Inhibition of Oxidative Hemolysis in Erythrocytes by Mitochondria-Targeted Antioxidants of SkQ Series
E. O. Omarova* and Y. N. Antonenko
Lomonosov Moscow State University, Belozersky Institute of Physico-Chemical Biology, Leninsky Gory 1/40, 119991 Moscow, Russia; fax: +7 (495) 939-3181; E-mail: omarova@belozersky.msu.ru* To whom correspondence should be addressed.
Received October 28, 2013; Revision received November 12, 2013
In the present work we studied the effect of antioxidants of the SkQ1 family (10-(6′-plastoquinonyl)decyltriphenylphosphonium) on the oxidative hemolysis of erythrocytes induced by a lipophilic free radical initiator 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) and a water-soluble free radical initiator 2,2′-azobis(2-methylpropionamidine) dihydrochloride (AAPH). SkQ1 was found to protect erythrocytes from hemolysis, 2 μM being the optimal concentration. Both the oxidized and reduced SkQ1 forms exhibited protective properties. Both forms of SkQ1 also inhibited lipid peroxidation in erythrocytes induced by the lipophilic free radical initiator AMVN as detected by accumulation of malondialdehyde. However, in the case of induction of erythrocyte oxidation by AAPH, the accumulation of malondialdehyde was not inhibited by SkQ1. In the case of AAPH-induced hemolysis, the rhodamine-containing analog SkQR1 exerted a comparable protective effect at the concentration of 0.2 μM. At higher SkQ1 and SkQR1 concentrations, the protective effect was smaller, which was attributed to the ability of these compounds to facilitate hemolysis in the absence of oxidative stress. We found that plastoquinone in the oxidized form of SkQ1 could be reduced by erythrocytes, which apparently accounted for its protective action. Thus, the protective effect of SkQ in erythrocytes, which lack mitochondria, proceeded at concentrations that are two to three orders of magnitude higher than those that were active in isolated mitochondria.
KEY WORDS: antioxidants, oxidative stress, SkQ1, hemolysis, lipid peroxidation, erythrocytes, AAPH, AMVNDOI: 10.1134/S0006297914020072
Abbreviations: AAPH, 2,2′-azobis(2-methylpropionamidine) dihydrochloride; AMVN, 2,2′-azobis(2,4-dimethylvaleronitrile); LPO, lipid peroxidation; MDA, malondialdehyde; ROS, reactive oxygen species; SkQ1, 10-(6′-plastoquinonyl)decyltriphenylphosphonium; SkQR1, 10-(plastoquinonyl)decylrhodamine 19.
Under normal physiological conditions, reactive oxygen species (ROS)
formed in a cell are opposed by the cellular multicomponent antioxidant
enzyme system. Any distortion of the existing balance, e.g. by
exogenous ROS sources, results in oxidative stress in cells. Many
cellular components are ROS targets, i.e. nucleic acids, proteins, and
membrane lipids. Enhancement of lipid peroxidation (LPO) is accompanied
by changes in the structure and properties of biological membranes and
in cell functioning. Disruption of lipid homeostasis of mitochondrial
and cellular membranes contributes to the initiation of apoptosis or
necrosis of cells. Oxidative stress experienced by biomolecules,
membranes, and tissues results in pathological conditions in the
organism, which then induce many diseases (coronary heart disease,
stroke, Alzheimer’s and Parkinson’s diseases, etc.) [1-3] and aging [4].
Mitochondria-targeted antioxidants of the SkQ family, being accumulated in energized mitochondria, can protect animal cells from the effects of oxidative stress. These compounds have shown their high antioxidant activity in such systems as artificial lipid membranes (liposomes and planar bilayers), isolated mitochondria, cell cultures, and in vivo models [5-7]. This leads to antiapoptotic effect of these compounds that has been shown, for example, in human cells of HeLa culture, where SkQ1 showed a protective effect against ROS-induced apoptosis in nanomolar and picomolar concentrations [6].
