Caenorhabditis elegans Eyes Absent Ortholog EYA-1 Is Required for Stress Resistance
Bing-ying Wang1,2, Xue-song Xu3, Yu-xiao Cui1,2, Hua Wang1,2, Ge Liu1,2, Zhizhuang Joe Zhao4, Jun-feng Ma1,2*, and Xue-qi Fu1,2*
1The State Engineering Laboratory of AIDS Vaccine, Jilin University, Changchun, P. R. China; fax: 86-431-85155152; E-mail: mjf@jlu.edu.cn; fxq@jlu.edu.cn2Key Laboratory for Molecular Enzymology and Engineering, Ministry of Education, Jilin University, Changchun, P. R. China; E-mail: wangbingyingicy@163.com; 363906218@qq.com; 253815665@qq.com; 874227993@qq.com
3China-Japan Union Hospital, Jilin University, Changchun 130033, P. R. China; E-mail: xuesongx@126.com
4Department of Pathology, University of Oklahoma Health Sciences Center, Oklahoma City, Oklahoma 73104, USA; E-mail: joe-zhao@ouhsc.edu
* To whom correspondence should be addressed.
Received March 8, 2014; Revision received April 2, 2014
Eyes absent (Eya) is a highly conserved transcription cofactor and protein phosphatase that regulates multiple developmental processes throughout the metazoans. It is a dual function protein, working as a transcription factor in the nucleus and as a tyrosine phosphatase in the cytoplasm. In this study, we isolated EYA-1 of Caenorhabditis elegans, the only homolog of Eyes absent, and set up an effective feeding-based RNAi (RNA interference) against the gene. We found that knockdown of EYA-1 decreased heat and oxidative stress tolerance and accelerated the onset of paralysis mediated by Aβ1-42 proteotoxicity and polyQ. Under heat stress (35°C), EYA-1 knockdown shortened the mean lifespan by 16.8%, which could be attributed to decrease in heat shock protein-16.2 (hsp-16.2) expression. Under oxidative stress, EYA-1 knockdown could shorten the mean lifespan by 18.7%, which could be attributed to intracellular ROS accumulation and the decrease of superoxide dismutase-3 (sod-3) protein expression. Moreover, EYA-1 knockdown animals also showed increased lipofuscin accumulation under oxidative stress. Further studies demonstrated that EYA-1 knockdown could not inhibit daf-16 nuclear accumulation in wild-type worms in response to stress. On the other hand, EYA-1 deficiency did not further reduce stress resistance of daf-16 mutants, which are stress sensitive. Quantitative real-time PCR results also showed that the expression of two daf-16 target genes, hsp-12.3 and sod-3, was downregulated in EYA-1 RNAi-treated worms under stress. All this evidence indicates EYA-1 is required for stress resistance of worms, and it might act downstream of daf-16 to regulate expression of stress resistance-associated genes.
KEY WORDS: EYA-1, Caenorhabditis elegans, RNA interference, stress resistanceDOI: 10.1134/S0006297914070074
Abbreviations: GFP, green fluorescent protein; HSP-16.2, heat-shock protein; NGM, nematode growth medium; RNAi, RNA interference; ROS, reactive oxygen species.
Eyes absent (eya) encodes a highly conserved
transcriptional coactivator and protein phosphatase. It was first
identified in Drosophila as a gene required for eye development
[1-3], and its homologs are
vital in the development of different organisms ranging from insects to
humans [4, 5]. In higher
animals, target genes of eya are implicated in the development
of multiple organs, such as the eyes, muscles, ears, heart, lungs,
endocrine glands, placodes, pharyngeal pouches, craniofacial skeleton,
and parathyroid [6]. In Drosophila, specific
and recessive eya mutations result in elimination of compound
eyes in viable flies, whereas eya null mutations are lethal to
embryos [1]. Mouse mutants for eya display
abnormal apoptosis and reduced cell proliferation during the
development of multiple tissues, such as kidney, muscle, and ear [7]. In humans, eya is also implicated in
several diseases, such as the multi-organ developmental disorder
branchio-oto-renal syndrome [8], congenital
cataracts [9], and late-onset deafness [7], and it is overexpressed in numerous types of
cancer [10, 11].
The genome of the nematode Caenorhabditis elegans contains a homolog of the “eye specification” gene, EYA-1. Furuya et al. [12] reported that loss of EYA-1 function by RNA interference (RNAi) and deletion mutations resulted in early larval mortality with incomplete penetrance, which was associated with defects in the differentiation and morphogenesis of several tissues and organs. Hirose et al. [13] reported that the C. elegans Six-family homeodomain protein (CEH-34) and the eyes absent ortholog (EYA-1) promote programmed cell death of a specific pharyngeal neuron. In this study, we isolated EYA-1 of C. elegans, the only homolog of Eyes absent (Eya), and set up an effective feeding-based RNA interference against the gene. Importantly, we found that although knockdown of EYA-1 in C. elegans could not change the lifespan of the worms under normal culture conditions, it could cause a significant shortening of the lifespan under stress condition. Therefore, we suggested that EYA-1 may be required for stress resistance in C. elegans. Our results also indicate that in C. elegans, EYA-1 is involved in daf-16-mediated stress resistance. Moreover, knockdown of EYA-1 reduced stress resistance by downregulating the expression of stress-resistance-associated genes, such as sod-3, hsp12.3, and hsp-16.2. We believe that these findings will provide new insights into the role of EYA proteins.
