Effect of Naphthalene on Photosystem 2 Photochemical Activity of Pea Plants
A. V. Lankin1, V. D. Kreslavski1,2*, A. Yu. Khudyakova3, S. K. Zharmukhamedov2*, and S. I. Allakhverdiev1,2*
1Timiryazev Institute of Plant Physiology, Russian Academy of Sciences, ul. Botanicheskaya 35, 127276 Moscow, Russia; fax: (499) 977-80182Institute of Basic Biological Problems, Russian Academy of Sciences, ul. Institutskaya 2, 142290 Pushchino, Moscow Region, Russia; fax: (4967) 330-532; E-mail: vkreslav@rambler.ru; watcher01@rambler.ru; allakhverdiev@gmail.com
3Institute of Biological Engineering, Russian Academy of Sciences, ul. Institutskaya 7, 142290 Pushchino, Moscow Region, Russia; fax: (4967) 330-522
* To whom correspondence should be addressed.
Received June 20, 2014; Revision received July 29, 2014
The effect of a typical polyaromatic hydrocarbon, naphthalene (Naph), on photosystem 2 (PS-2) photochemical activity in thylakoid membrane preparations and 20-day-old pea leaves was studied. Samples were incubated in water in the presence of Naph (0.078, 0.21, and 0.78 mM) for 0.5-24 h under white light illumination (15 μmol photons·m–2·s–1). The PS-2 activity was determined by studying fast and delayed chlorophyll (Chl) a fluorescence. Incubation of samples in water solutions at Naph concentrations of 0.21 and 0.78 mM led to a decrease in the maximum PS-2 quantum efficiency (Fv/Fm), noticeable changes in the polyphasic induction kinetics of fluorescence (OJIP), and a decrease in the amplitudes of the fast and slow components of delayed fluorescence of Chl a. The rate of release of electrolytes from leaves that were preliminarily incubated with Naph (0.21 mM) was also increased. Significant decrease in the fluorescence parameters in thylakoid membrane preparations was observed at Naph concentration of 0.03 mM and 12-min exposure of the samples. Chlorophyll (a and b) and carotenoid content (mg per gram wet mass) was insignificantly changed. The quantum yields of electron transfer from QA to QB (φET2o) and also to the PS-1 acceptors (φRE1o) were reduced. These results are explained by the increase in the number of QB-non-reducing centers of PS-2, which increased with increasing Naph concentration and exposure time of leaves in Naph solution. The suppression of PS-2 activity was partly abolished in the presence of the electron donor sodium ascorbate. Based on these results, it is suggested that Naph distorts cell membrane intactness and acts mainly on the PS-2 acceptor and to a lesser degree on the PS-2 donor side.
KEY WORDS: naphthalene, photosystem 2, variable and delayed fluorescence of chlorophyll, detached pea leaves, thylakoid membranes, photosynthetic pigmentsDOI: 10.1134/S0006297914110091
Abbreviations: Asc, sodium ascorbate; Chl, chlorophyll; DFl, delayed fluorescence; Fo, minimum fluorescence; Fm, maximum fluorescence; Fv, variable fluorescence (Fv = Fo – Fm); FFl, fast fluorescence; Naph, naphthalene; PA, photosynthetic apparatus; PAHs, polycyclic aromatic hydrocarbons; PS-2, photosystem 2; QA, primary quinone acceptor of PS-2; QB, secondary quinone acceptor of PS-2; RC, reaction center; TM, thylakoid membranes.
Great importance is now given to the study of environmental
contamination by different pollutants. Some of the most common and
toxic pollutants are polycyclic aromatic hydrocarbons (PAHs), i.e.
comprising at least two benzene rings. PAHs with two or three rings are
present in the atmosphere, mainly in the gas phase, whereas those with
four rings or more predominate in the solid state [1].
PAHs in atmosphere, such as naphthalene (two benzene rings) and phenanthrene (three benzene rings), penetrate into the intercellular spaces of plants leaves, mainly through the stomata. On one hand, due to the lipophilicity of the molecule, PAHs may accumulate in the lipid layer of the plants membranes, mainly from precipitation from the atmosphere onto the leaf surface [2]. On the other hand, in the form of water-soluble derivatives these agents reach the cell membrane, break its intactness, and increase permeability. This may be related to the change in the ratio of H+ and Ca2+ in the membranes and to decrease in the number of sulfhydryl groups (SH) and a corresponding increase in membrane protein disulfide bonds, and also with the formation of membrane lipid defect regions due to the accumulation of products of lipid peroxidation [3]. It is assumed that the effect of extreme factors such as PAHs leads to disruption of membrane functions and structures. Thus, there is a disruption of the functioning of the photosynthetic apparatus (PA), primarily photosystem 2 (PS-2), which is most vulnerable to the effects of stress factors and can therefore serve as a bio-indicator of PAH action [4, 5].
Effects of PAHs depend on the duration of exposure and the physicochemical properties of the pollutant molecule [4-6]. Redox properties and lipophilicity of the agent, characterized by distribution coefficient in octanol/water, determine the efficiency of penetration of PAHs to molecular targets.
Approaches for assessing the photochemical activity of PS-2 include measurement of “variable” fast fluorescence (FFl) (OJIP-transitions) and delayed fluorescence (DFl) of chlorophyll (Chl) a [7-9]. In this case, based on the kinetics of induction curves of millisecond DFl and fast fluorescence, different parameters characterizing the photochemical activity of PS-2 can be calculated. FFl measurements allow evaluation of the maximum quantum efficiency of PS-2 photochemistry, the efficiency of electron transfer at different sites of the electron transport chain (ETC), and a number of other parameters [8, 10, 11]. Based on the kinetics of DFl, the value of light-induced formation of the transmembrane proton gradient (ΔpH) through the thylakoid membranes and the rate of electron transfer on the PS-2 acceptor side can be estimated [7].
