing the decrease in reduced glutathione level in cells [17], thereby protecting cells from oxidative stress [27].
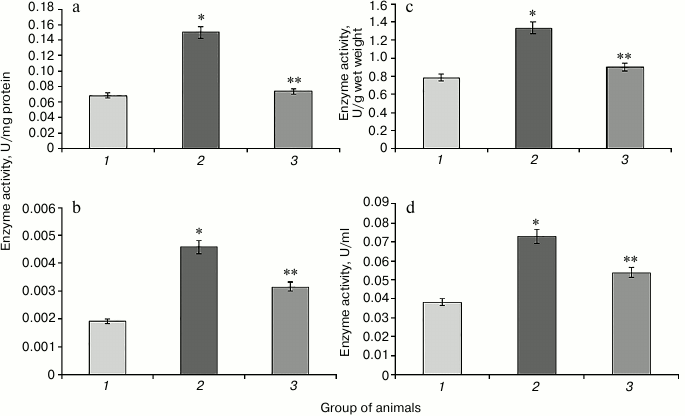
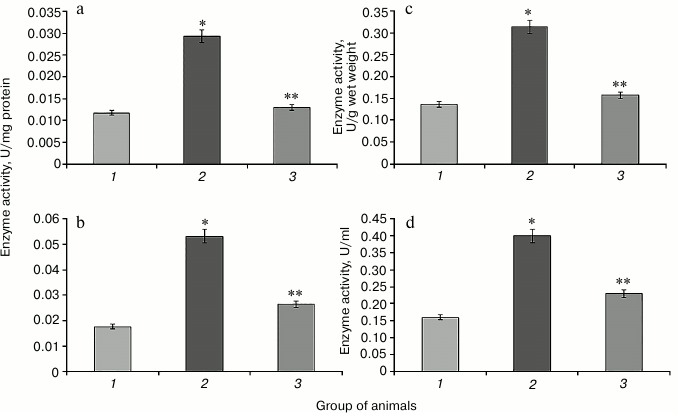
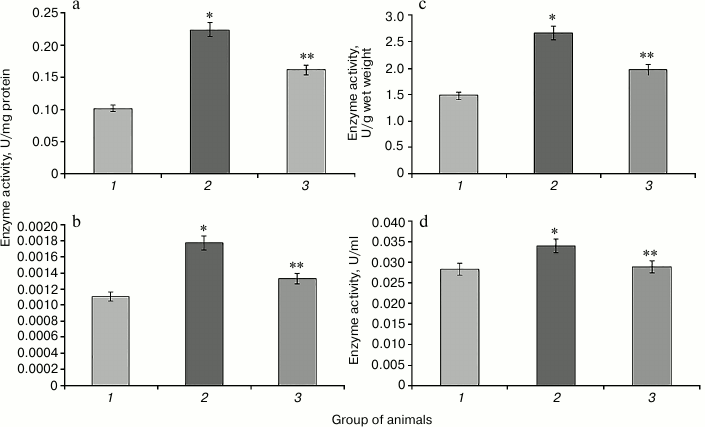
Hyperglycemia induced by administration of protamine sulfate also resulted in significant activation of enzymatic reactions of the glutathione system: GR, GP, and GT (Figs. 2-4, respectively). The stimulation of the glutathione system was probably caused by excessive formation of reactive oxygen species during the development of hyperglycemia. Amplification of free radical processes is likely to result from the activation of the polyol shunt, in which aldose reductase catalyzes the transformation of glucose into sorbitol. This process involves consumption of a large amount of NADPH required for reduction of glutathione. Sorbitol dehydrogenase catalyzes transformation of sorbitol into fructose, which is accompanied by an increase in NADPH/NADP ratio. This condition is called “reductive stress” or “hyperglycemic pseudo-hypoxia”, because similar changes occur in the development of tissue hypoxia [28].
Administration of SkQ1 was accompanied by a significant decrease in the activity of the enzymes of the glutathione system in rats with experimental hyperglycemia induced by protamine sulfate (Figs. 2-4). It is logical to assume that these effects of SkQ1 are directly related to its antioxidant properties, which contributed to reducing the burden on the studied enzymes.
