Uncoupling and Toxic Action of Alkyltriphenylphosphonium Cations on Mitochondria and the Bacterium Bacillus subtilis as a Function of Alkyl Chain Length
L. S. Khailova1, P. A. Nazarov1, N. V. Sumbatyan2, G. A. Korshunova1, T. I. Rokitskaya1, V. I. Dedukhova1, Yu. N. Antonenko1*, and V. P. Skulachev1,3
1Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, 119991 Moscow, Russia; E-mail: antonen@belozersky.msu.ru; antonen@genebee.msu.ru2Lomonosov Moscow State University, Faculty of Chemistry, 119991 Moscow, Russia
3Lomonosov Moscow State University, Institute of Mitoengineering, 119991 Moscow, Russia
* To whom correspondence should be addressed.
Received February 17, 2015; Revision received July 8, 2015
A series of permeating cations based on alkyl derivatives of triphenylphosphonium (Cn-TPP+) containing linear hydrocarbon chains (butyl, octyl, decyl, and dodecyl) was investigated in systems of isolated mitochondria, bacteria, and liposomes. In contrast to some derivatives (esters) of rhodamine-19, wherein butyl rhodamine possessed the maximum activity, in the case of Cn-TPP a stimulatory effect on mitochondrial respiration steadily increased with growing length of the alkyl radical. Tetraphenylphosphonium and butyl-TPP+ at a dose of several hundred micromoles exhibited an uncoupling effect, which might be related to interaction between Cn-TPP+ and endogenous fatty acids and induction of their own cyclic transfer, resulting in transport of protons across the mitochondrial membrane. Such a mechanism was investigated by measuring efflux of carboxyfluorescein from liposomes influenced by Cn-TPP+. Experiments with bacteria demonstrated that dodecyl-TPP+, decyl-TPP+, and octyl-TPP+ similarly to quinone-containing analog (SkQ1) inhibited growth of the Gram-positive bacterium Bacillus subtilis, wherein the inhibitory effect was upregulated with growing lipophilicity. These cations did not display toxic effect on growth of the Gram-negative bacterium Escherichia coli. It is assumed that the difference in toxic action on various bacterial species might be related to different permeability of bacterial coats for the examined triphenylphosphonium cations.
KEY WORDS: permeating cation, SkQ1, mitochondria, uncoupling, bacteria, cytotoxicityDOI: 10.1134/S000629791512007X
Abbreviations: CF, 5(6)-carboxyfluorescein; Cn-TPP+, alkyltriphenylphosphonium cation; DiS-C3-(5), potential-dependent carbocyanine probe; ΔµH+, mitochondrial transmembrane proton electrochemical gradient; ΔpH, pH gradient across the inner membrane of mitochondria; Δψ, mitochondrial membrane potential; FCCP, carbonyl cyanide p-trifluoromethoxyphenylhydrazone; Phe4P+, tetraphenylphosphonium cation; SkQ1, 10-(plastoquinonyl)decyltriphenylphosphonium.
Mitochondria-targeted antioxidants have become widespread not only in
studies investigating mitochondria in various physiological processes,
but also as therapeutic agents [1, 2]. These compounds are conjugates of some known
antioxidant (plastoquinone, ubiquinone, vitamin E, resveratrol) and a
permeating cation (triphenylphosphonium, rhodamine, etc.). Along with
antioxidant effect, such agents uncouple oxidative phosphorylation in
mitochondria, which is manifested as enhanced respiration and decrease
in membrane potential. It turned out that such an uncoupling effect in
some cases might not only be nontoxic, but also provide a protective
effect via partial mitochondrial uncoupling under pathologies related
to oxidative stress [3]. Such a phenomenon could be
due to dependence between generation of reactive oxygen intermediates
on membrane potential, and Korshunov et al. [4]
demonstrated this for the first time. Most probably, the mechanism of
the uncoupling effect of SkQ1 (a conjugate containing plastoquinone and
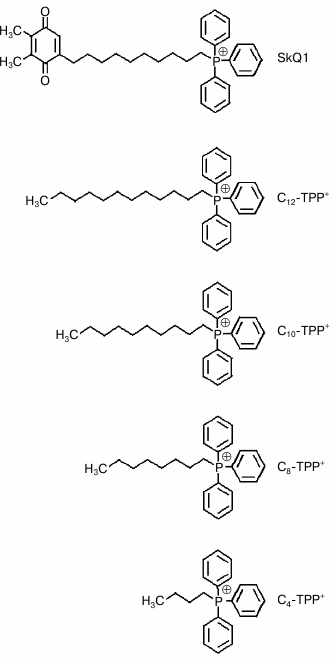
triphenylphosphonium connected via a decane linker; Fig. 1) comes down to its ability to interact with
endogenous fatty acids, thereby facilitating diffusion of fatty acid
anions across the mitochondrial membrane [5]. The
nature of such interaction is mostly electrostatic between the SkQ1
cation and fatty acid anions. Due to the fact that protonated fatty
acids easily permeate through membranes, such interaction allows the
transfer of protons similarly to routine protonophores such as
2,4-dinitrophenol. It was demonstrated that SkQ1 and its quinone-free
analog, C12TPP, exhibit an uncoupling effect on mitochondria
in a similar way [5]. In addition, it was suggested
that SkQ1 and its analogs at high concentrations were able to display
detergent effect, which, in turn, might stimulate respiration and
decrease mitochondrial potential [6].