The solubility of almost all PAHs in water is low. Among PAHs, naphthalene (Naph) has the highest solubility in water, which at 0 and 25°C is 0.148 and 0.268 mM, respectively [12]. It is assumed that Naph, as a typical representative of PAHs, due to its low solubility in water acts primarily on lipid membranes of cells. When Naph-like PAHs damage the membrane, this results in the development of oxidative stress. This leads to a decrease in PS-2 activity and degradation of some its components. It was shown that Naph causes a decrease in PS-2 activity more rapidly than phenanthrene and fluoranthene [13]. However, the mechanisms of PS-2 activity suppression under the action of Naph still unclear in many details. The targets of Naph in PS-2 and the fast reactions of PA, when formation of metabolites of Naph is unlikely, are insufficiently studied. It should be noted that experiments with PAHs in many studies take a long time (2-6 weeks) [4, 6], which complicates the understanding of the mechanisms of their effects on the activity of the PA.
In this work we studied the effect of different concentrations of Naph on the photochemical activity of PS-2 and the content of photosynthetic pigments, as well as on the rate of Naph absorption by leaves and the electrolytes’ output after a short incubation of leaves in a solution of Naph.
The data suggest that Naph damages intactness of cell membranes and acts primarily on the acceptor and to a lesser extent on the donor side of PS-2.
MATERIALS AND METHODS
Conditions for plant growth. Experiments were conducted on detached pea leaves (Pisum sativum L. cv. Moskovskii 559). Three-day-old seedlings were placed in plastic containers (25 × 25 × 14 cm, volume 8.75 liters) filled with moist sand, in which every seven days Knop medium (one fourth of total volume) have was added. Plants were grown for 20 days under white light illumination with intensity of 360 µmol photons·m–2·s–1 (from lamps HPS-400) and at temperature of 25 ± 1°C.
The leaves of the second and third tiers were detached and placed in Petri dishes with moist filter paper for 1 h to adapt to the white light fluorescent lamps (15 µmol photons·m–2·s–1) before the measurement. Then the leaves (6-7 pieces) were placed in sealed glass jars (100 ml) containing 80 ml of Naph solution with determined concentration and incubated under the same lighting conditions at 25 ± 1°C. After incubation of the leaves in the naphthalene solution for different time periods (0.5-24 h), all necessary investigations were carried out. Preparations of thylakoid membranes (TM) from peas (35 mg Chl/ml) were prepared as described previously [13] and incubated with Naph (0.03 and 0.08 mM) for 12 min.
Preparation of naphthalene samples. Since Naph has low solubility in water, a 1000-fold more concentrated solution of Naph in acetone (Sigma-Aldrich, USA) was first prepared. Then, the acetone solution was poured into distilled water to obtain final concentrations of 0.078 and 0.21 mM. The volume of 0.1 ml of stock solution was added to 100 ml of distilled water. In some experiments supersaturated solutions of Naph (Naph nominal concentration is 0.78 mM) were used in which some Naph was present in solution as a stable suspension. As a control, distilled water with the indicated concentration of acetone (0.1%) was used.
Measurements of chlorophyll fluorescence. The photosynthetic activity of PS-2 was assessed by measuring the fast and delayed fluorescence (FFl and DFl, respectively) of Chl a after incubation of leaves in a solution of Naph in the light for 1, 2, and 24 h. Prior to the measurements the leaves were fixed in the measuring cell and incubated in the dark for 15 min.
The Fo level and kinetics of photoinduced changes in the PS-2 chlorophyll fluorescence yield were measured with a PAM-fluorimeter (XE-PAM; Heinz Walz, Germany). Induction curves of the FFl and OJIP-transitions (increase in fluorescence yield of Chl a from minimum to maximum intensity – OJIP) were recorded with a fluorimeter as described previously [13]. The data is stored and processed with a computer.
Based on the Chl a FFl induction curves, the following fluorescence parameters were calculated: Fo, Fm, Fv, where Fo – the minimum level of fluorescence (F), Fm – maximum of F, and Fv – variable F defined as the difference between Fo and Fm [8]. Furthermore, based on the data the following parameters were calculated: ET2o/RC, ET2o/ABS, Ψo, TRo/RC, and ABS/RC (Table 1), where TR is the rate of exciton flow trapped by all the reaction centers (RC) of PS-2; TRo is the maximum (initial) exciton flow captured by all PS-2 RCs. The ratio TRo/RC designates the exciton capture rate (leading to the reduction of QA), i.e. the rate of QA reduction to QA– as a result of the absorption of an exciton in the PS-2 RC per RC; ABS/RC is the averaged flux of photons absorbed by PS-2 antenna chlorophyll per RC, the value of ABS/RC characterizes the size of the PS-2 antenna; ET2o is the maximum flow of electrons transferred in PS-2 from QA to QB; ratio ET2o/RC is the flow of electrons transferred from QA to QB per RC [8].
Table 1. Parameters (OJIP-parameters)
calculated based on induction curves of fast chlorophyll
fluorescence

Calculations were carried out in accordance with the following formulas:
ABS/RC = (Mo/VJ)·(Fv/Fm),
ETo/RC = (Mo/VJ)·(1 – VJ),
TRo/RC = Mo/VJ,
ETo/ABS = Fv/(Fm·(1 – VJ)),
Mo = 4 ms–1·(F300µs – Fo)/(Fm – Fo),
where VJ is relative fluorescence level in phase J; VJ = (F2ms – Fo)/(Fm – Fo); ET2o/TRo = Ψo = 1 – VJ. Here ABS is photon flux absorbed by chlorophyll of PS-2 antenna; J is the energy flux; 1 and 2 designate PS-1 and PS-2, respectively, as previously described [8].
Measurements DFl. Millisecond DFl of Chl a was measured using a disk-based phosphoroscope setup described in detail previously [14], with some modifications. Samples were irradiated with light of wavelength 645 nm and intensity at the surface equal to 1800 µmole quanta·m–2·s–1 from the red LED. The duration of illumination, the dark period, and the registration was 2, 4, and 2 ms, respectively. The photomultiplier signal was converted in a PCIe-6321 ADC (L-Card) and recorded every 50 µs during the registration period. The data was stored in the computer for further processing and analysis using functions of averaging and smoothing.