MATERIALS AND METHODS
Strains of C. elegans. All C. elegans strains were maintained at 20°C, except that of temperature-sensitive strain CL4176. The strains used were: N2 Bristol (wild type), CL2070 (hsp16.2::gfp), CF1553 muIs84 [pAD76(sod-3::gfp), BA17, fem-1(hc17) (fertile at 20°C, infertile at 25°C), CL4176 (dvIs27[pAF29(myo-3/Aβ 1-42/let UTR) + pRF4(rol-6(su1006)]), AM140 (rmIs132[P(unc-54) Q35::YFP]), TJ356 zIs356 [daf-16::gfp + rol-6] and CF1038 daf-16(mu86).
Molecular cloning of EYA-1. We first considered full sequences of the EYA-1 gene of C. elegans from www.wormbase.org and amplified the coding sequence with primers EYAf: 5′-AAAATGCTTCCAGATTCTGAGGGTCAAAA-3′ and EYAr: 5′-ATTATCCGCCCAACAAAAAGTTGTCCA-3′. PCR products of 1501-bp length were cloned into the vector pBluescript II KS. Sequencing analysis revealed that our sequences are identical to that in the database.
Knockdown of EYA-1. The full-length coding sequence of EYA-1 was cloned into the pPD129.36 vector. Plain p129.36 vector was used as the control throughout the study. Escherichia coli HT115 (DE3) were employed as hosts for the expression of double-stranded RNAs (dsRNAs), and 0.4 mM isopropyl β-D-1-thiogalactopyranoside (IPTG) was used to induce dsRNA expression. The efficiency of RNAi-mediated knockdown was confirmed by Western blot with anti-EYA-1 antibodies (prepared in Dr. Zhao’s lab).
Preparation of C. elegans protein extracts and Western blot analysis. Worms in a mixed population were collected and extracted by sonication in whole cell extraction buffer containing 25 mM 2-glycerol phosphate (pH 7.3), 5 mM EDTA, 5 mM EGTA, 0.1 M NaCl, 1% Triton X-100, 10 mM β-mercaptoethanol, and a cocktail of protease inhibitors (Roche Applied Science, Germany). Samples containing 20 μg of total protein were separated on 10% SDS gel and transferred to polyvinylidene difluoride (PVDF) membrane for Western blot analysis using the enhanced chemiluminescence method.
Immunohistochemistry. Worms were pelleted by brief centrifugation and then fixed overnight at 48°C with 4% paraformaldehyde, dehydrated in graded ethanol, embedded in paraffin, and sectioned at 3 μm thick. Sections were first heated with 0.01 M sodium citrate buffer (pH 6) in a water bath at 95°C for 15 min, and then sequentially reacted with primary and secondary antibodies (biotin-conjugated horse anti-mouse IgG; Vector Laboratories, USA).
Lifespan assays and stress resistance. Fertile adults were transferred to fresh nematode growth media (NGM) plates and allowed to lay eggs for 2 h for producing age-synchronized groups of worms. Age synchronized young larvae (L1) were cultured on NGM plates containing E. coli cells carrying the vector control or EYA-1 RNAi and maintained at 20 or 25°C. Once the worms reached adulthood, they were transferred daily for 6 days until egg laying ceased to avoid overlapping of generations. After these 6 days, the worms were transferred every second day. The worms were scored as dead if they did not respond to touch stimulus.
Heat-shock assays were performed at 30 and 35°C, using 4-day-old adults. Both control and RNAi-treated worms were cultured at 20°C for 4 days and then transferred to an incubator set to 30 or 35°C. The number of dead worms was counted every 2 h at 30°C, and every half an hour at 35°C.
High concentrations of juglone (5-hydroxy-1,4-naphthoquinone), which is a generator of reactive oxygen species (ROS), have a damaging effect on cells and organisms. Age synchronized young larvae (L1) were cultured on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi and maintained at 20°C for 4 days. Then both control and RNAi-treated worms were transferred into 96-well plates containing 200 μM of juglone in S-medium. The number of dead worms was counted every hour. For all lifespan assays, every experiment was repeated three times and conducted in double-blind manner.
Fluorescence quantification and microscopy of transgenic C. elegans (CL2070, hsp-16.2::gfp). The expression of hsp-16.2::gfp (green fluorescent protein) in CL2070 worms was investigated by fluorescence microscopy. Both control and RNAi-treated worms were treated at 35°C for 1 h and allowed to recover for 24 h. For quantification, the worms were anesthetized with sodium azide (10 mM) on a 5% agarose pad on a glass slide, and the fluorescence was viewed under a microscope (Olympus, Japan) with excitation at 488 nm and emission at 500 to 530 nm. The fluorescence intensity was analyzed using the Image-Pro Plus software (Media Cybernetics). Each experiment was repeated twice, and 15 worms per group were used in each experiment. The data represent mean ± standard deviation (n = 30) of the two biological experiments.
Measurement of intracellular reactive oxygen species (ROS) in C. elegans. Intracellular ROS in C. elegans were measured with H2DCF-DA (2′,7′-dichlorodihydrofluorescein diacetate) as a molecular probe. For ROS detection under normal culture conditions, both control and RNAi-treated worms were incubated at 20°C for 4 days. On day 4, for the ROS test under oxidative stress, worms were treated with 200 μM juglone for 2 h. At the end of the specified treatment time, the worms were collected into 100 μl phosphate-buffered saline (PBS) with 1% Tween 20 in Eppendorf tubes. They were then pipetted into the wells of a Costar 96-well microtiter plate (black, clear, flat-bottom wells) containing H2DCF-DA (final concentration 200 μM in PBS). Samples were read in a Thermo Labsystems Fluoroskan Ascent microplate reader at 37°C with excitation 485 nm and emission 530 nm.