Fig. 1. Chemical structures of cationic SkQ1 and Cn-TPP+.
In a series of studies with rhodamine-19-containing hydrophobic cations and hydrocarbon chains of various length, it was demonstrated that for cations with short chains an uncoupling effect might result from their specific interaction with protein transporters in the inner mitochondrial membrane [7]. The most active was the butyl ester of rhodamine-19 (C4R1), whereas more hydrophobic analogs of rhodamine-19 (C8R1, C10R1, C12R1) were shown to be much less effective. Zoratti et al. demonstrated that resveratrol conjugated with triphenylphosphonium was able to uncouple isolated mitochondria or mitochondria inside cells via interacting with ATPase, which mediated proton efflux [8]. The latter was suggested by data showing that an uncoupling effect induced by permeating cations was sensitive to oligomycin specifically inhibiting ATPase. Altogether, these data motivated us to return to an issue regarding the mechanism of an uncoupling effect for SkQ1, which at least partially could be accomplished independently of fatty acids through SkQ1 specifically interacting with protein transporters at the inner mitochondrial membrane. For this, we synthesized alkyl derivatives of triphenylphosphonium (Cn-TPP+) containing hydrocarbon chain of various length, such as n-butyl-, n-octyl-, n-decyl-, and n-dodecyl-triphenylphosphonium (Fig. 1). Moreover, during our study we also examined the effects of these hydrophobic cations on bacteria, which could result in uncoupling of oxidative phosphorylation followed by suppressing their growth due to similarity of bioenergetics processes between bacteria and mitochondria. It was noted that earlier results demonstrated that permeating cations of plant origin such as berberine and palmatine display strong antibacterial effect [9, 10]. Moreover, it was shown that some quaternary phosphonium salts also have antibacterial activity [11-13]. Thus, the task of this study was to examine the effect of two compounds derived from Cn-TPP+ on mitochondria and bacteria. In particular, it was demonstrated that Cn-TPP as well as SkQ1 had antibacterial effect on Bacillus subtilis, whereas E. coli was resistant to Cn-TPP+ and SkQ1. Uncoupling and toxic effects of Cn-TPP+ were enhanced with growing length of alkyl chain, thereby suggesting that these compounds lack specific interaction with protein transporters.
MATERIALS AND METHODS
Synthesis of n-butyl-triphenylphosphonium (C4-TPP+), n-octyl-triphenylphosphonium (C8-TPP), n-decyl-triphenylphosphonium (C10-TPP), and n-dodecyl-triphenylphosphonium (C12-TPP; Fig. 1) bromides was done by adding the appropriate alkyl bromide acting on triphenylphosphine according to earlier described methods [14].
Unless specified, all other reagents were purchased from Sigma-Aldrich (USA).
Carboxyfluorescein efflux from liposomes. Liposomes loaded with 5(6)-carboxyfluorescein (CF) (Sigma) at concentration of fluorescence self-quenching were prepared from total E. coli-derived lipids (Avanti Polar Lipids, USA) suspended in 100 mM CF solution by extrusion through a polycarbonate filter with 100-nm pores as previously described [15]. The final liposomes were washed free from unbound CF by passing through a column with Sephadex G-50 using buffer containing 100 mM KCl, 10 mM Tris, 10 mM MES, pH 7.4. To measure CF efflux, liposomes were diluted to final concentration 5 µg/ml in the same buffer. Then fluorescence was measured at 520 nm (excitation at 490 nm) using a Panorama fluorimeter (Lumex, Russia). At the end of each experiment, 0.1% Triton X-100 solution was added to record complete efflux of CF. The magnitude of CF efflux was calculated as follows: (Ft – F0)/(F100 – F0), where F0 and Ft – fluorescence intensity at initial and random time points, F100 – fluorescence intensity after complete efflux of CF after adding Triton X-100.
Mitochondria from rat liver were isolated as described earlier [16]. Then the mitochondria were resuspended in media containing 250 mM sucrose, 5 mM MOPS-KOH, pH 7.4, and 1 mM EGTA. Protein concentration was measured using the biuret method with BSA as a standard protein.
Mitochondrial respiration rate was measured by polarographic analysis with a Clark-type electrode on a Strathkelvin Instruments unit (USA), model 872, at 25°C. The incubation medium contained 250 mM sucrose, 5 mM MOPS-KOH, pH 7.4, and 1 mM EGTA. The concentration of mitochondria was 0.6-0.7 mg protein per ml. Oxygen consumption is presented as O2 nmol/min per mg protein.