Mammalian erythrocytes are a traditional model widely used to study the mechanisms and effects of oxidative stress, as well as the protective effects of new antioxidants [8]. This model and artificial model systems (liposomes, planar bilayer membranes) used in studying oxidative membrane damages have one common feature: they have no membrane systems other than the plasma membrane [8-10]. However, unlike liposomes, the erythrocyte plasma membrane consists of natural lipids, and these lipids are in contact with the cytoplasm containing most of the enzymes of the cellular antioxidant protection system, in particular, such proteins as glutathione peroxidase, catalase, superoxide dismutase, etc. Studies on the antioxidant effects of any compound include not only the analysis of its ability to inhibit LPO accumulation in membranes of erythrocytes, but also such parameter as protection against hemolysis caused by oxidative stress [8]. In this work, we studied the antioxidant effects of SkQ1 and its analogs in human erythrocytes exposed to oxidative stress, including their protective effect against oxidative hemolysis of the cells.
MATERIALS AND METHODS
Mitochondria-targeted antioxidants of the SkQ1 family were synthesized in our institute by Korshunova and Sumbatyan as described in [6]. The reduced form SkQ1red was prepared before the experiment by adding an alcoholic solution of sodium borohydride (NaBH4) to the oxidized form SkQ1ox. Other reagents were from Sigma-Aldrich (USA).
Blood samples from a healthy donor were collected in phosphate-buffered saline (PBS) (pH 7.4) containing EDTA (1 mM), and the erythrocytes were then washed twice in PBS without EDTA. The resulting erythrocytes were resuspended in PBS so that hematocrit values would reach 5-10%.
The lipophilic free radical initiator 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) was used as an oxidative stress inducer to initiate hemolysis. Hemolysis was induced by introduction of 4 mM AMVN into 2.5% (hematocrit) erythrocyte suspension with subsequent incubation at 37ºC with occasional stirring. Hemolysis was registered by hemoglobin release, which was measured after centrifugation at 3000 rpm (5 min) by the hemoglobin absorption at 540 nm in the supernatant using an Ultrospec 1100 spectrophotometer (Amersham). Complete hemolysis was caused by the addition of 1% Triton. The experiments were repeated three times.
The water-soluble free radical initiator 2,2′-azobis(2-methylpropionamidine) dihydrochloride (AAPH) was also used as an oxidative stress inducer to initiate hemolysis. Hemolysis was induced by introduction of 40 mM AAPH to 0.1% (hematocrit) erythrocyte suspension with subsequent incubation at 37ºC under constant stirring. Hemolysis was detected by the reduction in light absorption by the erythrocyte suspension at 650 nm; it was registered using the Ultrospec 1100 spectrophotometer. Hemolysis rate was characterized by hemolysis half-time – the time at which the absorption value reached half value. Hemolysis experiments were repeated 4-6 times to obtain statistically reliable results.
Lipid peroxidation (LPO) induced by the free radical initiators AMVN or AAPH was assessed by the level of malondialdehyde (MDA) accumulation, which was recorded by the fluorescence of the colored adduct with thiobarbituric acid according to [11].
A 5-10% suspension of human erythrocytes in PBS buffer (pH 7.4) was incubated under oxidizing conditions (4 mM AMVN or 80 mM AAPH) in the presence and absence of antioxidants at 37ºC for 2.5 h with periodical shaking. After the addition of trichloroacetic acid (3.4%), the suspension was thoroughly dispersed on a vortex mixer, and the precipitate was separated by centrifugation for 10 min at 10,000 rpm. Then thiobarbituric acid (0.35%) and HCl (0.07 M) were successively added. The mixture was boiled in a water bath for 30 min. Fluorescence was measured at 550 nm (excitation 515 nm) using a Panorama fluorat-02 fluorometer (Lumex, Russia). The reaction with 1,1,3,3-tetraethoxypropane was used to calibrate the method. The MDA content was normalized for the amount of hemoglobin that was determined spectrophotometrically at 540 nm using the extinction coefficient 13.8 mM-1·cm-1. The experiments were repeated four times.
Reduction of the oxidized form of SkQ1 (absorption maximum at 274 nm) in erythrocytes in PBS and the appearance of the reduced form of SkQ1 (absorption at 290 nm) were recorded spectrophotometrically [6] on the Ultrospec 1100 spectrophotometer. The oxidized form of SkQ1 (2 μM) was incubated with erythrocyte suspension for 5 min, and the precipitate was collected by centrifugation. Hydrogen peroxide (0.006%) was used for further oxidation of the reduced form of SkQ1. The experiments were repeated three times.