Lipofuscin accumulation and fluorescence quantification of transgenic C. elegans (CF1553, sod-3::gfp). Age synchronized BA17 or CF1553 worms were cultured on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi and maintained at 20°C for 4 days, followed by 200 μM juglone for 2 h. Then individuals were anesthetized with sodium azide (10 mM) on 5% agarose pad on glass slides and the fluorescence was viewed under a microscope (Olympus, Japan) with excitations at 488 nm and emissions at 500 to 530 nm. The fluorescence intensity was analyzed using Image-Pro Plus software. Each experiment was repeated twice and 15 worms per group were used in each experiment. Data represent mean ± standard deviation (n = 30) of two biological experiments.
Worm paralysis assays. For strain CL4176 (dvIs27[pAF29(myo-3/Aβ 1-42/let UTR) + pRF4(rol-6(su1006)]), worms were prepared by synchronous egg laying and induced to express Aβ as third-stage larvae by upshift from 15 to 25°C on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi. All paralysis plots were done in triplicate with 100 worms per condition. For strain AM140 (rmIs132[P(unc-54) Q35::YFP]), age synchronized worms were cultured on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi at 20°C. Paralyzed worms were identified as those failing to make forward or backward movement in response to stimulation by tail-prodding.
Quantitative real-time PCR. Total RNA was extracted from adult worms with TRIzol reagent (Invitrogen, USA), and cDNA was produced by oligo(dT) priming. The reverse transcription PCR (RT-PCR) primers were as follows: act-1, 5′-GTCATGGTCGGTATGGGACA-3′ and 5′-TTCGTAGATTGGGACGGTGT-3′; hsp-12.3, 5′-ACAACTTCTTGCCAAACGA-3′ and 5′-CTGCCATCCAACTTGCTC-3′; sod-3, 5′-CAAAAGCATCATGCCACCTA-3′ and 5′-CTCCCAAACGTCAATTCCA-3′. We used act-1 as an internal control for equal RNA loading. The mRNA expression was assessed by quantitative real-time PCR on a Bio-Rad IQ5 multicolor real-time PCR detection system using SYBR green as the detection method. The gene expression data were analyzed using the comparative 2−ΔΔCt method, with act-1 mRNA as the control.
Statistical analysis. Statistical comparison between control and RNAi-treated worms was performed with unpaired t-test using the GraphPad Prism software (GraphPad Software, USA). Differences with p < 0.05 were considered significant.
RESULTS
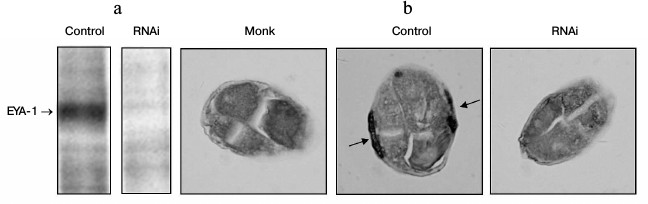
In this study, the full-length coding sequence of EYA-1 was cloned into the pPD129.36 vector that contains the T7 promoter at both 5′ and 3′ ends to drive transcription. The wild-type N2 worms were fed with HT115 cells that contained the vector control or EYA-1 RNAi. EYA-1 protein expression was detected by Western blot and immunohistochemistry with anti-EYA-1 antibodies. As shown in Fig. 1a, treatment of worms with E. coli cells expressing dsRNA derived from a full-length EYA-1 cDNA resulted in almost total deletion of EYA-1. Moreover, the protein of EYA-1 was mainly expressed in body wall muscle cells in the control, but it decreased to near absence in RNAi-treated worms (Fig. 1b). These data indicated the high efficiency of the feeding-based RNAi in knocking down the gene expression of EYA-1.
Fig. 1. RNAi-mediated knockdown resulted in almost total deletion of EYA-1. Normal N2 worms were fed with E. coli cells carrying the vector control, full-length EYA-1. a) Crude cell extracts containing equal amounts of total protein were subjected to Western blot analyses with anti-EYA-1 antibodies and then with horseradish peroxidase (HRP)-conjugated anti-mouse secondary antibodies. b) Paraffin-embedded worm sections were stained with anti-EYA-1 antibody and then with biotin-conjugated horse anti-mouse IgG. Arrows indicate that EYA-1 was expressed in the body wall muscle cells.
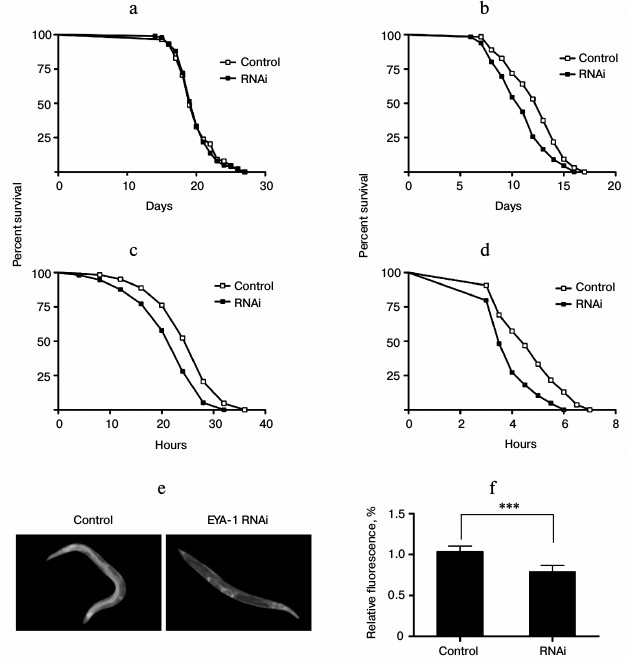
Knockdown of EYA-1 did not change the lifespan expectancy significantly at 20°C, but it shortened the lifespan of worms at 25, 30, and 35°C in a temperature-dependent manner compared with control (Fig. 2, b-d). Among the temperatures examined, knockdown of EYA-1 caused the greatest effect on lifespan at 35°C, and it reduced adult mean lifespan to 16.8%. Therefore, we consider that EYA-1 of C. elegans may be required for thermotolerance.