Mitochondrial membrane potential was measured using the safranine-O probe [17]. The assay medium contained 250 mM sucrose, 5 mM MOPS-KOH, pH 7.4, 1 mM EGTA, 5 mM succinate, 2 µM rotenone, oligomycin (1 µg/ml), and 15 µM safranine-O. The concentration of mitochondria was 0.6-0.7 mg protein per ml. Absorbance was measured at 555 versus 523 nm using an Aminco DW-2000 spectrophotometer in dual wavelength mode.
Bacteria were grown in LB medium overnight at 37°C in a shaker (200 rpm) until reaching optical density 1.5 (at 600 nm), which was measured on an Ultrospec 1100 pro spectrophotometer (Amersham Biosciences Corp., USA). To plot bacterial growth curves, cells from overnight culture were seeded into fresh LB medium and cultured for 5-24 h at 37°C in a shaker (200 rpm) with periodic measurement of absorbance at 600 nm.
Membrane potential in Bacillus subtilis was measured by estimating fluorescence of the potential-dependent probe DiS-C3-(5) [18]. Bacillus subtilis strain BR151 from the overnight culture were seeded into fresh LB medium followed by growth for 2-4 h until reaching optical density 0.8 at 600 nm. Then the bacteria were diluted 20-fold in a buffer containing 100 mM KCl, 10 mM Tris, pH 7.4. Fluorescence was measured at 690 nm (excitation at 622 nm) by using a Panorama Fluorat-02 fluorimeter.
RESULTS
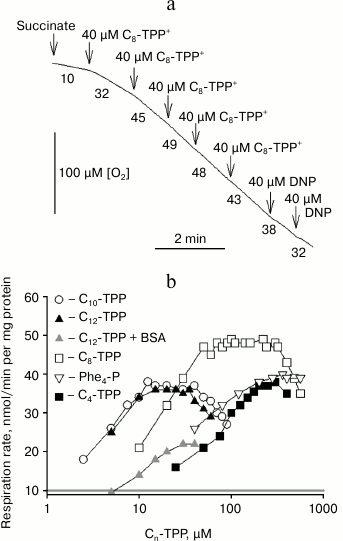
Uncoupling effect of alkyltriphenylphosphonium cations on mitochondria. Previously, it was shown that micromolar concentrations of SkQ1 or C12-TPP+ were able to stimulate respiration and decrease membrane potential in rat liver mitochondria and yeasts [6, 19]. A representative kinetic curve of oxygen consumption by rat liver mitochondria after adding increasing concentrations of C8-TPP+ is shown in Fig. 2a. It was found that after stimulating respiration in response to ~100 µM C8-TPP+, further increase in C8-TPP+ concentration no longer augmented respiration. The dependences between rate of rat mitochondrial respiration and concentrations of C12-TPP+ and other Cn-TPP+ of various alkyl chain length are presented in Fig. 2b. In contrast to rhodamine-19 esters, wherein butyl ester displayed the maximum activity, the uncoupling effect for Cn-TPP+ increased with growing length of the alkyl radical. By examining tetraphenylphosphonium or C4-TPP+, an uncoupling effect was reached only at high concentrations. The maximum uncoupling effect was observed at relatively low concentration of long-chain Cn-TPP+, whereas for tetraphenylphosphonium cation and C4-TPP+ the maximum mitochondrial uncoupling was detected at more than one order higher concentration. According to this, the half-maximal effective concentration for Cn-TPP+ in mitochondria decreased with increasing alkyl chain length. In addition, the activity of C12-TPP+ in incubation medium containing BSA (gray curve) is depicted in Fig. 2b. Note that BSA significantly reduces C12-TPP+-stimulated mitochondrial respiration.
Fig. 2. a, b) Dependence of mitochondrial respiration rate in rat liver mitochondria on concentration of permeating Cn-TPP+ cations. Succinate (5 mM) was used as a respiratory substrate in the presence of rotenone (2 µM). Representative polarographic data on oxygen consumption in mitochondria after adding C8-TPP at increasing concentrations are presented in panel (a). The incubation medium contained 250 mM sucrose, 5 mM MOPS-KOH, pH 7.4, and 1 mM EGTA.
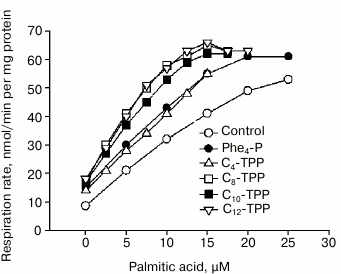
The dependence of mitochondrial respiration rate on palmitate concentration in control and 5 µM Cn-TPP+-treated mitochondria is shown in Fig. 3. It was found that Cn-TPP+ stimulated the palmitate-mediated uncoupling effect, and the efficiency of the stimulation increased with increasing value of n, reaching saturation at n = 10. These data support the hypothesis that endogenous fatty acids are involved in the uncoupling process.