RESULTS
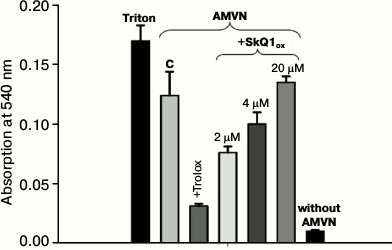
Oxidative hemolysis of erythrocytes induced by AMVN. Incubation of erythrocytes with the lipophilic free radical initiator AMVN (4 mM) for 2.5 h causes considerable hemolysis (control column C, Fig. 1). Hemolysis of erythrocytes in the supernatant was recorded spectrophotometrically by absorbance of hemoglobin at 540 nm. Figure 1 shows that the addition of 2 μM SkQ1ox (SkQ1 oxidized form) to the erythrocyte suspension had a protective effect. It will be shown below that the reduction of the protective effect of SkQ1 at concentrations exceeding 2 μM (4 and 20 μM) is due to its ability at high concentrations to cause hemolysis of erythrocytes in the absence of oxidative stress. Addition of 3 mM Trolox, a classic antioxidant, protected against oxidative hemolysis.
Fig. 1. Protective effects of SkQ1 and Trolox against oxidative hemolysis of erythrocytes induced by the lipophilic free radical initiator AMVN. Hemolysis was recorded after erythrocyte precipitation by the absorption of hemoglobin at 540 nm in the presence of AMVN (4 mM) (control) or in the presence of pairs: AMVN and oxidized form SkQ1ox (2, 4, and 20 μM); AMVN and Trolox (3 mM). Complete hemolysis was caused by treatment with 1% Triton. Each column in the figure represents the average value of three experiments and shows the standard error of the mean.
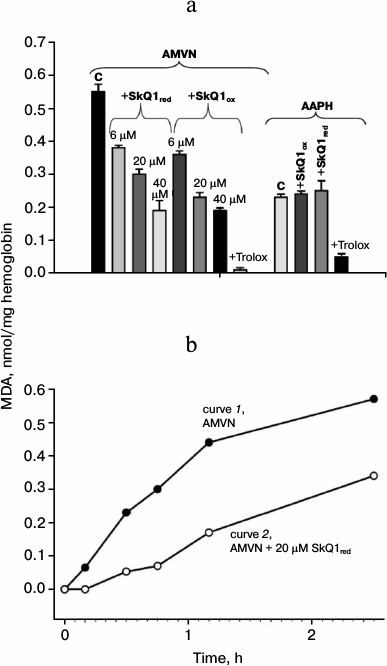
Antioxidant effect of SkQ1 on erythrocytes. LPO level was assessed by the accumulation of MDA 2.5 h after the addition of the lipophilic free radical initiator AMVN or the water-soluble free radical initiator AAPH to obtain significant MDA accumulation. Figure 2a shows that in the case of AMVN induction in the presence of either oxidized or reduced SkQ1, the accumulation of MDA was reduced. Statistical processing of the results indicated with confidence interval p > 0.999 that SkQ1 protects erythrocyte lipids from LPO in this system regardless of its initial reduction level. The efficacy of the protective effect of the oxidized form SkQ1ox compared to the reduced form SkQ1red at concentrations 6 and 40 μM did not differ with confidence probability p > 0.95. High concentrations of SkQ are known to have a prooxidant effect [6]. For this reason, we carried out experiments on the effect of 50 μM SkQ1ox (in the absence of free radical initiators), and we found that SkQ1 caused no accumulation of MDA (data not shown). In the case of AAPH induction, the MDA level in the presence of either the oxidized or reduced forms of SkQ1 was practically the same (Fig. 2a).