Hsp-16.2 can serve as a stress-sensitive reporter to predict longevity in C. elegans [14]. Higher levels of hsp-16.2::gfp predict longer mean remaining longevity of C. elegans. To study the effect of EYA-1 knockdown on hsp-16.2, C. elegans strains CL2070 (hsp-16.2::gfp) were used. Compared with the control group, the RNAi-treated group showed lower hsp-16.2::gfp intensity in pictures taken with an Olympus fluorescence microscope (Fig. 2e). After quantification using Image-Pro Plus, the data showed that knockdown of EYA-1 significantly downregulated hsp-16.2::gfp expression by 24.5% in CL2070 (Fig. 2f) (p < 0.001 compared with the control). We consider that knockdown of EYA-1 downregulates the expression of the hsp-16.2::gfp reporter gene in CL2070 under heat stress.
Fig. 2. Knockdown of EYA-1 shortens the lifespan of worms under heat stress. a, b) Survival curves of normal N2 worms cultured on NGM plates containing E. coli cells that carry the vector control or EYA-1 RNAi from day 0 to death at 20 (a) and 25°C (b). The number of dead worms was counted every day. c, d) Survival curves of normal N2 worms under heat stress. Worms were cultured on NGM plates containing E. coli cells that carry the vector control or EYA-1 RNAi at 20°C for 4 days and then transferred to an incubator set to 30 (c) and 35°C (d). This time point was represented by 0. The number of dead worms was counted every 2 h at 30°C and every half an hour at 35°C. All survival curves are presented based on three individual experiments. The data were processed with the GraphPad Prism software (n = 250), p < 0.001. e, f) Effect of EYA-1 knockdown on the expression of hsp-16.2::gfp in C. elegans. Both control and RNAi-treated worms were cultured at 20°C for 4 days and then transferred to an incubator set to 35°C for 1 h and allowed to recover for 24 h. e) Image of hsp-16.2::gfp expression in control and RNAi-treated worms. f) Quantified hsp-16.2::gfp intensity (mean ± standard deviation) in CL2070 was from three experiments in each group, with 20 worms in each experiment, as quantified by Image-Pro Plus; p < 0.001.
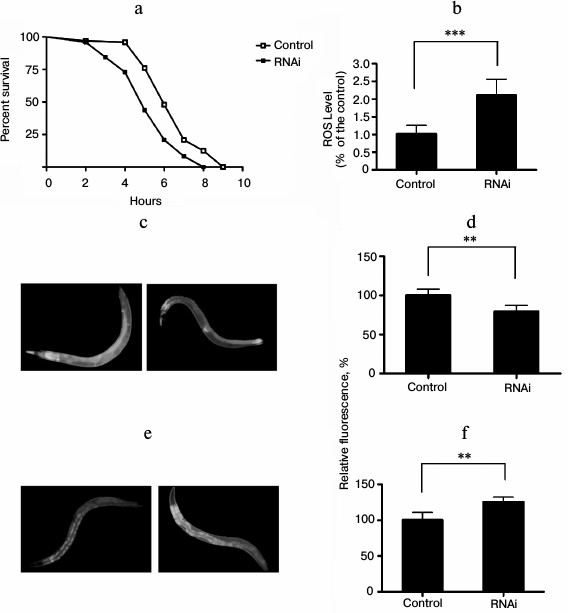
In C. elegans and other organisms, environmental conditions or genetic changes that result in increased lifespan are frequently associated with enhanced resistance to stress, including heat stress and oxidative stress [15]. To explore whether knockdown of EYA-1 can affect C. elegans against vulnerability to oxidative stress, we performed oxidative stress resistance assays. We observed that knockdown of EYA-1 caused a significant shortening of the lifespan in C. elegans under oxidative stress generated by juglone. The data showed that the mean survival rate was significantly shortened by 18.7% (Fig. 3a). Therefore, we consider that EYA-1 may be required for oxidative stress resistance in C. elegans. Furthermore, we determined intracellular ROS accumulation in 4-day-old adults, and an increase in ROS accumulation was found in RNAi-treated worms in comparison to control using a fluorescence well-plate reader to measure DCF fluorescence (Fig. 3b). According to the “free radical theory of aging” [15], ROS are a crucial factor for aging, and the intracellular amount of ROS can be correlated to stress. Thus, the increased amount of ROS in RNAi-treated worms may explain the decreased survival rate under oxidative stress conditions.
The mechanism by which EYA-1 affects oxidative stress was further studied using transgenic CF1553 worms expressing sod-3::gfp. Sod-3 is a typical scavenger enzyme in oxidative stress, and it catalyzes disproportionation of reactive superoxide anion. Fluorescence microscopy revealed a decreased expression of sod-3 by EYA-1 knockdown in CF1553. In subsequent studies, we quantified the sod-3::gfp expression using the Image-Pro Plus software. Knockdown of EYA-1 significantly downregulated the sod-3::gfp expression by 17.43% in CF1553 (Fig. 3d) (0.001 < p < 0.05 compared with the control). We consider that knockdown of EYA-1 downregulates the expression of sod-3::gfp in CF1553 under oxidative stress.
Lipofuscin is the fine yellow-brown pigment in granules composed of lipid-containing residues of lysosomal digestion. It is a product of oxidative damage and autophagy that is found in liver, kidney, heart muscle, adrenals, neurons, and ganglion cells. In C. elegans, lipofuscin is detectable as autofluorescent granules in the intestine, and its accumulation is a well-established marker of the biological age of C. elegans [16]. To estimate the effect of RNAi treatment on lipofuscin levels, the C. elegans strain BA17 was used. In comparison to the control group, the RNAi-treated group showed a significantly increased accumulation of lipofuscin (Fig. 3f) (0.001 < p < 0.05 compared with control).