Contents of Chl a and b as well as carotenoids (mg per gram wet weight) was measured in extracts of ethanol using known absorption coefficients [15].
Naphthalene accumulation in the leaves. The rate of Naph accumulation in the pea leaves was evaluated on the basis of lowering Naph initial concentration in the aqueous solutions after incubation of 6-7 leaves in a solution of naphthalene (80 ml) for 24 h. Decrease in Naph initial concentrations (0.078 and 0.21 mM) was determined with a Spectronic Genesis 10UV spectrophotometer (Thermo Scientific, USA) in the main Naph absorption wavelength at 219 nm. As a control (taken as 100%), data on changes in the concentration of similar Naph solutions in the absence of leaves were used.
Rate of release of electrolytes. The cell membrane permeability was judged by the rate of electrolyte exit (REE) from detached leaves in an aqueous solution where they were placed. The conductivity of the aqueous solution was measured by conductometry. The units of measure was microSiemens per gram fresh weight (µSm/g). REE was measured as follows. Detached pea leaves (6-7 leaves) were incubated in bidistilled water and a solution of Naph (0.078 and 0.21 mM) (volumes of solutions were 80 ml) for 2 h at light intensity of 12 W/m2. After washing with distilled water, the leaves were placed in 80 ml bidistilled water and incubated for 3 h at room temperature. The conductivity was measured immediately after placing the leaves in bidistilled water and during incubation of the leaves for 3 h.
To clarify the nature of the DFl components and the effect of Naph on PS-2, experiments were conducted in the presence of (i) the inhibitor of PS-2 Diuron (3-(3,4-dichlorophenyl)-1,1-dimethylurea) (blocking electron transfer between the primary and secondary quinone electron acceptors PS-2, QA and QB, respectively); (ii) the PS-2 exogenous electron donor sodium ascorbate. Leaves were incubated in a solution of sodium ascorbate (3 mM) or Diuron (30-50 µM) for 30 min.
In the tables and figures, the arithmetic means of values and their standard errors (±S.E.) are presented. At least three biological and at least 4-9 analytical replicates were used. The reliability of differences between the variants was determined by Student’s t-test at significance level p < 0.05.
RESULTS
Effects of Naph on photochemical activity of PS-2 in pea leaves as well as on the activity of the PS-2 preparations of thylakoid membranes were investigated both after short-term (1 h) incubation in the presence of Naph (Table 2) and in after long-term (24 h) experiments (Figs. 1 and 2 and Tables 3 and 4).
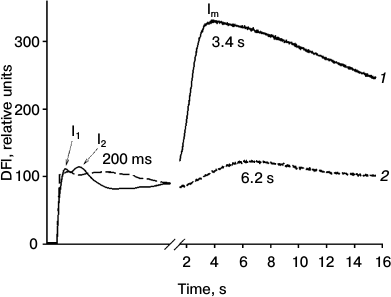
Fig. 1. Effect of Naph on DFl curves of detached pea leaves. Leaves were incubated in aqueous Naph solution (0.78 mM) for 24 h (2). Control leaves were incubated without Naph for 24 h (1). Maxima in the figure labeled as I1, I2, and Im. Typical curves of nine replicates are shown.
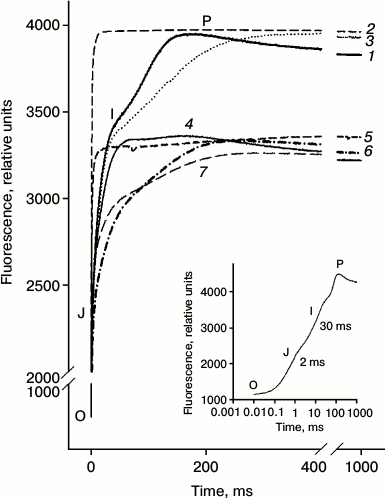
Fig. 2. Kinetics of chlorophyll FFl (OJIP-transitions) measured on detached leaves. Leaves were incubated with distilled water (1) for 24 h; then a portion of the leaves was incubated for 60 min in a solution of Diuron (2) or 30 min in a solution of sodium ascorbate (Asc) (3). Another portion of the leaves was incubated in a Naph solution (4) for 24 h. Then part of the leaves were incubated for 1 h in a solution of Diuron (5) or 30 min in a solution of Asc (6) as well as in the mixed solution Diuron (1 h) + Asc (30 min) (7). Concentrations of Naph, Diuron, and Asc were 0.21 mM, 40 µM, and 3 mM, respectively. In the lower right corner a typical OJIP curve measured on the untreated leaves is shown.
Table 2. Effect of naphthalene (Naph) on the
ratio (Fv/Fm) and the relative amplitude of DFl
slow component ((Im – D)/D), as well as absorption
Naph by leaves incubated for 2 h in a solution at different Naph
concentrations under light intensity of 12 W/m2. Naph
absorption in solution without leaves was taken as 100%. Values in
parentheses are standard errors
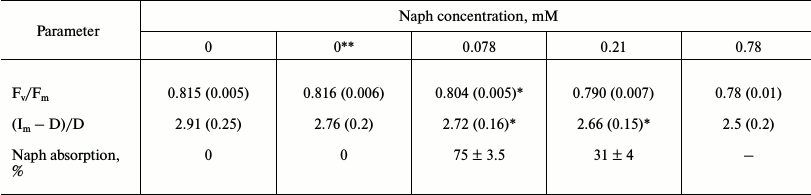
Note: Relationship Fv/Fm are amended with inserted
acetone – 0.1 and 1%, respectively, n = 8.
* Difference is insignificant (p > 0.05).
** Acetone is added: 0.1% in the measurements of variable fluorescence
and 1% – for DFl.