Fig. 3. Influence of Cn-TPP+ (5 µM) on dependence between mitochondrial respiration rate and concentration of palmitic acid. The experiment was done under conditions noted in the legend to Fig. 2.
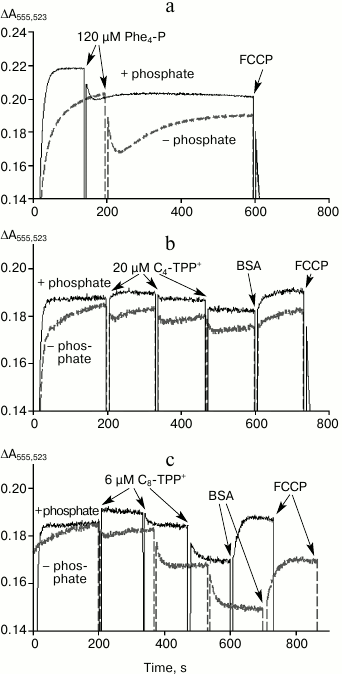
Because Cn-TPP+ cations must easily penetrate through lipid membranes in an electrophoretic manner, then at the initial time point adding them to mitochondria should result in a decrease in membrane potential and stimulated respiration. In the absence of interaction with fatty acids, the magnitude of membrane potential and respiration rate should be restored after accumulation of cations inside the mitochondria is completed. Changes in mitochondrial potential measured by applying the potential-sensitive probe safranin after adding tetraphenylphosphonium cations C4-TPP+ and C8-TPP+ with/without 1 mM phosphate are shown in Fig. 4. Adding 120-µM tetraphenylphosphonium in the absence of phosphate resulted in a decrease in Δψ, which then was restored within several minutes (Fig. 4a, dashed line). A similar effect was observed after adding 20 µM C4-TPP+ and 6 µM C8-TPP+ (Fig. 4, b and c, dashed lines). In the presence of phosphate, no time-dependent decrease and restoration of membrane potential was detected, apparently due to very rapid kinetics of such processes (solid lines). Adding BSA into the medium resulted in partial restoration of membrane potential, especially in the presence of phosphate (Fig. 4, b and c). Accelerated recovery of membrane potential after adding a permeating cation in the presence of phosphate was documented earlier in case of calcium ions, which easily permeate through mitochondrial membrane via protein transporters [20]. This effect was linked to activity of electroneutral phosphate transporter, which rapidly restores pH magnitude in the matrix, thus preventing generation of a large pH gradient on the inner mitochondrial membrane [21]. In accordance with this, the magnitude of membrane potential was higher in the presence of phosphate (Fig. 4). Further increase in concentration of C4-TPP+ or C8-TPP+ results in an irreversible decrease in membrane potential, even in the presence of phosphate (Fig. 4, b and c), which can be interpreted as initiation of an uncoupling mechanism involving fatty acids.
Fig. 4. Influence of tetraphenylphosphonium (Phe4-P) (a), C4-TPP+ (b), and C8-TPP+ (c) on mitochondrial potential estimated using safranin with/without phosphate (1 mM KH2PO4, bold and dashed line, respectively) in the assay medium. Y-axis: difference in absorbance at 555 and 523 nm. The mitochondria were energized by adding succinate (5 mM) in the presence of rotenone (2 µM). The incubation medium contained 250 mM sucrose, 5 mM MOPS, 1 mM EGTA, 1 µM oligomycin, pH 7.4, 0.3 mg/ml BSA, and 200 nM FCCP. The concentration of mitochondrial protein was 0.6 mg/ml.
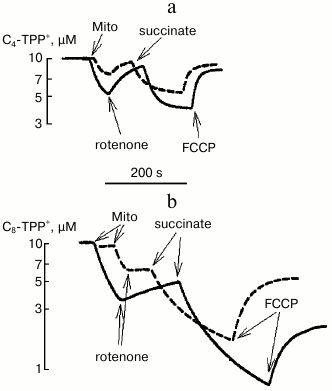
Using a TPP-selective electrode for measuring concentration of C4-TPP+ and C8-TPP+ in solution, it was possible to estimate their absorbance by energized and de-energized mitochondria depending on the presence of phosphate in the medium (Fig. 5). It was noted that both in the case of C4-TPP+ and of C8-TPP+ cations, protons were pumped in much better in the presence of phosphate. Also, much more C8-TPP+ was bound to de-energized and energized mitochondria. The phosphate-related effect may be because the rate of proton pump activity was mainly accounted by pH gradient across the mitochondrial membrane, which decreased much faster in the presence of phosphate.
Fig. 5. Redistribution of C4-TPP+ (a) and C8-TPP+ (b) between medium and mitochondria after energizing mitochondria by adding succinate (3 mM, in the presence of 2 µM rotenone) and de-energizing process by adding uncoupling agent FCCP (0.2 µM) depending on the presence of phosphate in the medium (2 mM KH2PO4, solid curve; no phosphate in the medium, dashed curve). Measurements were done using a TPP+-selective electrode. The incubation medium contained 250 mM sucrose, 10 mM MOPS, 3 mM MgCl2, 1 mM EGTA, pH 7.4. The concentration of mitochondrial (Mito) protein was 0.6 mg/ml.