Fig. 3. Knockdown of EYA-1 reduces resistance to oxidative stress in C. elegans. a) Survival curves of normal N2 worms cultured on NGM plates containing E. coli cells that carry the vector control or EYA-1 RNAi at 20°C for 4 days, then both control and RNAi-treated worms were transferred into 96-well plate containing 200 μM of juglone in S-medium. This time point was taken as 0. The number of dead worms was counted every hour. The data were processed with the GraphPad Prism software (n = 250), *** p < 0.001. b) Knockdown of EYA-1 significantly increased the intracellular ROS level in C. elegans under oxidative stress. Both control and RNAi-treated worms were incubated at 20°C for 4 days, and after 2 h under oxidative stress induced by 200 μM juglone the RNAi-treated worms showed a significantly higher DCF (2,7-dichlorofluorescein) fluorescence in comparison to control (n = 120), *** p < 0.001. All the data are presented based on three individual experiments. c, d) Effect of EYA-1 knockdown on the expression of sod-3::gfp in C. elegans. Both control and RNAi-treated worms were cultured at 20°C for 4 days followed by 200 μM juglone for 2 h. c) Image of sod-3::gfp expression in control and RNAi-treated worms. d) Quantified sod-3::gfp intensity (mean ± standard deviation) in CF1553 was from three experiments in each group, with 20 worms in each experiment, as quantified by Image-Pro Plus; ** 0.001 < p < 0.05. e) Intestinal autofluorescence images of lipofuscin in control and RNAi-treated worms. f) Quantified mean fluorescence intensity from intestinal lipofuscin (mean ± standard deviation) in BA17 was from three experiments in each group, with 20 worms in each experiment, as quantified by Image-Pro Plus; ** 0.001 < p < 0.05.
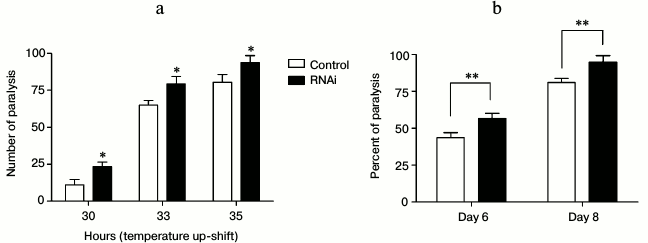
Aggregation of misfolded proteins increases with aging and causes chronic proteotoxic stress, which is the main reason for a variety of age-related neurodegenerative diseases, such as Alzheimer’s disease and Huntington’s disease. As knockdown of EYA-1 has a negative effect under conditions of heat and oxidative stress, we investigated if knockdown of EYA-1 can also cause internal proteotoxic stress in C. elegans. Two C. elegans models of human proteotoxic diseases were exploited here: the strains CL4176 (dvIs27 [pAF29(myo-3/Ab 1-42/let UTR) + pRF4(rol-6(su1006)]) and AM140 (rmIs132[P(unc-54) Q35::YFP]). The strain CL4176 is engineered to provide temperature-inducible muscle expression of a human β-amyloid peptide (Aβ) transgene, resulting in a readable paralysis phenotype of Aβ toxicity upon temperature upshift to 25°C. Synchronized eggs were cultured on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi at 15°C. Aβ induction was induced at L3 stage when the temperature was shifted to 25°C. Paralysis at several time points was monitored. A moderate acceleration in paralysis was observed in RNAi-treated transgenic animals at both time durations (Fig. 4a), suggesting that knockdown of EYA-1 can exacerbate the Aβ toxicity. Worms expressing 35 glutamine residues conjugated to YFP in body wall muscle (unc-54p::Q35::YFP) were cultured on NGM plates containing E. coli cells carrying vector control or EYA-1 RNAi at 20°C. As expected, knockdown of EYA-1 accelerated the onset of Q35::YFP-induced paralysis (Fig. 4b). Thus, we suggest that EYA-1 is required for resistance to multiple stressors, including proteotoxic injury.
Fig. 4. Knockdown of EYA-1 exacerbates proteotoxicity-mediated paralysis. a) The paralysis associated with muscle Aβ1-42 expression is exacerbated by EYA-1 knockdown from hatching in the transgenic strain CL4176. Shown is the non-average paralysis percentages in three independent experiments with 100 animals/experiment at indicated time points after temperature upshift to 25°C; * p < 0.05. b) Paralysis associated with Q35 expression is significantly deteriorated by EYA-1 knockdown from hatching in the transgenic strain AM140. A representative assay of more than three independent experiments is shown. Error bars indicates SD among the paralysis percentages of three independent experiments; ** 0.001 < p < 0.05.