Fig. 2. Protective effect of SkQ1 and Trolox against peroxidation of erythrocyte lipids caused by the lipophilic free radical initiator AMVN or the hydrophilic free radical initiator AAPH, as determined by the content of malondialdehyde colored adduct. Each column on the figure represents the average value of three experiments and shows the standard error of the mean. a) Content of malondialdehyde colored adduct formed after incubation with AMVN (4 mM) or in the presence of pairs: AMVN + SkQ1red (6, 20, and 40 μM); AMVN + SkQ1ox (6, 20, and 40 μM); AMVN + Trolox (3 mM). The content of malondialdehyde colored adduct formed after incubation with AAPH (80 mM) and in the presence of pairs: AAPH + SkQ1ox (50 μM); AAPH + SkQ1red (50 μM); AAPH + Trolox (3 mM). b) Kinetics of accumulation of malondialdehyde colored adduct formed after incubation with 4 mM AMVN (curve 1) or in the presence of the pair AMVN + 20 μM SkQ1red (curve 2).
We also found that the well-known antioxidant Trolox at 3 mM concentration almost completely inhibited the formation of MDA products (Fig. 2a). The concentration of 300 μM and less had no inhibitory effect on MDA product accumulation in the case of initiation by AAPH (data not shown).
Figure 2b shows that the addition of the lipophilic free radical initiator AMVN caused a steady increase in MDA product accumulation for 2.5 h (curve 1). In the presence of 20 μM SkQ1red, the MDA content was reduced during the entire experiment (curve 2), and at early times the decrease was more pronounced.
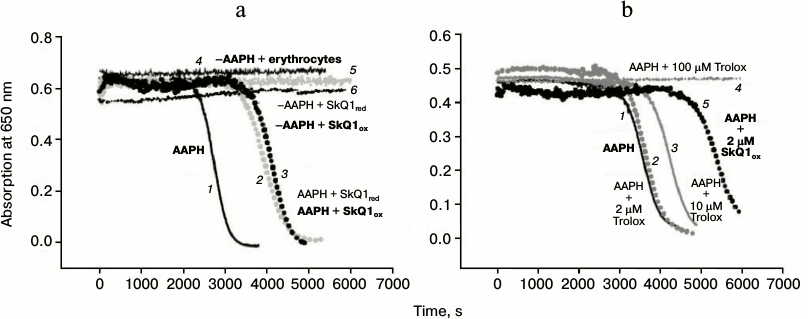
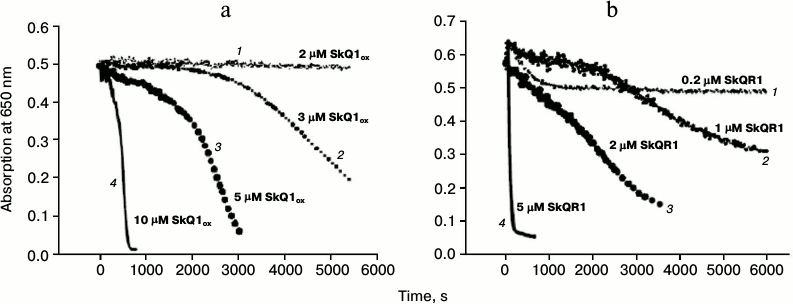
Oxidative hemolysis of erythrocytes induced by AAPH. Incubation of erythrocytes with the water-soluble free radical initiator AAPH (40 mM) caused their hemolysis on the time scale of hours (black solid curve 1, Fig. 3a). Hemolysis was detected by the reduction of the absorption of the erythrocyte suspension at 650 nm (i.e. at a wavelength where hemoglobin has practically no absorbance). The hemolysis curve has two phases: 1) a lag-phase with practically no change in absorption (to 2300 s on the solid curve, Fig. 3a); 2) phase of rapid decrease in absorption, which reflects the progress of changes in cell shape (after 2300 s, Fig. 3a). No hemolysis was observed in the absence of the free radical initiator (upper curve 4, Fig. 3a). Figure 3a shows that the addition of 1 μM SkQ1red to the erythrocyte suspension increased the time of the beginning of the second stage, i.e. it had a protective effect (gray dashed curve 2, Fig. 3a). The efficacy of the protective effect of oxidized form SkQ1ox (black dashed curve 3, Fig. 3a) was practically the same. In the absence of AAPH, this SkQ1 concentration had no hemolytic effect in either the oxidized form (black dotted curve 6, Fig. 3a) or in the reduced form (gray dotted curve 5, Fig. 3a).