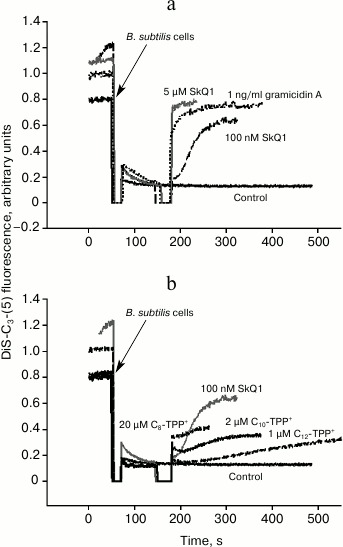
Uncoupling effect of alkyltriphenylphosphonium cations in Bacillus subtilis. To measure membrane potential on Bacillus subtilis cells, the potential-sensitive probe DiS-C3-(5) was applied. It is known that this cationic probe accumulates inside B. subtilis in the presence of membrane potential (negative inside), and its fluorescent signal fades due to its aggregation [18]. DiS-C3-(5)-mediated fluorescence in response to increasing concentrations of SkQ1 was recorded (Fig. 6a). At submicromolar concentration, SkQ1 caused a relatively slow decline in membrane potential in B. subtilis, whereas by adding 5-µM SkQ1 the potential dropped instantly (Fig. 6). The decrease in membrane potential in B. subtilis after adding C12-TPP+, C10-TPP+, and C8-TPP+ is plotted in Fig. 6b: the agents were added at concentration empirically shown to result in quite slow decrease in membrane potential. In the case of tetraphenylphosphonium cation and C4-TPP+, even the highest concentration (100 µM) did not result in decrease in membrane potential. For the other Cn-TPP+ ions, this activity was displayed at 1 µM (C12-TPP+), 2 µM (C10-TPP+), and 20 µM (C8-TPP+) concentrations. Thus, to significantly decrease potential in B. subtilis, a steadily decreasing concentration of cations is required with growing alkyl chain length in Cn-TPP+.
Fig. 6. Influence of SkQ1 (a) and Cn-TPP+ (b) on membrane potential in B. subtilis evaluated by DiS-C3-(5)-mediated fluorescence intensity. b) Representative kinetic curves of DiS-C3-(5)-induced fluorescence changes after adding Cn-TPP+ at concentrations resulting in “intermediate” effect on bacterial membrane potential, i.e. for each compound the lower concentrations (compare to the “intermediate” one) triggered weaker/no effect, whereas the higher concentrations resulted in rapid and complete loss of membrane potential. Gramicidin A effect resulting in complete loss of membrane potential is shown on panel (a). The assay medium contained 100 mM KCl, 10 mM Tris, pH 7.4. The bacterial cell concentration was 2·107/ml.
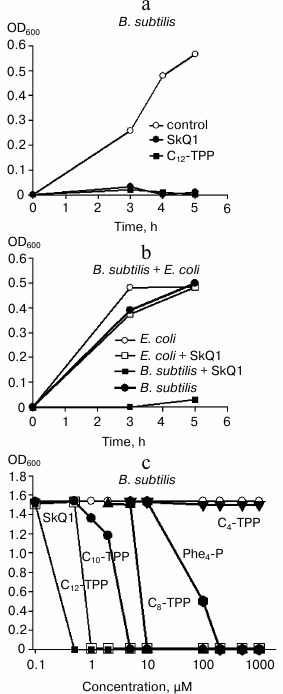
Membrane potential is required for bacteria to provide normal metabolism, and its inhibition should be toxic to bacteria. This assumption was tested by plotting growth curves for B. subtilis and E. coli in control setting and after adding permeating cations (Fig. 7). Indeed, SkQ1 and C12-TPP+ at micromolar concentration were shown to suppress growth of B. subtilis (Fig. 7a). However, they did not affect growth of E. coli (Fig. 7b). In addition, experiments evaluating the influence of SkQ1 on activity of both bacterial strains (expressed in colony-forming units, CFU) demonstrated that adding 1 µM SkQ1 decreased CFU value in B. subtilis by 10-fold, whereas it had virtually no effect on E. coli (data not shown). The dependence between growth of B. subtilis and concentration of SkQ1 and Cn-TPP+ compounds is shown in Fig. 7c. In particular, it was found that inhibitory concentrations of Cn-TPP+ gradually decreased with growing hydrocarbon chain length. For C4-TPP+, no toxicity was exhibited even at 1 mM concentration (Fig. 7c, triangles).
Fig. 7. a, b) Effects of SkQ1 and Cn-TPP+ on growth of B. subtilis and E. coli in LB medium evaluated by absorbance at 600 nm. The compounds were added at “0” time point at the concentration of 1 µM (a, b) or in the range of 0.1-1000 µM (c)). c) Dependence between absorbance at 600 nm after 5-h growth of suspended B. subtilis and concentration of SkQ1 and Cn-TPP+.