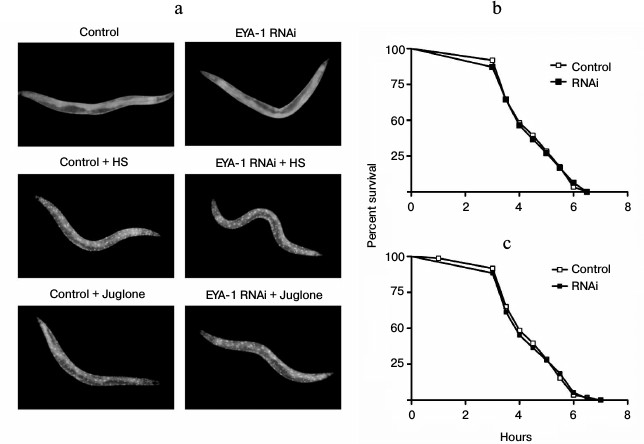
Daf-16/forkhead transcription factor, the downstream target of insulin-like signaling in C. elegans, is indispensable for lifespan regulation, stress tolerance, and resistance to proteotoxicity [17]. The daf-16 protein normally lies in the cytoplasm, and in response to stress daf-16 will translocate from the cytoplasm to the nucleus. In the nucleus daf-16 can activate transcription of a large number of genes including hsp-16.2 and sod-3, thus increasing stress resistance and longevity [18]. To examine whether or not EYA-1 plays a role in directing daf-16 to the nucleus in response to stress, we used the TJ356 (daf-16::gfp) strain to assess the effect of EYA-1 knockdown on daf-16 nuclear localization. As a result, both control and RNAi-treated group did not direct daf-16::gfp to the nucleus under normal culturing conditions, but both heat treatment at 35°C for 1 h and 200 μM juglone for 1 h resulted in rapid nuclear localization of daf-16::gfp (Fig. 5a). Our results showed that knockdown of EYA-1 could not inhibit daf-16 nuclear translocation in response to stress. Hence, EYA-1 is dispensable in directing daf-16 to the nucleus in response to stress. Consequently, we suggest that EYA-1 does not act upstream of the insulin-like signaling pathway to regulate stress resistance. Alternatively, it is also possible that EYA-1 does not act in other pathways, which can direct daf-16 to the nucleus in response to stress. On the other hand, we tested the lifespan in a daf-16 null mutant strain, which is stress sensitive. As shown in Fig. 5 (b and c), knockdown of EYA-1 did not further reduce stress resistance of daf-16 mutants, suggesting that EYA-1 knockdown decreases stress resistance of the worms in a daf-16-dependent manner. In conclusion, we consider that EYA-1 may act downstream of daf-16 to carry out its functions in response to stress.
Fig. 5. Knockdown of EYA-1 cannot inhibit daf-16 nuclear localization and shorten the lifespan of daf-16 null mutants. a) daf-16::gfp distribution was observed in daf-16::gfp animals grown on NGM plates containing E. coli cells carrying vector control and EYA-1 RNAi at 20°C (upper panel), or immediately after heat shock at 35°C for 1 h (HS) (middle panel), or 200 μM juglone for 1 h (lower panel) (n = 20). b, c) Survival curves of daf-16 (mu86) mutants cultured on NGM plates containing E. coli cells that carry the vector control or EYA-1 RNAi at 20°C for 4 days and then transferred to an incubator set to 35°C (b) or 96-well plate containing 200 μM of juglone in S-medium (c). This time point was taken as 0. The number of dead worms was counted every half an hour (n = 250). A representative assay of more than three independent experiments is shown.
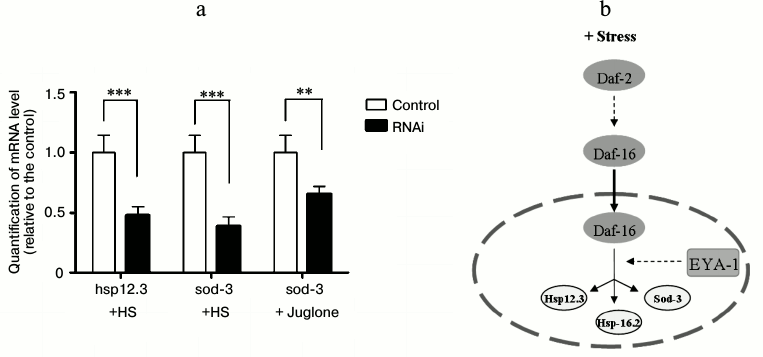
Daf-16 shows its functions by transcriptional regulation of a large number of target genes [18]. The role of EYA-1 in daf-16-dependent gene expression was tested in the context of two well-known daf-16 targets, hsp-12.3 and sod-3. Hsp-12.3 is a small heat shock protein that is activated by daf-16 at the transcriptional level. Sod-3, one of the downstream effectors of daf-16, is also an important regulator of lifespan and stress resistance in C. elegans. Under heat stress conditions, EYA-1 RNAi led to decreased mRNA levels of two genes positively regulated by daf-16 (Fig. 6a). On the other hand, under oxidative stress conditions, EYA-1 RNAi led to decreased mRNA level of sod-3 (Fig. 6a), which helped explain why knockdown of EYA-1 caused a significant shortening of lifespan of C. elegans under stress conditions. Moreover, these results imply that EYA-1 is involved in daf-16-mediated stress resistance for carrying out its functions by transcriptional regulation of daf-16-dependent target genes.
Fig. 6. EYA-1 is required for optimal daf-16-dependent gene expression. a) Normal N2 worms were cultured on NGM plates containing E. coli cells that carry the vector control or EYA-1 RNAi at 20°C for 4 days and then transferred to 35°C for 2 h or 200 μM juglone for 2 h. Relative mRNA levels of hsp-12.3 and sod-3 were measured by quantitative RT-PCR. The representative of three independent experiments with similar results is shown. Error bars indicate standard deviation among three replicates in RT-PCR; t-test, ** 0.001 < p < 0.05, *** p < 0.001. b) Sketch: EYA-1 presumably acts downstream of daf-16 and function during stress. See text for details.