Fig. 2. Activity of glutathione reductase in rats of the control group (1), rats with hyperglycemia induced by administration of protamine sulfate (2), and rats with the pathology treated by SkQ1 (3). a) Specific activity of the enzyme in liver; b) specific activity of the enzyme in blood serum; c) activity of the enzyme expressed in U/g liver wet weight; d) activity of the enzyme expressed U/ml blood serum. The figure shows mean values and standard deviations. Differences at p ≤ 0.05: * relative to the control group; ** relative to the group with experimental hyperglycemia.
Fig. 3. Activity of glutathione peroxidase. Designations as in Fig. 2.
Fig. 4. Activity of glutathione transferase. Designations as in Fig. 2.
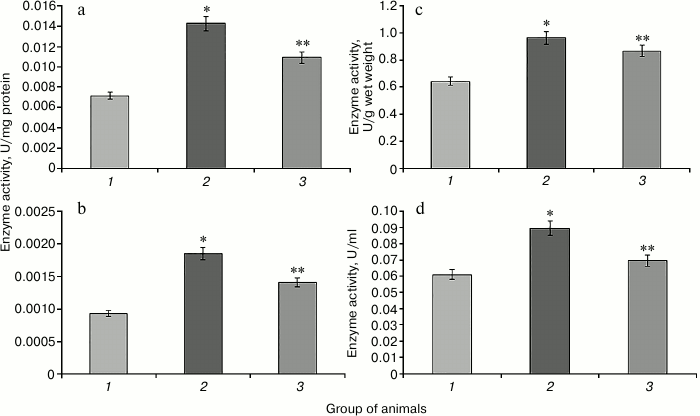
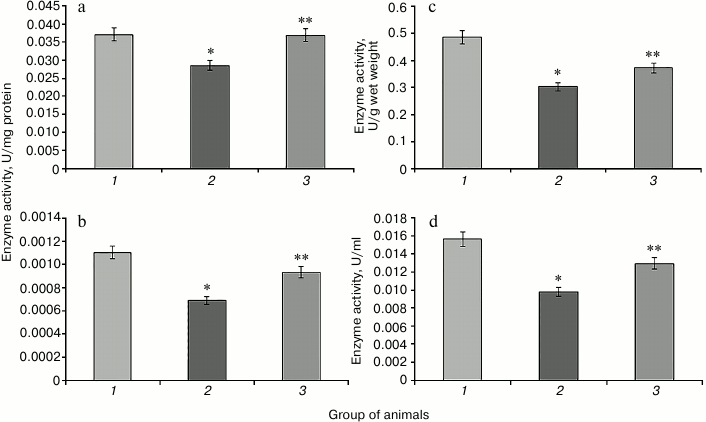
Increase in the activity of NADP-IDH in rat liver and blood serum is observed in hyperglycemia induced by protamine sulfate (Fig. 5). This enzyme is one of the NADPH suppliers for the glutathione system, which apparently has adaptive character reflecting the need to provide reducing equivalents for this system under oxidative stress. SkQ1 prevented the increase in NADP-IDH activity (Fig. 5). This might be related to the ability of the antioxidant to decrease excessive production of reactive oxygen species inhibiting the activity of the key enzymes of the Krebs cycle [16], which is accompanied by change in activity of the studied enzyme in the direction of control values.
Fig. 5. Activity of NADP-isocitrate dehydrogenase. Designations as in Fig. 2.
It is known that the activity of G6PDH significantly decreases in many tissues in experimental DM2. This phenomenon might be due to several causes. The level of glucose in blood in known to play an important role in the regulation of G6PDH activity [29]. Hyperglycemia might inhibit this enzyme activity due to increased activity of adenylate cyclase leading to cAMP accumulation and activation of protein kinase A (PKA), which phosphorylates and inhibits G6PDH. Another mechanism of G6PDH regulation involves enhancement of protein glycation in hyperglycemia, which leads to a sharp decrease in G6PDH activity [30]. Protamine sulfate caused significant reduction in G6PDH activity compared to control (Fig. 6). In the case of animals with pathology, administration of SkQ1 also changed the activity of this enzyme towards control values (Fig. 6).
Fig. 6. Activity of glucose-6-phosphate dehydrogenase. Designations as in Fig. 2.
Apparently, SkQ1-mediated normalization of free radical homeostasis of an organism suffering from DM2 resulted in the recovery of the activity of the pentose phosphate pathway, which was reflected in increased G6PDH activity.