Fig. 3. Protective effects of SkQ1 (a) and Trolox (b) against oxidative hemolysis of erythrocytes induced by water-soluble free radical initiator AAPH. The figure shows the results of a typical experiment. a) Absorption of erythrocyte suspension in the presence of 40 mM AAPH (black solid curve 1) and in the presence of pairs: AAPH + 1 μM SkQ1red (gray dashed curve 2); AAPH + 1 μM SkQ1ox (black dashed curve 3). Control in the absence AAPH: erythrocytes without SkQ1 (upper curve 4); erythrocytes in the presence of 1 μM SkQ1red (gray dotted curve 5); erythrocytes in the presence of 1 μM SkQ1ox (black dotted curve 6). b) Absorption of erythrocyte suspension in the presence of 40 mM AAPH (black solid curve 1) and in the presence of pairs: AAPH + Trolox (2 μM – gray dashed curve 2, 10 μM – gray solid curve 3, 100 μM – gray dotted curve 4); AAPH in the presence of 2 μM SkQ1ox (black dashed curve 5).
In accordance with data found in the literature, addition of 100 μM Trolox, the classic antioxidant, protected against hemolysis (gray dotted curve 4, Fig. 3b). Trolox at 2 μM concentration had no protective effect on this time scale (gray dashed curve 2, Fig. 3b), and 10 μM Trolox had a much weaker protective effect (gray solid curve 3, Fig. 3b) than 2 μM SkQ1 (black dashed curve 5, Fig. 3b).
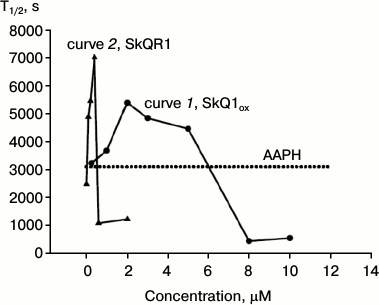
As customary in the literature [12], the hemolysis process can be quantitatively characterized by the time corresponding to 50% reduction in the erythrocyte suspension absorption, i.e. hemolysis half-time T1/2. Curve 1 on Fig. 4 shows the dependence of T1/2 on SkQ1 concentration. The figure shows the effect of the oxidized form; the effect of the reduced form practically coincided. The oxidized form SkQ1ox shows the maximal protective effect at 2 μM concentration (curve 1, Fig. 4). The dotted line on Fig. 4 shows the level of T1/2 without added SkQ (in the presence of AAPH). Statistical processing of the results indicated with confidence interval p > 0.999 that 2 μM SkQ1 increases the hemolysis half-time, the average T1/2 increase being 40%. It will be shown below that the decrease in the protective effect of SkQ1 at concentrations above 2 μM is due to its ability at high concentrations to cause hemolysis of erythrocytes in the absence of oxidative stress.
Fig. 4. Dependence of erythrocyte hemolysis half-time on concentrations of SkQ1ox (curve 1) and rhodamine analog SkQR1 (curve 2) in the presence of 40 mM AAPH. The dotted line shows the level of T1/2 without added SkQ1 (in the presence of AAPH). The figure shows the results of a typical experiment.
Curve 2 of Fig. 4 shows the dependence of T1/2 on the concentration of the rhodamine analog SkQR1, where the triphenylphosphonium group is replaced by rhodamine 19. It is evident that SkQR1 also increases T1/2, but at significantly lower concentrations. The protective effect of SkQR1 was maximal at 0.4 μM concentration: the hemolysis half-time increased more than twofold (curve 2, Fig. 4). Already 0.2 μM SkQR1 (curve 2, Fig. 4) had the same protective effect as 2 μM SkQ1ox (curve 1, Fig. 4). Statistical analysis of the results showed that with confidence interval p > 0.99, 0.2 μM SkQR1 increases hemolysis half-time; the average increase in T1/2 was also 40%.