Table 3. Effect of naphthalene (Naph),
Diuron, and sodium ascorbate (Asc) on the fluorescence parameters
calculated based on induction curves of chlorophyll FFl,
OJIP-transitions. Values in parentheses are standard errors
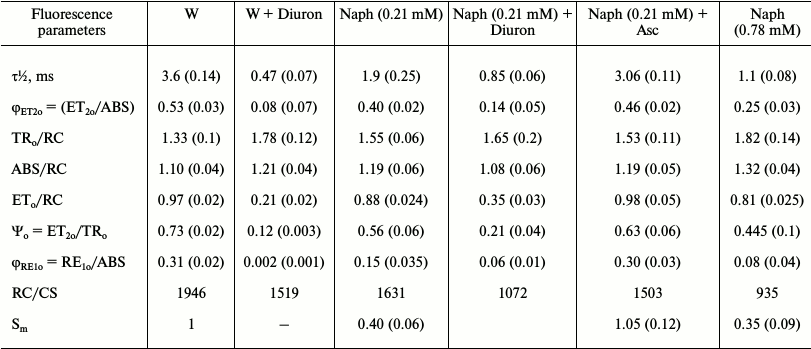
Note: The value of Sm in the control is taken as unity. The
leaves were incubated in the light (15 µmol
photons·m–2·s–1) in a
Naph solution of two concentrations (0.21 and 0.78 mM) or
distilled water (W) for 24 h. Then the leaves were kept in Diuron
solution (30 µM), Asc (3 mM) for 30 min (variants:
W + Diuron, Naph + Diuron, and Naph + Asc),
n = 5.
Table 4. Effect of naphthalene (Naph), and
sodium ascorbate (Asc) on the relative values of the fluorescent
parameters (Fo, Fm and
Fv/Fm), and the content of photosynthetic
pigments. Values in parentheses are standard errors
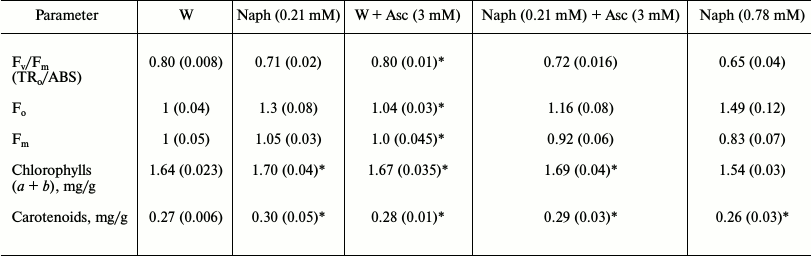
Note: The values of Fo and Fm in the control taken
as unity. Leaves were exposed to light (15 µmol
photons·m–2·s–1) in a
Naph solution of two concentrations (0.21 and 0.78 mM) or
distilled water (control, W) for 24 h. Then part of the leaves were
kept in Asc solution (3 mM) for 30 min (variants: W +
Asc and Naph + Asc), n = 8.
* Difference is insignificant (p > 0.05).
Using the DFl method, typical DFl curves were recorded including fast components with maxima I1 and I2 and the slow component with maximum Im (Fig. 1). In control leaves the positions of maxima corresponded to the following time values, I1 – 35 (±5) ms, I2 – 100 (±15) ms, and Im – 3.4 (±1) s. The relative amplitudes of the fast (I2) and the slow component were 0.3 and 3.0 relative units, respectively. After incubation of leaves in the presence of Naph for 24 h, the relative amplitudes of the fast and slow components decreased from 0.3 to 0.1 and from 3.0 to 0.8 relative units, respectively, and their maxima were at 200 ms and 6.2 s, respectively. In preparations of TM, only the fast component (maximum at 80 ms) was detected, which was reduced by half when incubated in 0.03 mM Naph and almost to zero in 0.08 mM Naph (data not shown).
Figure 2 (curve 1) shows a typical growth curve of FFl obtained in the absence of Naph (control), the so-called OJIP transitions – a rapid increase in the Chl a fluorescence from baseline (O) in a few milliseconds to a level J, and then relatively slow phases from J to I and from I to P, as shown previously [10, 11]. After incubation of the leaves in aqueous Naph solution for 24 h, the OJIP kinetics significantly changed (see Fig. 2, curve 2). The OJ phase is characterized with the least change, whereas the amplitudes of the JI and IP phases significantly decreased and their maxima shifted to longer times.
The effects of Naph on time of the photoinduced increase in the yield of PS-2 chlorophyll fluorescence from the initial (Fo) to maximum (Fm) levels (the rate of increase ΔF) were investigated. In control samples after incubation of leaves for 24 h in an acetone solution (0.1%) (which is used as a solvent for Naph), time (τ½) of increase of the F yield to a level equal to 50% from the maximum level (Fm(½)) was 3.6 ms. In leaves incubated for 24 h in the presence of Naph, this time was reduced to 1.90 ms (0.21 mM) and 1.1 ms (0.78 mM).
Values of the maximum quantum efficiency of PS-2 photochemistry (the ratio (Fm – Fo)/Fm) were calculated based on induction curves. This value for the leaves before Naph treatments was 0.80 ± 0.01. After incubation of leaves for 24 h in water containing acetone (0.1%), in the absence of Naph (control), this value was reduced to 0.78 ± 0.01. Only at Naph concentrations of 0.21 and 0.78 mM, and only after 2 h of incubation, significant reduction in quantum efficiency Fv/Fm was observed, while after 1 h of incubation in an aqueous solution of 0.21 mM Naph no significant effect was observed. At the same time, the contribution to this ratio of each of the variables, Fm and Fo, was assessed. The level Fo mainly increased, whereas the decrease in the maximum intensity Fm was less significant (Table 3). In TM preparations, a similar trend was observed: at 0.03 mM Naph concentration Fo is increased by 30% and Fm was little changed.