According to the results of our study, the use of mitochondria-targeted antioxidant SkQ1 in rats with hyperglycemia caused by protamine sulfate leads to decrease in the activity of glutathione system enzymes, and the increase in GSH content in tissues when compared to these parameters in the pathology. Apparently, the antioxidant effect of the protector due to the suppression of free radicals in mitochondria reduces pressure on the antioxidant system, leading to decrease in activation of its glutathione part and reduction in NADPH required for the functioning of this system. Thus, the use of mitochondria-targeted antioxidants to prevent and eliminate the negative effects of oxidative stress caused by hyperglycemia seems to be very promising.
We express gratitude to the director of the Belozersky Institute of Physico-Chemical Biology, Academician V. P. Skulachev, for his interest in this study and valuable comments during the preparation of the article.
This work was supported by the Russian Science Foundation (project No. 14-24-00107).
REFERENCES
1.Stumvoll, M., Goldstein, B. J., and Van Haeften, T.
W. (2005) Type 2 diabetes: principles of pathogenesis and therapy,
Lancet, 365, 1333-1346.
2.Roglic, G., Unwin, N., Bennett, P. H., Mathers, C.,
Tuomilehto, J., Nag, S., Connolly, V., and King, H. (2005) The burden
of mortality attributable to diabetes realistic estimates for the year
2000, Diabetes Care, 28, 2130-2135.
3.Dedov, I. I., Balabolkin, M. I., and Klebanova, Ye.
M. (2004) Current aspects of transplantation of pancreatic islets in
diabetes, Sakhar. Diabet, 2, 34-41.
4.Osipov, A., Azizova, O., and Vladimirov, Yu. (1990)
Reactive oxygen species and their role in the organism, Usp. Biol.
Khim., 31, 180-208.
5.Vincent, A. M., Russell, J. W., Low, P., and
Feldman, E. L. (2004) Oxidative stress in the pathogenesis of diabetic
neuropathy, Endocrin. Rev., 25, 612-628.
6.Popova, T. N., Pashkov, A. N., Semehikhina, A. V.,
Popov, S. S., and Pakhmanova, T. I. (2008) Free-Radical Processes in
Biosystems: a Textbook [in Russian], Kirillitsa, Stary Oskol.
7.Balabolkin, M. I. (2002) The role of protein
glycation, oxidative stress in pathogenesis of vascular complications
in diabetes, Sakhar. Diabet, 4, 4-16.
8.Lu, S. C. (1999) Regulation of hepatic glutathione
synthesis: current concepts and controversies, FASEB J.,
13, 1169-1183.
9.Upton, J., Edens, F., and Ferket, P. (2009) The
effects of dietary oxidized fat and selenium source on performance,
glutathione peroxidase, and glutathione reductase activity in broiler
chickens, J. Appl. Poultry Res., 18, 193-202.
10.Mannervik, B. (1980) Thioltransferases, in
Enzymatic Basis of Detoxication (Jacoby, W., ed.) Academic
Press, New York, Vol. 2, pp. 229-244.
11.Habig, W. H., Pabst, M. J., and Jakoby, W. B.
(1974) Glutathione S-transferases. The first enzymatic step in
mercapturic acid formation, J. Biol. Chem., 249,
7130-7139.
12.Schrauwen, P., and Hesselink, M. K. (2004)
Oxidative capacity, lipotoxicity, and mitochondrial damage in type 2
diabetes, Diabetes, 53, 1412-1417.
13.Kelley, D. E., He, J., Menshikova, E. V., and
Ritov, V. B. (2002) Dysfunction of mitochondria in human skeletal
muscle in type 2 diabetes, Diabetes, 51, 2944-2950.
14.Petersen, K. F., Dufour, S., Befroy, D., Garcia,
R., and Shulman, G. I. (2004) Impaired mitochondrial activity in the
insulin-resistant offspring of patients with type 2 diabetes, N.
Engl. J. Med., 350, 664-671.
15.Green, K., Brand, M. D., and Murphy, M. P. (2004)
Prevention of mitochondrial oxidative damage as a therapeutic strategy
in diabetes, Diabetes, 53, 110-118.
16.Antonenko, Y. N., Roginsky, V., Pashkovskaya, A.,
Rokitskaya, T., Kotova, E., Zaspa, A., Chernyak, B., and Skulachev, V.
(2008) Protective effects of mitochondria-targeted antioxidant SkQ in
aqueous and lipid membrane environments, J. Membr. Biol.,
222, 141-149.
17.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L.