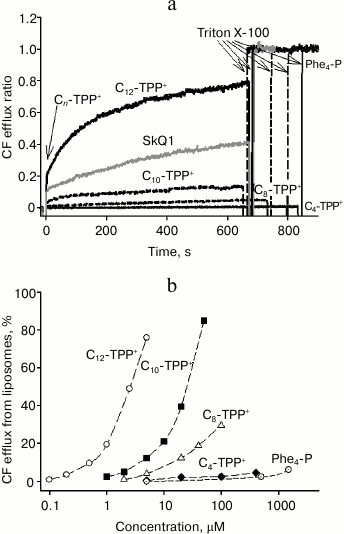
Effect of alkyltriphenylphosphonium cations on release of CF from liposomes. We demonstrated that with growing length of hydrocarbon chain, the effect of Cn-TPP+ ions on mitochondria and bacteria increased. This suggests that an uncoupling effect did not involve specific interaction with any enzymes. If so, then the uncoupling mechanism might primarily be related to binding to endogenous fatty acids, which has been described previously [5]. To confirm this mechanism, the effect of Cn-TPP+ on CF efflux from liposomes was examined (to note, CF is an organic anion like fatty acids) [15]. Liposomes were loaded with CF at fluorescence self-quenching concentration followed by estimating the increase in fluorescence as a measure of CF release. Earlier, we found that C12-TPP and SkQ1 at micromolar concentrations triggered CF efflux from liposomes [15]. In the present study, we found that C10-TPP+ induced CF efflux at concentrations approximately one order of magnitude higher than those of C12-TPP+, i.e. tens of micromoles (Fig. 8). SkQ1 was also active at somewhat higher concentrations compared to C12-TPP+. The efficacy of other Cn-TPP+ cations declined along with decreasing length of the hydrocarbon chain, but tetraphenylphosphonium cation was somehow more active than C4-TPP+. Earlier, it was shown that Cn-TPP+ (n = 1-6) trigger efflux of the anionic probe calcein from liposomes, the value of efflux enhancing with increase in hydrocarbon chain length [22].
Fig. 8. a) Time dependence of carboxyfluorescein (CF) efflux from liposomes triggered by 5 µM SkQ1 and Cn-TPP+. Curves were plotted as the ratio of fluorescence registered (at 520 nm) to the fluorescence level after adding Triton X-100, causing complete efflux of CF from the liposomes. The liposomes were prepared from total E. coli-derived lipids containing 100 mM CF, a self-quenching concentration. b) Dependence of CF efflux on Cn-TPP+ concentration 600 s after adding the permeating cation. The assay medium contained 100 mM KCl, 10 mM Tris, pH 7.4. The lipid concentration was 3 µg/ml.
DISCUSSION
During the first studies, it was already found that permeating cations are able to decrease mitochondrial potential and stimulate respiration due to their ability to pass across the membrane in a potential-dependent manner, which lowers mitochondrial potential followed by stimulated activity of proton pumps [23]. Most obviously, this effect was evident on adding 100 µM calcium ions to suspended energized mitochondria, which resulted in an initial decrease in membrane potential (and stimulation of respiration) followed by recovery of to the baseline level within about a minute (without induction of mitochondrial pores) [20]. It should be noted that the recovery time of the potential greatly depends on the presence of phosphate in the medium, sharply increasing without phosphate [20]. This is because the inner mitochondrial membrane contains a very active electroneutral phosphate transporter, which equalizes pH value via co-transporting protons with phosphate. Activation of proton pumps upon entry of permeating cation generates a pH gradient across the mitochondrial membrane (additional matrix alkalinization) and subsequently lowers the level of confining potential, as the activity of proton pumps depends on proton-motive force ΔµH+, being the sum of Δψ plus ΔpH. The activity of the phosphate transporter restores the matrix pH value to the baseline level, which contributes to the recovery of the initial magnitude of Δψ due to unchanged ΔµH+.
In contrast to the reversible decrease in mitochondrial membrane potential described above, high concentrations of permeating cations result in irreversible decrease in membrane potential and stimulation of respiration. In our study, we demonstrated that the uncoupling effect of some Cn-TPP+ in mitochondria steadily increases with increasing hydrocarbon chain length. The same effect was observed with liposomes, when the rate of efflux for carboxyfluorescein increased with increasing alkyl chain length in Cn-TPP+. Moreover, uncoupled respiration and decrease in membrane potential after adding Cn-TPP+ were sensitive to the presence of fatty acids and BSA in the medium. Altogether, this indicates that fatty acids are involved in the uncoupling effect exhibited by Cn-TPP+. Hence, the role for Cn-TPP+ cations involves formation of complexes with anions of endogenous fatty acids and stimulation of their transport across the mitochondrial membrane. Increased uncoupling effect observed along with increasing hydrocarbon chain length in Cn-TPP+ may be related to increased strength of the complex between the cation and fatty acid anions. However, a molecular dynamics study of this interaction indicated that a Cn-TPP+ cation and a fatty acid anion have low affinity for each other, and it is mainly accounted for by electrostatic factors [5]. In addition, it was also supposed that varying hydrocarbon chain length in Cn-TPP+ does not affect the constant of cation transmembrane diffusion, and membrane affinity increases with increasing hydrocarbon chain length [19]. Thus, the most likely mechanism underlying the increased efficacy of the uncoupling effect by Cn-TPP+ with increasing n value is merely related to increase in local concentration of Cn-TPP+ in the mitochondrial membrane and increased probability of interaction between Cn-TPP+ and an anion of highly lipophilic fatty acids.