In some experiments Diuron, an inhibitor of electron transport between the primary quinone QA and secondary QB electron acceptors of PS-2, and sodium ascorbate, an exogenous electron donor to PS-2, were used. Diuron significantly (see Fig. 2, curves 2 and 5) and, to a lesser extent, sodium ascorbate (curve 3) changed the appearance of the induction curve of FFl, reducing the time to achieve level Fm(½). In the presence of Diuron, growth of FFl from Fo to Fm markedly accelerated. In control leaves the increase in the quantum yield of FFl to a level Fm(½) occurred in 3.6 ms, whereas in leaves treated with Diuron it occurred in only 0.47 ms. Adding the electron donor sodium ascorbate had not so much influence on the time of half-growth of FFl (τ½). After 30 min of incubation in a solution of sodium ascorbate, for leaves preincubated for 24 h in the presence of Naph (0.21 mM) the τ½ value is increased from 1.9 to 3.06 ms. Incubation of leaves in the presence of sodium ascorbate in the absence of Naph during the same time had practically no effect on the fluorescence parameters (Table 3). The τ½ magnitude changes only slightly, from 3.6 to 4.2 ms.
Based on the FFl induction curves, other parameters characterizing the PS-2 photochemical activity were also calculated (Table 1) including (i) flow of electrons transferred from QA to QB per PS-2 RC (value ET2o/RC), (ii) maximum quantum yields of electron transport at the site QA–QB (φET2o, ET2o/ABS) and from QA to PS-1 electron acceptors (φRE1o), and (iii) ΨET2o – the efficiency with which the capture of exciton by components of the PS-2 reaction center leads to electron transfer (ETo) from QA to QB (Table 2) [8]. All these values were decreased after incubation of leaves in Naph solution for 24 h and more so with increase in Naph concentration. The quantum yield φRE1o was most notably decreased – 4-fold at a Naph concentration of 0.78 mM and 2-3-fold at a Naph concentration of 0.21 mM. The area (Sm) over the OJIP curve characterizing the size of the plastoquinone molecule pool in PS-2 was decreased by 2.5-3.0-fold. The ratio of the number of active RCs to PS-2 absorption cross-section (RC/CS) was also decreased. The following values were also calculated: TRo/RC – the exciton capture rate, which reflects the rate of QA to QA– reduction because of exciton absorption in PS-2 RC per RC; ABS/RC – the rate of photon absorption by PS-2 antenna chlorophyll per RC. Both values were increased under (i) incubation of leaves in 0.21 mM Naph and (ii) increase in Naph concentration to 0.78 mM. In the presence of Diuron, changes in the values of TR0/RC and ABS/RC were similar to the changes observed in the presence of Naph (Table 2). An electron donor to PS-2, on the contrary, led to partial withdrawal of the effects on the parameters of the FFl of Naph treated leaves (Tables 3 and 4 and Fig. 3).
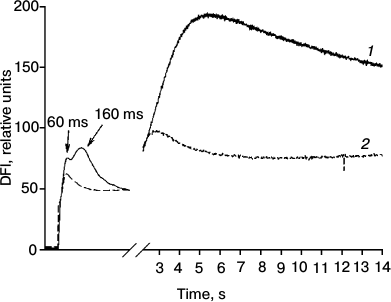
Fig. 3. Effect of Diuron on DFl curves of detached pea leaves. The leaves were incubated in a solution of Diuron (40 µM) for 30 min (2). Control leaves were incubated in distilled water with no Diuron for 30 min (1). Typical curves are shown.
The maximum quantum efficiency of PS-2 (Fv/Fm) after 24 h of incubation of untreated leaves in darkness was 0.78-0.81, which corresponds to the values of Fv/Fm characteristic of a healthy plant. After incubation of leaves with Naph (0.21 mM) the value of Fv/Fm decreased, starting with a 2-h incubation (Table 2) at the expense of increasing Fo level and decreasing Fm level. Increasing the Naph concentration resulted in further reduction in the quantum efficiency of PS-2. The Fo level was increased with increasing Naph concentration and incubation time (Table 4). The maximum fluorescence, Fm, varied little at 0.21 mM concentration and was reduced at 0.78 mM concentration.
Total Chl content (a + b) after detached leaves were incubated in aqueous solution of 0.78 mM Naph was reduced slightly, from 1.64 to 1.54 mg/g, whereas at 0.21 mM Naph concentration it was practically unchanged (Table 4).
We also investigated the effect of Diuron on DFl of pea leaves. In the presence of Diuron the amplitudes of the fast and slow components of the DFl induction curves were decreased. The fastest DFl component ((I1 – D)/D) with maximum I1 characterized by time 50 ms was the least susceptible to change compared with the slower components (150 ms and 4 s) with maxima I2 and Im (Fig. 3).
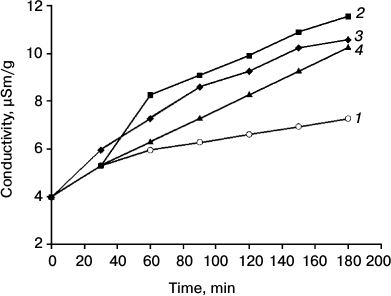
Study of the conductivity of the aqueous solution in the presence of leaves depending on the time of incubation in aqueous naphthalene solution (0.21 mM) revealed that Naph increased conductivity, and this effect was observed when the leaves were preincubated for 15 min with Naph (Fig. 4). Significant reduction in the optical density of the Naph solution (0.21 mM) in the presence of leaves was also observed after 15-20 min, whereas a 50% reduction was observed after 5 h.
Fig. 4. Dynamics of conductivity of an aqueous solution in the presence of pea leaves depending on the time of preincubation in 0.21 mM naphthalene. Duration of incubation (min): 1) 0; 2) 15; 3) 30; 4) 60. Time on abscissa – the time of registration of electrical conductivity after inserting in bidistilled water of leaves incubated (2-4) and not incubated (1) in Naph solution. In the absence of the leaf conductivity varied in the range of 0.1. The standard error of the measurements did not exceed 5%.