Effects of SkQ1 and its analogs on erythrocyte hemolysis in the absence of free radical initiator. We stated above that high concentrations of SkQ1 and its rhodamine analog showed hemolytic effects. Figure 5a shows that 10 μM SkQ1 caused hemolysis in the absence of free radical initiator on the time scale of tens of minutes (solid curve 4, Fig. 5a). SkQR1 already exhibited its strong hemolytic action at a concentration of 5 μM (solid curve 4, Fig. 5b). Figure 5 also shows hemolysis curves for different SkQ1 and SkQR1 concentrations. It can be seen that SkQR1 had hemolytic effect at much lower concentrations than SkQ1. It should be noted that Trolox had only minimal protective effect in the case of SkQ1-induced hemolysis (data not shown).
Fig. 5. Erythrocyte hemolysis caused by SkQ1 (a) and SkQR1 (b) in the absence of free radical initiator AAPH. The figure shows the results of a typical experiment. a) 2 μM SkQ1ox (dotted curve 1); 3 μM SkQ1ox (medium-dashed curve 2); 5 μM SkQ1ox (dashed curve 3); 10 μM SkQ1ox (solid curve 4). b) 0.2 μM SkQR1 (dotted curve 1); 1 μM SkQR1 (medium-dashed curve 2); 2 μM SkQR1 (dashed curve 3); 5 μM SkQR1 (solid curve 4).
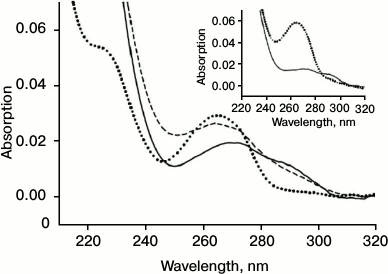
Reduction of oxidized SkQ1 in erythrocytes. Figure 6 shows the spectrum of the oxidized form SkQ1ox (dotted curve, Fig. 6). Incubation of SkQ1ox with erythrocytes resulted in the reduction of the absorption maximum at 274 nm and the appearance of a “shoulder” near 290 nm (solid curve, Fig. 6), indicating the formation of the reduced form SkQ1red. Subsequent oxidation SkQ1red by hydrogen peroxide again resulted in the appearance of the absorption maximum at 274 nm and the reduction of the “shoulder” at 290 nm (dashed curve, Fig. 6). It should be noted that incubation with erythrocytes causes the appearance of light scattering in the spectrum, which is apparently due to incomplete washing of erythrocytes and their fragments in the course of centrifugation. The inset in Fig. 6 shows the control spectra of SkQ1ox (dotted curve) and SkQ1red reduced by sodium borohydride (solid curve).
Fig. 6. Absorption spectrum SkQ1ox (2 μM) (dotted curve), the appearance of SkQ1red after incubation with erythrocytes (solid curve), and subsequent oxidation of SkQ1red by hydrogen peroxide (0.006%) (dashed curve). Inset: absorption spectrum of SkQ1ox (dotted curve) and spectrum of SkQ1red after reduction by sodium borohydride (solid curve).
DISCUSSION
Plastoquinone, being a part of both SkQ1 and SkQR1, is a classic antioxidant that effectively breaks the chain of peroxidation due to the reduction of lipid radical LO2· to hydroperoxide LOOH in the reaction:
LO2· + QH2 → LOOH + QH·,
which competes with the reaction of chain propagation [13]:
LO2· + LH → LOOH + L·.
Only the reduced quinone form (quinol) takes part in this reaction, while the oxidized form has no antioxidant effect. We have shown that the plastoquinone moiety of SkQ1ox can be reduced by erythrocytes (Fig. 6). Thus, the observed antioxidant effect of SkQ1ox (Fig. 2a) as well as its anti-hemolytic effect (Figs. 1 and 3) should be attributed to the process of plastoquinone reduction in erythrocytes. This process can result both from the interaction with the enzyme systems present in erythrocytes (such as NADPH-oxidase) and the interaction with the reduced form of glutathione present in erythrocytes in millimolar concentrations.