Interestingly, the maximum respiration rate was found after adding C8-TPP+ (Fig. 2), and, although C10-TPP+ was active at lower concentration, the maximum respiration rate was lower. It might be assumed that C10-TPP+ and C12-TPP+ inhibit succinate dehydrogenase, revealed as a lower maximal respiration rate compared to C8-TPP+.
In addition, we were also able to demonstrate that SkQ1 and some Cn-TPP+ agents suppressed growth of B. subtilis, without affecting growth of E. coli. In the series from C4-TPP+ to C12-TPP+, the toxic effect steadily increased with increasing alkyl radical length. It should be noted that Cn-TPP+ agents and cationic surface-active agents (SAA) bear some structural similarity. It is known that SAAs exhibit antiseptic and antibacterial activity [24]. Among them, benzalkonium is a representative SAA, consisting of a mixture of alkyl benzyl dimethyl ammonium-based quaternary amines with alkyl substituent length varying from C8 to C18. By evaluating the effect of Cn-TPP+ on potential in B. subtilis, it was found that such agents effectively decreased potential, wherein the efficacy of the compounds was parallel with the degree of their toxicity. Thus, we conclude that cytotoxicity of Cn-TPP+ is due to the ability of Cn-TPP+ to dissipate membrane potential, thereby suppressing energy turnover in bacteria. Moreover, by analogy with mitochondria, this effect seems to be accounted for by participation of endogenous fatty acids.
We acknowledge Dr. E. A. Omarova for help and Dr. S. A. Novgorodov for fruitful discussion of results.
This study was done with financial support from the Institute of Mitoengineering, Moscow State University (project No. 14-24-00107), and the Russian Science Foundation (project No. 14-50-00029).
REFERENCES
1.Skulachev, V. P., Antonenko, Y. N., Cherepanov, D.
A., Chernyak, B. V., Izyumov, D. S., Khailova, L. S., Klishin, S. S.,
Korshunova, G. A., Lyamzaev, K. G., Pletjushkina, O. Y., Roginsky, V.
A., Rokitskaya, T. I., Severin, F. F., Severina, I. I., Simonyan, R.
A., Skulachev, M. V., Sumbatyan, N. V., Sukhanova, E. I., Tashlitsky,
V. N., Trendeleva, T. A., Vyssokikh, M. Y., and Zvyagilskaya, R. A.
(2010) Prevention of cardiolipin oxidation and fatty acid cycling as
two antioxidant mechanisms of cationic derivatives of plastoquinone
(SkQs), Biochim. Biophys. Acta, 1797, 878-889.
2.Murphy, M. P., and Smith, R. A. (2007) Targeting
antioxidants to mitochondria by conjugation to lipophilic cations,
Annu. Rev. Pharmacol. Toxicol., 47, 629-656.
3.Cunha, F. M., Caldeira da Silva, C. C., Cerqueira,
F. M., and Kowaltowski, A. J. (2011) Mild mitochondrial uncoupling as a
therapeutic strategy, Curr. Drug Targets, 12,
783-789.
4.Korshunov, S. S., Skulachev, V. P., and Starkov, A.
A. (1997) High protonic potential actuates a mechanism of production of
reactive oxygen species in mitochondria, FEBS Lett., 416,
15-18.
5.Severin, F. F., Severina, I. I., Antonenko, Y. N.,
Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M. Y.,
Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova, G.
A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
Penetrating cation/fatty acid anion pair as a mitochondria-targeted
protonophore, Proc. Natl. Acad. Sci. USA, 107,
663-668.
6.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E.,
Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Y.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Y., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vyssokikh, M.
Y., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Mitochondria-targeted plastoquinone derivatives as tools to
interrupt execution of the aging program. 1. Cationic plastoquinone
derivatives: synthesis and in vitro studies, Biochemistry
(Moscow), 73, 1273-1287.
7.Khailova, L. S., Silachev, D. N., Rokitskaya, T.