DISCUSSION
Typically, levels that characterize the maximum quantum efficiency of PS-2 (Fv/Fm) are closely correlated with the rate of photosynthesis in intact leaves. For healthy leaves the ratio Fv/Fm usually ranges from 0.75 to 0.84 [16]. This ratio for the leaves used by us for the treatments was 0.80 ± 0.01. After 24-h incubation of leaves in water containing acetone (0.1%) in the absence of Naph (control), it was reduced slightly to 0.785 ± 0.015, while after 2-h incubation it was practically unchanged, indicating the stability of the functional activity of PA in the studied leaves. Incubation of leaves in 0.21 mM Naph resulted in a decrease in this ratio after 2 h, which is in agreement with the earlier observed increase in hydrogen peroxide content in leaves after 30-min incubation at a given Naph concentration [13]. Comparing these data, one can suggest that oxidative stress precedes the reduction of the photochemical activity of PS-2. This supposition is supported by data that phenanthrene, a PAH containing three rings, decreases activity of PS-2 in Arabidopsis leaves, and this process is accompanied by accumulation of hydrogen peroxide [17].
PS-2 is heterogeneous in structure and in function [18, 19]. Two types of PS-2 heterogeneity are known: heterogeneity of antenna and heterogeneity of the reducing (acceptor) side [19, 20].
To investigate the possible mechanism of action of naphthalene on PS-2 activity, we used an inhibitor of electron transport on the acceptor side of PS-2, Diuron, and exogenous electron donor, sodium ascorbate. Diuron displaces the secondary quinone acceptor QB from its binding site on the D1-protein of PS-2 [21] and thereby blocks the electron transfer from QA to QB. Inhibition of reduced QA reoxidation is accompanied by characteristic changes in the kinetics of induction curves (OJIP) (Fig. 2). In this case, in the curves practically only one phase, J, is observed. This phase corresponds to the reduction of the primary electron acceptor QA. The next phases after the J phase of the induction curve are mainly related to the reduction of plastoquinone at the QB-site and the accumulation of reduced plastoquinone in the membrane pool of plastoquinone.
With regard to sodium ascorbate, its concentration in chloroplasts is rather high, perhaps reaching 20 mM [22]. However, as a result of treatment with PAH, which leads to oxidative stress due to increased membrane permeability, the content of sodium ascorbate in chloroplasts can be remarkably reduced, and even its relatively low concentration (3 mM) can act on the activity of PS-2.
Based on the analysis of the kinetics of FFl induction curves, we suggest that Naph affects primarily the acceptor side of PS-2, presumably through decrease in efficiency of electron transfer from QA to QB and further to the ETC, but not by reducing the efficiency of electron transfer to QA, which was not reduced during treatment. This suggestion is supported by all the results obtained on the leaves after incubation in the presence of Naph, namely data on the reduction of the area over the induction curve of FFl, which characterizes the size of the pool of active plastoquinone molecules; decrease in values of τ½; and that, after processing, the fast component (OJ) associated with the reduction of QA is virtually unchanged, whereas the amplitude of the slower component is reduced.
To take into account the reduced amplitude of the DFl fast component in TM preparations, we suggest that the effect of Naph on thylakoid membranes leads to an increase in the proportion of non-reduced QB centers of PS-2, i.e. it changes the heterogeneity of the acceptor side of PS-2. This suggestion was made previously [19] concerning effects of fluoranthene on peas leaves and some actions of PAHs on leaves of Arabidopsis plants [20]. The conclusion that the inhibition of electron transfer on the acceptor side is also consistent with data on the effect of Naph on the parameters calculated from the OJIP kinetics in the variant with simultaneous action on the PA of leaves of two agents, Diuron and Naph. Typical effects of Diuron in leaves treated with Naph, compared with untreated leaves, were less pronounced (Table 3). This is because Naph has an inhibitory effect on the activity of PS-2, which is identical to the influence of Diuron. On the other hand, inhibition of Naph-pretreated leaves in the presence of the exogenous electron donor sodium ascorbate resulted in a partial restoration of the shape of the OJIP curve and parameters calculated based on it. These data suggest that Naph can inhibit not only the acceptor, but also partly the donor side of PS-2. Conclusions about the action of PAHs including Naph and anthracene on both the acceptor and donor sides of PS-2 were made also in papers by Axmann and Tukaj [23] and by Kummerova et al. [24].
The redox state of the primary quinone electron acceptor of PS-2, QA, defines the fluorescence yield of Chl a and the intensity change in the OJ phase of OJIP-transition [25]. Under stationary conditions, measured reconstitution of the formulations of PS-2 QA by a saturating light reaches its maximum after 2 ms (I2ms) after turning on the light. We have shown that the rate of QA to QA– reduction as a result of exciton absorption in PS-2 RC per RC (TR0/RC value) and the rate of photon absorption by PS-2 antenna chlorophyll per RC (ABS/RC value) increased after incubation of leaves in Naph solution. A similar increase in TR0/RC and ABS/RC values was previously observed under the influence of drought [26]. The increase in the QA to QA– reduction rate observed by us may be associated with an increase in the number of inactive RCs. The increase in the ABS/RC value can be attributed to a decrease in the number of active PS-2 centers.
It was shown that Naph led to a decrease in quantum yield of electron transport at the QA–QB site (φET2o) and from QA to electron acceptors of PS-1 (φRE1o) that, along with a decrease in the pool of reduced plastoquinone carriers (Tables 3 and 4), indicates the inhibition of the PS-2 acceptor side activity by Naph. This result is consistent with a reduction in the amplitude of the fast component DFl, I2, as a result of exposure to Naph. We found a significant reduction in the magnitude φRE1o compared with the value φET2o in leaves after their incubation in a Naph solution. This suggests that Naph reduces the rate of electron transport in not only the site from QA to QB, but also elsewhere in the photosynthetic electron transport chain, probably in sites between the PS-2 and PS-1. In this case, there is a correlation between the decrease in the value of φET2o and ratio RC/CS with increasing concentration of Naph. Probably decrease in both quantities is due to reduction in the number of active QB-reducing centers of PS-2.
Previously, it was shown [13] that Naph acts on thylakoid membranes, causing in them certain disorders. We assume that Naph also acts on the associated PS-2 antenna complex. This can lead to disruption of conjugation between the Chl molecules in the antenna complex and as a result, enhanced antenna Chl fluorescence, which may increase the Fo level. Inactivation of the PS-2 can also lead to detachment of the PS-2 antenna complex from this photosystem [24] and, thus, increase Fo.