DISCUSSION
The Eyes absent (Eya) proteins, first identified in Drosophila, are now implicated in processes as disparate as organ development, innate immunity, DNA damage repair, photoperiodism, angiogenesis, and cancer metastasis. It is expressed in the nuclei of a subset of anterior cells, which includes pharyngeal and body wall muscle cells, starting from the morphogenesis stage in embryogenesis [6]. In this study, we have cloned a C. elegans ortholog of Eya, EYA-1. We found that knockdown of EYA-1 using feeding-based RNAi leads to a nearly total loss of EYA-1 expression (Fig. 1a) but could not change the lifespan of C. elegans under normal culture condition (Fig. 2a). Interestingly, EYA-1 knockdown caused a significant shortening of lifespan under heat stress and oxidative stress. Because hsp-16.2 can serve as heat stress-sensitive reporter to predict longevity in C. elegans [14], the effects of EYA-1 RNAi on the expression of hsp-16.2 protein were investigated. RNAi-treated worms showed significantly lower expression of heat shock proteins as determined through analysis of gfp reporter expression, which might explain why knockdown of EYA-1 decreased thermotolerance in the C. elegans. In C. elegans pretreated with juglone, a generator of ROS, a subsequently increased ROS accumulation was observed. Therefore, oxidative stress generated by juglone can result in a shortened lifespan in N2 worms. Does EYA-1 knockdown reduce oxidative stress resistance of C. elegans to shorten its lifespan by accumulating ROS? We got a positive answer from this study. We found that EYA-1 knockdown significantly upregulated ROS levels under oxidative stress (Fig. 3b). Combined, these results suggested that the sensitivity of RNAi-treated worms to oxidative stress is a specific sensitivity resulting from their decreased ability to detoxify ROS. In addition, EYA-1 knockdown could downregulate the expression of sod-3 protein in C. elegans, which might explain why EYA-1 knockdown could significantly decrease the survival of C. elegans under oxidative stress. Another oxidative damage marker investigated in this study was lipofuscin, also called age pigment. It is a brown-yellowish autofluorescent material, which accumulates in postmitotic cells [19]. The current study showed that knockdown of EYA-1 could increase the amount of lipofuscin in the worms’ intestines (Fig. 3e). Previously, oxidative stress, a condition describing the production of oxygen radicals beyond a threshold for proper neutralization by antioxidant, has been implicated as a pathologic condition in several neurodegenerative disorders, such as Alzheimer’s disease and Huntington’s disease [20]. The present study demonstrated that EYA-1 RNAi accelerated the onset of paralysis mediated by Aβ1-42 proteotoxicity and polyQ in CL4176 and AM140, respectively. Oxidative stress is involved in Alzheimer’ and Huntington’ pathogenesis; moreover, accumulation of ROS along with the aggregation of toxic amyloid species and Q35-YFP (Q35) is proposed to exacerbate the condition in Alzheimer and Huntington patients [21-23]. Previous study has shown that EYA-1 knockdown reduced oxidative stress resistance in C. elegans, which might be one of the contributing factors for the increased onset of paralysis in the worms.
In C. elegans, daf-16 regulates a wide variety of genes involved in longevity, stress tolerance, resistance to proteotoxicity, metabolism, and development [16-18]. In response to stress, daf-16 translocates from the cytoplasm to the nucleus. Once daf-16 enters the nucleus, it enhances the expression of numerous target genes to prevent damage from harmful stresses, which would then confer increased stress resistance and help to maintain normal life in C. elegans. Approaching the question on the mode of action of EYA-1, we used transgenic worms TJ356 (daf-16::gfp) in which a reporter protein (green fluorescent protein, gfp) is fused to the predicted last amino acid of the daf-16 polypeptide chain [24]. Under normal conditions daf-16::gfp remains in the cytoplasm, but under thermal stress (35°C, 1 h), or oxidative stress (200 μM juglone, 1 h), a strong nuclear localization of daf-16::gfp was observed in control and RNAi-treated worms (Fig. 5a). These results suggest that EYA-1 knockdown could not impair stress-induced daf-16 nuclear localization. However, the lifespan of daf-16 null mutants showed that knockdown of EYA-1 decreased stress resistance to heat shock and oxidative stress in a daf-16-dependent manner (Fig. 5b). Therefore, we suggest that EYA-1 may act downstream of daf-16 to carry out its functions in response to stress. The daf-16 transcription factor is known to upregulate a large number of genes that increase stress resistance and extend the lifespan of worms. Knockdown of EYA-1 has been shown to reduce the stress resistance of C. elegans in a daf-16-dependent manner, and these effects may be due to inhibition of daf-16-dependent gene expression that reduces stress resistance. As expected, quantitative real-time PCR results suggested that knockdown of EYA-1 significantly downregulated the expression of the stress resistance-associated genes hsp-12.3 and sod-3 (Fig. 6a), which in turn explained why knockdown of EYA-1 significantly shortened the lifespan of the worms under stress.
In conclusion, EYA-1 is involved in daf-16-mediated stress resistance, and it may act downstream of daf-16 through regulating stress-resistance-associated genes such as hsp-12.3 and sod-3 to carry out its functions (Fig. 6b). Our findings reveal a novel role of the EYA-1 protein in stress resistance.
This work was supported by the National Natural Science Foundation of China (grant No. 31000358 for J. Ma) and Project of State Key Laboratory of Superhard Materials of Jilin University (No. 201306).
REFERENCES
1.Bonini, N. M., Leiserson, W. M., and Benzer, S.
(1993) The eyes absent gene: genetic control of cell survival
and differentiation in the developing Drosophila eye,
Cell, 72, 379-395.
2.Rayapureddi, J. P., Kattamuri, C., Steinmetz, B.
D., Frankfort, B. J., Ostrin, E. J., Mardon, G., and Heqde, R. S.
(2003) Eyes absent represents a class of protein tyrosine phosphatases,
Nature, 426, 295-298.
3.Tootle, T. L., Silver, S. J., Davies, E. L.,
Newman, V., Latek, R. R., Mills, I. A., Selenqut, J. D., Parlikar, B.
E., and Rebay, I. (2003) The transcription factor Eyes absent is a
protein tyrosine phosphatase, Nature, 426, 299-302.