I., Avetisyan, A. V., Lyamsaev, K. G., Severina, I. I.,
Il’yasova, T. M., Gulyaev, M. V., Dedukhova, V. I., Trendeleva,
T. A., Plotnikov, E. Y., Zvyagilskaya, R. A., Chernyak, B. V., Zorov,
D. B., Antonenko, Y. N., and Skulachev, V. P. (2014) A short-chain
alkyl derivative of rhodamine 19 acts as a mild uncoupler of
mitochondria and a neuroprotector, Biochim. Biophys. Acta,
1837, 1739-1747.
8.Sassi, N., Mattarei, A., Azzolini, M.,
Szabo’, I., Paradisi, C., Zoratti, M., and Biasutto, L. (2014)
Cytotoxicity of mitochondria-targeted resveratrol derivatives:
interactions with respiratory chain complexes and ATP synthase,
Biochim. Biophys. Acta, 1837, 1781-1789.
9.Severina, I. I., Muntyan, M. S., Lewis, K., and
Skulachev, V. P. (2001) Transfer of cationic antibacterial agents
berberine, palmatine, and benzalkonium through bimolecular planar
phospholipid film and Staphylococcus aureus membrane, IUBMB
Life, 52, 321-324.
10.Schmeller, T., Latz-Bruning, B., and Wink, M.
(1997) Biochemical activities of berberine, palmatine, and sanguinarine
mediating chemical defence against microorganisms and herbivores,
Phytochemistry, 44, 257-266.
11.Galkina, I. V., and Egorova, S. N. (2009)
Biological activity of quaternary salts of phosphonium and perspectives
of their medical application, Farmatsiya, 9, 142-145.
12.Galkina, I. V., Bakhtiyarova, Y. V., Shulaeva, M.
P., Pozdeev, O. K., Egorova, S. N., Cherkasov, R. A., and Galkin, V. I.
(2013) Synthesis and antimicrobial activity of carboxylate
phosphabetaines derivatives with alkyl chains of various lengths, J.
Chem., doi: 10.1155/2013/302937.
13.Kanazawa, A., Ikeda, T., and Endo, T. (1994)
Synthesis and antimicrobial activity of dimethyl-substituted and
trimethyl-substituted phosphonium salts with alkyl chains of various
lengths, Antimicrob. Agents Chemother., 38, 945-952.
14.Ross, M. F., Prime, T. A., Abakumova, I., James,
A. M., Porteous, C. M., Smith, R. A., and Murphy, M. P. (2008) Rapid
and extensive uptake and activation of hydrophobic triphenylphosphonium
cations within cells, Biochem. J., 411, 633-645.
15.Rokitskaya, T. I., Sumbatyan, N. V., Tashlitsky,
V. N., Korshunova, G. A., Antonenko, Y. N., and Skulachev, V. P. (2010)
Mitochondria-targeted penetrating cations as carriers of hydrophobic
anions through lipid membranes, Biochim. Biophys. Acta,
1798, 1698-1706.
16.Johnson, D., and Lardy, H. (1967) Isolation of
liver or kidney mitochondria, Methods Enzymol.,
10, 94-96.
17.Akerman, K. E., and Wikstrom, M. K. (1976)
Safranine as a probe of the mitochondrial membrane potential, FEBS
Lett., 68, 191-197.
18.Miller, J. B., and Koshland, D. E., Jr. (1977)
Sensory electrophysiology of bacteria: relationship of the membrane
potential to motility and chemotaxis in Bacillus subtilis,
Proc. Natl. Acad. Sci. USA, 74, 4752-4756.
19.Trendeleva, T. A., Rogov, A. G., Cherepanov, D.
A., Sukhanova, E. I., Il’yasova, T. M., Severina, I. I., and
Zvyagilskaya, R. A. (2012) Interaction of tetraphenylphosphonium and
dodecyltriphenylphosphonium with lipid membranes and mitochondria,
Biochemistry (Moscow), 77, 1021-1028.
20.Lotscher, H. R., Winterhalter, K. H., Carafoli,
E., and Richter, C. (1980) The energy state of mitochondria during the
transport of Ca2+, Eur. J. Biochem., 110,
211-216.
21.Kramer, R., and Palmieri, F. (1989) Molecular
aspects of isolated and reconstituted carrier proteins from animal
mitochondria, Biochim. Biophys. Acta, 974, 1-23.
22.Yang, Q., Liu, X. Y., Umetani, K., Kamo, N., and
Miyake, J. (1999) Partitioning of triphenylalkylphosphonium homologues
in gel bead-immobilized liposomes: chromatographic measurement of their
membrane partition coefficients, Biochim. Biophys. Acta,
1417, 122-130.
23.Bakeeva, L. E., Grinius, L. L., Jasaitis, A. A.,
Kuliene, V. V., Levitsky, D. O., Liberman, E. A., Severina, I. I., and
Skulachev, V. P. (1970) Conversion of biomembrane-produced energy into
electric form. II. Intact mitochondria, Biochim. Biophys Acta,
216, 13-21.
24.Thorsteinsson, T., Loftsson, T., and Masson, M.
(2003) Soft antibacterial agents, Curr. Med. Chem., 10,
1129-1136.