The effect of Naph on PS-2 activity was also investigated by measuring DFl induction curves. A typical Chl a DFl curve (see Fig. 1) is characterized by several phases: fast (characterized by amplitudes I1 and I2 and times of the order of 30-150 ms) and slow (maximum at 3-10 s, characterized by amplitude Im), which are separated by a minimum, denoted as D. The relative amplitude of the DFl I2 component (ratio (I2 – D)/D) reflects the rate of electron transport on the acceptor side of PS-2 depending on the redox state of QA, QB, and the plastoquinone pool PQ [27, 28]. We observed a decrease in this ratio with increasing concentration of Naph, most likely due to increased number of non-reducing QB reaction centers of PS-2. The ratio of (Im – D)/D reflects the photoinduced formation of a proton gradient in the thylakoid membranes. Formation of ΔpH, needed for a high level of non-photochemical quenching, requires the functional integrity of thylakoid membranes [29]. Thus, Naph induces oxidative stress, leading to a significant disruption of the integrity of the thylakoid membrane, which was observed by Kreslavski et al. [13].
Depending on the measuring conditions and the functional state of the PS-2 oxygen-evolving complex (OEC), DFl of Chl a can be illuminated as a result of charge recombination in pairs [QA–P680+], [QA–YZ+], where P680 and YZ+ are the primary and secondary PS-2 electron donors, chlorophyll P680, and tyrosine 161 of D1 protein, respectively; as well as in pairs [QA–Sn+], [QB–P680+], [QB–YZ+], and [QB–Sn+], where Sn are the S-states of the PS-2 OEC [9]. In the presence of Diuron blocking electron transfer from QA to QB, millisecond DFl arises mainly as a result of the charge recombination between QA– and positively charged components of the donor side of PS-2. According to our data, the relative amplitude of the fast component of DFl ((I1 – D)/D) with a maximum of 50 ms (which most appropriately correlates with the [QA–YZ+] pair lifetime) is reduced by treatment with Diuron to the least extent (see Fig. 3). Apparently, the fastest component I1 is related to the recombination of charges in the [QA–YZ+] pair to a greater extent than other components.
Membrane reactions, in particular enhanced membrane permeability, occur rather rapidly and are a primary cell response to various stress factors. The structure of the cell membrane of a plant determines its properties, physiological activity, and resistance to stress, while antioxidants, embedded in the membrane, protect it from destruction by oxidants and maintain optimum regulatory functions [30].
It is assumed that photosynthetic membranes are targets for the action of PAHs [31]. The increase in the conductivity of aqueous solutions with the introduction of the leaves incubated in Naph solution (see Fig. 4) seen by us is consistent with this idea and indicates a change in the permeability of cell membranes after incubation of leaves with Naph. Changes in conductivity was observed after 15 min and a significant increase in the pool of H2O2 was observed after 30 min. It is likely that increase in the permeability of membranes under the influence of Naph associated with subsequent changes in the content of H2O2 and other reactive oxygen species (ROS) occurs due to a decrease in PS-2 activity. Although reduction of the Chl and carotenoid content at high Naph concentrations was only slight, it may also contribute to reducing the activity of the PS-2 by prolonged Naph exposure that was demonstrated in the work of Jajoo et al. [20].
Why does an increase in ROS affect electron transfer from QA– and further and partly on the PS-2 donor side? It is known that ROS generated during stress caused by the influence of stress factors of different nature inhibit the synthesis of photosynthetic proteins de novo [32]. Proteins of PS-2 RC, D1 and D2, are the most vulnerable. Because these proteins are involved in the organization of the PS-2 RC, the structure of PS-2 is broken, which can reduce the number of active PS-2 RCs, i.e. QB-reducing RCs. In particular, disruption of D1 protein synthesis may reduce the effectiveness of electron transfer from QA to QB.
This work was supported by the Russian Foundation for Basic Research (grants 14-04-92690 and 12-04-01035) and the program of the Presidium of the Russian Academy of Sciences “Molecular and Cell Biology”.
REFERENCES
1.Fang, G.-C., Wu, Y.-S., Chen, J.-C., Chang, C.-N.,
and Ho., T.-T. (2006) Characteristic of polycyclic aromatic hydrocarbon
concentrations and source identification for fine and coarse
particulates at Taichung Harbor near Taiwan Strait during 2004-2005,
Sci. Total Environ., 366, 729-738.
2.Simonich, S., and Hites, R. A. (1994) Importance of
vegetation in removing polycyclic aromatic hydrocarbons from the
atmosphere, Nature, 370, 49-51.
3.Chirkova, T. V. (2002) Physiological Basis of
Plant Resistance [in Russian], St. Petersburg State University, St.
Petersburg.
4.Kummerova, M., Krulova, J., Zezulka, S., and
Triska, J. (2006) Evaluation of fluoranthene phytotoxicity in pea
plants by Hill reaction and chlorophyll fluorescence,
Chemosphere, 65, 489-496.
5.Kummerova, M., and Vanova, L. (2007) Chlorophyll
fluorescence as an indicator of fluoranthene phototoxicity, Plant
Soil Environ., 53, 430-436.
6.Jajoo, A., Rao, M. N., Tomar, R. S., Grieco, M.,
Tikkanen, M., and Aro, E.-M. (2014) Inhibitory effects of polycyclic
aromatic hydrocarbons (PAHs) on photosynthetic performance are not
related to their aromaticity, J. Photochem. Photobiol. B,
135, 151-155.
7.Bigler, W., and Schreiber, U. (1990) Chlorophyll
luminescence as an indicator of stress-induced damage to the
photosynthetic apparatus, Photosynth. Res., 25,
161-171.
8.Stirbet, A., and Govindjee (2011) On the relation
between the Kautsky effect (chlorophyll a fluorescence induction) and
photosystem II: basics and applications of the OJIP fluorescence
transient, J. Photochem. Photobiol. B, 104, 36-57.