E., Chernyak, B. V., Vhertkov, V. A., Domnina, L. V., Ivanova, O. Yu.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pushkovskaya, A.
A., Pletyushkina, O. Yu., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaaaeva, I. V., Tashlitsky, V. N., Vassiliev, I. M., Wyssololh, M.
Yu., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Mitochondria-targeted plastoquinone derivative as tools to
interrupt execution of the aging program. 1. Cationic plastoquinone
derivatives: synthesis and in vitro studies, Biochemistry
(Moscow), 73, 1273-1287.
18.Galkin, I. I., Pletjushkina, O. Y., Zinovkin, R.
A., Zakharova, V. V., Birjukov, I. S., Chernyak, B. V., and Popova, E.
N. (2014) Mitochondria-targeted antioxidants prevent TNFα-induced
endothelial cell damage, Biochemistry (Moscow) 79,
124-130.
19.Plotnikov, E. Y., Morosanova, M. A., Pevzner, I.
B., Zorova, L. D., Manskikh, V. N., Pulkova, N. V., Galkina, S. I.,
Skulachev, V. P., and Zorov, D. B. (2013) Protective effect of
mitochondria-targeted antioxidants in an acute bacterial infection,
Proc. Natl. Acad. Sci. USA, 110, 3100-3108.
20.Skulachev, V. P. (2013) Cationic antioxidants as
a powerful tool against mitochondrial oxidative stress, Biochem.
Biophys. Res. Commun., 441, 275-279.
21.Paglialunga, S., Van Bree, B., Bosma, M.,
Valdecantos, M. P., Amengual-Cladera, E., Jorgensen, J. A., Van
Beurden, D., Den Hartog, G. J., Ouwens, D. M., Briede, J. J.,
Schrauwen, P., and Hoeks, J. (2012) Targeting of mitochondrial reactive
oxygen species production does not avert lipid-induced insulin
resistance in muscle tissue from mice, Diabetologia, 55,
2759-2768.
22.Ulyanov, A. M., and Tarasov, Yu. A. (2000)
Insular system in animals with chronic heparin shortage, Vopr. Med.
Khim., 46, 149-154.
23.Buzlama, V. S. (1997) Manual to Study the
Processes of Lipid Peroxidation and Systems of Antioxidant Protection
in Animals [in Russian], VNIVIPFiT, Voronezh, p. 35.
24.Voronkova, Ya. G., Popova, T. N., Agarkov, A. A.,
and Skulachev, M. V. (2015) Effect of
10-(6′-plastoquinonyl)decyltriphenylphosphonium (SkQ1) on
oxidative status in rats with hyperglycemia induced by administration
of protamine sulfate, Biochemistry (Moscow), 80,
1606-1613.
25.McLennan, S. V., Heffernan, S., Wright, L., Rae,
C., Fisher, E., Yue, D. K., and Turtle, J. R. (1991) Changes in hepatic
glutathione metabolism in diabetes, Diabetes, 40,
344-348.
26.Menshikova, E. B., Lankin, V. Z., Zenkov, N. K.,
Bondar, I. A., Krugovykh, N. F., and Trufakin, V. A. (2006)
Oxidative Stress. Prooxidants and Antioxidants [in Russian],
Slovo, Moscow.
27.Fetisova, E. K., Avetisyan, A. V., Izyumov, D.
S., Korotetskaya, M. V., Tashlitsky, V. N., Skulachev, V. P., and
Chernyak, B. V. (2011) P-glycoprotein providing multidrug resistance,
prevents the expression of anti-apoptotic effect of
mitochondria-targeted antioxidant SkQR1, Tsitologiya, 53,
488-497.
28.Baynes, J. W., and Thorpe, S. R. (1999) Role of
oxidative stress in diabetic complications: a new perspective on an old
paradigm, Diabetes, 48, 1-9.
29.Zhang, Z., Apse, K., Pang, J., and Stanton, R. C.
(2000) High glucose inhibits glucose-6-phosphate dehydrogenase via cAMP
in aortic endothelial cells, J. Biol. Chem., 275,
40042-40047.
30.Bulteau, A.-L., Verbeke, P., Petropoulos, I.,
Chaffotte, A.-F., and Friguet, B. (2001) Proteasome inhibition in
glyoxal-treated fibroblasts and resistance of glycated
glucose-6-phosphate dehydrogenase to 20S proteasome degradation in
vitro, J. Biol. Chem., 276, 45662-45668.