We have shown that SkQ1 in a certain range of concentrations has a protective effect in AMVN-induced hemolysis (Fig. 1), protecting erythrocyte membrane lipids from peroxidation induced by the lipophilic free radical initiator AMVN (Fig. 2a). SkQ1 also had a protective effect in hemolysis induced by the water-soluble free radical initiator AAPH (Fig. 4), but it had no antioxidant effect when the process was assessed by lipid peroxidation (Fig. 2a). This may be due to the fact that not only the membrane lipids, but also many other erythrocyte components are targets of water-soluble oxidation inducer AAPH: the level of reduced glutathione decreases, hemoglobin is oxidized, and membrane proteins are degraded [9]. Moreover, some authors believe that each of these events alone cannot cause AAPH-mediated hemolysis [11, 16], indicating the complex character of the oxidative effect of AAPH on the cells. In contrast to AAPH, the free radical initiator AMVN, due to its lipophilicity, exhibits substantially different effects on cells [8, 17]: the efficiency of their impacts on membrane components is very different [4, 18]. Efficiency of antioxidant protective effects is determined both by the properties of the oxidative stress inducers and by the properties of the antioxidants per se. Hydrophilic ascorbic acid efficiently extinguishes radicals in an aqueous medium prior to their reaching the membrane. This results in the dose-dependent inhibition of hemolysis [19]. However, due to the limited ability to extinguish radicals in lipid membranes, it does not suppress the oxidation of polyunsaturated fatty acids of micelles, phospholipid liposomal membranes, and lipoproteins in the case of oxidation caused by lipophilic radicals formed directly in lipids [19]. Hydrophilic ascorbic acid and lipophilic tocopherol were shown to have a synergistic effect: oxidation was suppressed for a longer time than in the presence of only tocopherol, and the consumption rate of the latter was significantly decreased in the presence of ascorbic acid. Lipid-soluble quercetin was more effective in protecting against AAPH-induced hemolysis than water-soluble dihydroquercetin [21]. Properties of lipophilic antioxidants determine their preferential localization in the surface- or deeper layers of lipid membranes of cells [4]. The lipophilic character of SkQ1, which has a high partition coefficient between lipid and water (about 13,000 : 1) [6], is likely to determine its more efficient antioxidant effect in the case of the more lipophilic free radical initiator AMVN.
Hemolytic effects of SkQ1 and SkQR1 are probably related to the ability of these compounds, when in high concentrations, to disrupt the barrier properties of lipid membranes, i.e. to have detergent-like effects [14]. Indeed, these compounds are long molecules; on one hand, they have a positive charge, which makes them hydrophilic, and on the other hand, they have rather long lipophilic segments. As a result, when in micromolar concentrations, they form micelles and have the ability to induce lipid membrane permeability [14]. However, it had been shown earlier on the system of isolated mitochondria that high SkQ1 concentrations had prooxidant effect [6]. This property can also contribute to the induction of erythrocyte hemolysis at high concentrations of SkQ1 and SkQR1. It was shown that 40 μM and higher concentrations of SkQ1 caused swelling of isolated mitochondria; the authors attributed this phenomenon to its prooxidant and detergent activity [15]. We found that in the absence of free radical initiator, 50 μM SkQ1 caused no significant MDA accumulation (data not shown), and Trolox had only minimal protective effect in the case of SkQ1-induced hemolysis (data not shown), so the hemolytic effect of high concentrations of SkQ1 should be attributed to its detergent-like action.
It should be noted that SkQ1 was effective at several micromolar concentrations in the case of its antioxidant and anti-hemolytic effects on erythrocytes (Figs. 1, 2, and 4). Similar concentrations had a considerable protective effect on artificial lipid membranes [7]. However, these concentrations were approximately two to three orders of magnitude higher than those that were active in the system of isolated mitochondria and in protecting from peroxide-induced apoptosis [6]. This difference emphasizes the role of SkQ1 and SkQR1 accumulation in mitochondria, which results from electrophoretic transport of SkQ1 and SkQR1 across the inner mitochondrial membrane, which has high electric potential (about –180 mV). The potential across the erythrocyte membrane is very low; it is only about –10 ± 3 mV [22, 23].