4.Bessarab, D. A., Chong, S. W., and Korzh, V. (2004)
Expression of zebrafish six1 during sensory organ development
and myogenesis, Dev. Dyn., 230, 781-786.
5.Xu, P. X., Woo, I., Her, H., Beier, D. R., and
Maas, R. L. (1997) Mouse eya homologues of the Drosophila
eyes absent gene require Pax6 for expression in lens and nasal
placode, Development, 124, 219-231.
6.Tadjuidje, E., and Hegde, R. S. (2012) The Eyes
absent proteins in development and disease, Cell Mol. Life Sci.,
70, 1897-1913.
7.Zou, D., Silvius, D., Rodrigo-Blomqvist, S.,
Enerback, S., and Xu, P. X. (2006) Eya1 regulates the growth of otic
epithelium and interacts with Pax2 during the development of all
sensory areas in the inner ear, Dev. Biol., 298,
430-441.
8.Abdelhak, S., Kalatzis, V., Heilig, R., Compain,
S., Samson, D., Vincent, C., Weil, D., Cruaud, C., Sahly,
I., Leibovici, M., Bitner-Glindzicz, M., Francis, M., Lacombe,
D., Vigneron, J., Charachon, R., Boven, K., Bedbeder,
P., Van Regemorter, N., Weissenbach, J., and Petit, C.
(1997) A human homologue of the Drosophila eyes absent gene
underlies branchio-oto-renal (BOR) syndrome and identifies a novel gene
family, Nat. Genet., 15, 157-164.
9.Azuma, N., Hirakiyama, A., Inoue, T., Asaka, A.,
and Yamada, M. (2000) Mutations of a human homologue of the
Drosophila eyes absent gene (EYA1) detected in patients
with congenital cataracts and ocular anterior segment anomalies,
Hum. Mol. Genet., 9, 363-366.
10.Pandey, R. N., Rani, R., Yeo, E. J., Spencer, M.,
Hu, S., Lang, R. A., and Heqde, R. S. (2010) The Eyes absent
phosphatase-transactivator proteins promote proliferation,
transformation, migration, and invasion of tumor cells,
Oncogene, 29, 3715-3722.
11.Robin, T. P., Smith, A., McKinsey, E., Reaves,
L., Jedlicka, P., and Ford, H. L. (2012) EWS/FLI1 regulates EYA3 in
Ewing sarcoma via modulation of miRNA-708, resulting in increased cell
survival and chemoresistance, Mol. Cancer Res., 10,
1098-1108.
12.Furuya, M., Qadota, H., Chisholm, A. D., and
Sugimoto, A. (2005) The C. elegans eyes absent ortholog EYA-1 is
required for tissue differentiation and plays partially redundant roles
with PAX-6, Dev. Biol., 286, 452-463.
13.Hirose, T., Galvin, B. D., and Horvitz, H. R.
(2010) Six and Eya promote apoptosis through direct transcriptional
activation of the proapoptotic BH3-only gene egl-1 in
Caenorhabditis elegans, Proc. Natl. Acad. Sci.
USA, 107, 15479-15484.
14.Hsu, A. L., Murphy, C. T., and Kenyon, C. (2003)
Regulation of aging and age-related disease by DAF-16 and heat-shock
factor, Science, 300, 1142-1145.
15.Lithgow, G. J., White, T. M., Melov, S., and
Johnson, T. E. (1995) Thermotolerance and extended life-span conferred
by single-gene mutations and induced by thermal stress, Proc. Natl.
Acad. Sci. USA, 92, 7540-7544.
16.Gerstbrein, B., Stamatas, G., Kollias, N., and
Driscoll, M. (2005) In vivo spectrofluorimetry reveals
endogenous biomarkers that report healthspan and dietary restriction in
Caenorhabditis elegans, Aging Cell, 4,
127-137.
17.Kenyon, C. J. (2010) The genetics of ageing,
Nature, 464, 504-512.
18.Murphy, C. T., McCarroll, S. A., Bargmann, C. I.,
Fraser, A., Kamath, R. S., Ahringer, J., Li, H., and Kenyon, C. (2003)
Genes that act downstream of DAF-16 to influence the lifespan of
Caenorhabditis elegans, Nature, 424, 277-283.
19.Terman, A., and Brunk, U. T. (1998) Lipofuscin:
mechanisms of formation and increase with age, APMIS,
106, 265-276.
20.Borlongan, C. V., Kanning,
K., Poulos, S. G., Freeman, T. B., Cahill, D.
W., and Sanberg, P. R. (1996) Free radical damage and oxidative
stress in Huntington’s disease, J. Fla. Med. Assoc., 83,
335-341.
21.Charvin, D., Vanhoutte, P., Pages,
C., Borrelli, E., and Caboche, J. (2005) Unraveling a role
for dopamine in Huntington’s disease: the dual role of reactive
oxygen species and D2 receptor stimulation, Proc. Natl. Acad. Sci.
USA, 102, 12218-12223.
22.Shukla, V., Mishra, S. K., and Pant, H. C. (2011)
Oxidative stress in neurodegeneration, Adv. Pharmacol. Sci.,
2011, 572634.
23.Murakami, K., Murata, N., Noda, Y., Tahara, S.,
Kaneko, T., Kinoshita, N., Hatsuta, H., Murayama,
S., Barnham, K. J., Irie, K., Shirasawa, T., and
Shimizu, T. (2011) SOD1 (copper/zinc superoxide dismutase) deficiency
drives amyloid β protein oligomerization and memory loss in mouse
model of Alzheimer disease, J. Biol. Chem., 286,
44557-44568.
24.Henderson, S. T., and Johnson, T. E. (2001)
daf-16 integrates developmental and environmental inputs to mediate
aging in the nematode Caenorhabditis elegans, Curr.
Biol., 11, 1975-1980.