9.Kalaji, H. M., Golstev, V., Bosa, K.,
Allakhverdiev, S. I., Strasser, R. J., and Govindjee (2012)
Experimental in vivo measurements of light emission in plants: a
perspective dedicated to David Walker, Photosynth. Res.,
114, 69-96.
10.Strasser, R. J., Srivastava, A., and
Tsimilli-Michael, M. (2000) in Probing Photosynthesis:
Mechanisms, Regulation and Adaptation (Yunus, M., Pathre,
U., and Mohanty, P., eds.) Taylor and Francis, London, pp. 445-483.
11.Strasser, R. J., Tsimilli-Michael, M., and
Srivastava, A. (2004) in Chlorophyll a Fluorescence: a Signature of
Photosynthesis, Advances in Photosynthesis and Respiration
(Papageorgiou, G. C., and Govindjee, eds.) Springer, Dordrecht, pp.
321-362.
12.Perlman, R. S., Yalkowsky, S. H., and Banerjee,
S. (1984) Water solubilities of polynuclear aromatic and heteroaromatic
compounds, J. Phys. Chem. Ref. Data, 13,
555-562.
13.Kreslavski, V. D., Lankin, A. V., Vasilyeva, G.
K., Lyubimov, V. Yu., Semenova, G. N., Schmitt, F.-J., Friedrich, T.,
and Allakhverdiev, S. I. (2014) Effects of polyaromatic hydrocarbons on
photosystem II activity in pea leaves, Plant Physiol. Biochem.,
81, 135-142.
14.Mehta, P., Kreslavsky, V., Bharti, S.,
Allakhverdiev, S. I., and Jajoo, A. (2011) Analysis of salt stress
induced changes in photosystem II heterogeneity by prompt fluorescence
and delayed fluorescence in wheat (Triticum aestivum) leaves,
J. Photochem. Photobiol. B, 104, 308-313.
15.Lichtenthaler, H. K., and Wellburn, A. R. (1987)
Chlorophylls and carotenoids: pigments of photosynthetic
biomembranes, Methods Enzymol., 148, 350-382.
16.Genty, B., Briantais, J-M., and Baker, N. R.
(1989) The relationship between the quantum yield of photosynthetic
electron transport and quenching of chlorophyll fluorescence,
Biochim. Biophys. Acta, 990, 87-92.
17.Liu, H., Weisman, D., Ye, Y. B., Cui, B., Huang,
Y. H., Colon-Carmona, A., and Wang, Z. H. (2009) An oxidative stress
response to polycyclic aromatic hydrocarbon exposure is rapid and
complex in Arabidopsis thaliana, Plant Sci., 176,
357-382.
18.Bukhov, N. G., and Carpentier, R. (2000)
Heterogeneity of photosystem II reaction centers as influenced by heat
treatment of barley leaves, Physiol. Plant., 110,
279-285.
19.Singh-Tomar, R., and Jajoo, A. (2013) Alteration
in PS II heterogeneity under the influence of polycyclic aromatic
hydrocarbon (fluoranthene) in wheat leaves (Triticum aestivum),
Plant Sci., 209, 58-63.
20.Jajoo, A., Mekala, N. R., Tomar, R. S., Grieco,
M., Tikkanen, M., and Aro, E.-M. (2014) Inhibitory effects of
polycyclic aromatic hydrocarbons (PAHs) on photosynthetic performance
are not related to their aromaticity, J. Photochem. Photobiol.
B, 137, 151-155.
21.Velthuys, B. R. (1981) Electron-dependent
competition between plastoquinone and inhibitors for binding to
photosystem II, FEBS Lett., 126, 277-281.
22.Smirnoff, N., and Wheeler, G. L. (2000) Ascorbic
acid in plants: biosynthesis and function, Crit. Rev. Plant
Sci., 19, 267-290.
23.Aksmann, A., and Tukaj, Z. (2008) Intact
anthracene inhibits photosynthesis in algal cells: a fluorescence
induction study on Chlamydomonas reinhardtii cw92 strain,
Chemosphere, 74, 26-32.
24.Kummerova, M., Vanova, L., Krulova, J., and
Zezulka, S. (2008) The use of physiological characteristics for
comparison of organic compounds phytotoxicity, Chemosphere,
71, 2050-2059.
25.Van Gorkom, H. J. (1986) in Light Emission by
Plants and Bacteria (Govindjee, Amesz, J., and Fork, D. C., eds.)
Academic, Orlando, pp. 267-289.
26.Rathod, D. P., Brestic, M., and Shao, H. B.
(2011) Chlorophyll a fluorescence determines the drought
resistance capabilities in two varieties of mycorrhized and
non-mycorrhized Glycine max Linn, Afr. J. Microbiol.
Res., 5, 4197-4206.
27.Goltsev, V. N., Kuzmanova, M. A., Kaladzhi, H.
M., and Allahverdiev, S. I. (2014) Variable and Delayed Fluorescence
of Chlorophyll a – Theoretical Basis and Practical
Application in the Study of Plants [in Russian], Institute of
Computer Science, Izhevsk-Moscow.
28.Goltsev, V., Zaharieva, I., Chernev, P., and
Strasser, R. J. (2009) Delayed fluorescence in photosynthesis,
Photosynth. Res., 101, 217-232.
29.Zhang, L., and Xing, D. (2008) Rapid
determination of the damage to photosynthesis caused by salt and
osmotic stresses using delayed fluorescence of chloroplasts,
Photochem. Photobiol. Sci., 7, 352-360.
30.Chirkova, T. V. (1997) Cell membranes and plant
resistance to stresses, Soros Obrazovat. Zh., 9,
12-17.
31.Duxbury, C. L., Dixon, D. G., and Greenberg, B.
M. (1997) Effects of simulated solar radiation on the bioaccumulation
of polycyclic aromatic hydrocarbons by the duckweed Lemna gibba,
Environ. Toxicol. Chem., 16, 1739-1748.
32.Murata, N., Takahashi, S., Nishiyama, Y., and
Allakhverdiev, S. I. (2007) Photoinhibition of photosystem II under
environmental stress, Biochim. Biophys. Acta, 1767,
414-421.