We have shown that the protective SkQR1 concentrations in the system of erythrocyte hemolysis were about 10 times lower than those of SkQ1. This corresponds to the observation that in human fibroblast cells the protective effect of SkQR1 was higher than that of SkQ1 [6]. In the system of artificial lipid membranes, the antioxidant effect of quinone-containing compounds hardly depends on the other substituent [13], i.e. the antioxidant effects of SkQR1 and SkQ1 should be similar. This indicates that the difference in their protective effects on erythrocytes can be related to the better penetrating ability of SkQR1 or its greater ability to interact with intracellular enzymes. It has been shown previously that SkQR1 had about 10 times higher permeability across the lipid membranes than SkQ1 [14].
The authors would like to express their gratitude to Professor V. P. Skulachev for his help and fruitful discussion of the results.
This work was financially supported by the Moscow State University Research Institute of Mitoengineering and the Russian Foundation for Basic Research (grant 12-04-00199).
REFERENCES
1.Aruoma, O. I. (1998) J. Am. Oil Chem. Soc.,
75, 199-212.
2.Vincent, A. M., Russell, J. W., Low, P., and
Feldman, E. L. (2004) Endocr. Rev., 25, 612-628.
3.Chirico, E. N., and Pialoux, V. (2012) IUBMB.
Life, 64, 72-80.
4.Niki, E. (2010) Free Radic. Biol. Med.,
49, 503-515.
5.Skulachev, V. P. (2007) Biochemistry
(Moscow), 72, 1385-1396.
6.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E.,
Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Yu.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Yu., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vysokikh, M.
Yu., Yaguzhinsky, L. S., Zamyatnin, A. A., and Skulachev, V. P. (2008)
Biochemistry (Moscow), 73, 1273-1287.
7.Antonenko, Y. N., Roginsky, V. A., Pashkovskaya, A.
A., Rokitskaya, T. I., Kotova, E. A., Zaspa, A. A., Chernyak, B. V.,
and Skulachev, V. P. (2008) J. Membr. Biol., 222,
141-149.
8.Niki, E., Komuro, E., Takahashi, M., Urano, S.,
Ito, E., and Terao, K. (1988) J. Biol. Chem., 263,
19809-19814.
9.Zou, C. G., Agar, N. S., and Jones, G. L. (2001)
Life Sci., 69, 75-86.
10.Shiva Shankar Reddy, C. S., Subramanyam, M. V.,
Vani, R., and Asha, D. S. (2007) Toxicol. in vitro, 21,
1355-1364.
11.Sato, Y., Kamo, S., Takahashi, T., and Suzuki, Y.
(1995) Biochemistry, 34, 8940-8949.
12.Kinosita, K. Jr., and Tsong, T. T. (1977)
Proc. Natl. Acad. Sci. USA, 74, 1923-1927.
13.Roginsky, V., Barsukova, T., Loshadkin, D., and
Pliss, E. (2003) Chem. Phys. Lipids, 125, 49-58.
14.Rokitskaya, T. I., Klishin, S. S., Severina, I.
I., Skulachev, V. P., and Antonenko, Y. N. (2008) J. Membr.
Biol., 224, 9-19.
15.Sukhanova, E. I., Trendeleva, T. A., and
Zvyagilskaya, R. A. (2010) Biochemistry (Moscow), 75,
139-144.
16.Sandhu, I. S., Ware, K., and Grisham, M. B.
(1992) Free Radic. Res. Commun., 16, 111-122.
17.Krainev, A. G., and Bigelow, D. G. (1996) J.
Chem. Soc. Perkin Trans., 2, 747-754.
18.Dean, R. T., Hunt, J. V., Grant, A. J., Yamamoto,
Y., and Niki, E. (1991) Free Radic. Biol. Med., 11,
161-168.
19.Niki, E. (1991) Am. J. Clin. Nutr.,
54, 1119S-1124S.
20.Konovalova, G. G., Lankin, V. Z., Tikhaze, A. K.,
Nezhdanova, I. B., Lisina, M. O., and Kukharchuk, V. V. (2003) Byul.
Eksp. Biol. Med., 136, 142-144.
21.Chen, Y., and Deuster, P. (2009) Chem. Biol.
Interact., 182, 7-12.
22.Mikkelsen, R. B., Tanabe, K., and Wallach, D. F.
(1982) J. Cell Biol., 93, 685-689.
23.Gedde, M. M., and Huestis, W. H. (1997)
Biophys. J., 72, 1220-1233.