Basolateral Expression of GRP94 in Parietal Cells of Gastric Mucosa
R. M. Arin1*, Y. Rueda1, O. Casis2, M. Gallego2, A. I. Vallejo1, and B. Ochoa1
1Department of Physiology, Faculty of Medicine and Dentistry, University of the Basque Country UPV/EHU, Barrio Sarriena s/n, 48940 Leioa, Spain; fax: +34-9460-15662; E-mail: rosamaria.arin@ehu.es; yuri.rueda@ehu.es; anaivallejo@gmail.com; begona.ochoa@ehu.es2Department of Physiology, Faculty of Pharmacy, University of the Basque Country UPV/EHU, Paseo de la Universidad 7, 01006 Vitoria-Gasteiz, Spain; fax: +34-9460-15662; E-mail: oscar.casis@ehu.es; monica.gallego@ehu.es
* To whom correspondence should be addressed.
Received June 3, 2013; Revision received July 8, 2013
GRP94 is a member of the heat shock protein family normally confined to the endoplasmic reticulum that sometimes escapes the KDEL-mediated retention system. It is overexpressed in some gastric and other gastrointestinal carcinomas, but little is known about the physiological role of GRP94 in gastric mucosa. We investigated the membrane presence of GRP94 in parietal cells, which secrete acid into the gastric lumen, using subcellular fractionation, selective solubilization of membrane proteins, Western blotting, and radio-ligand binding and provided evidence of functional GRP94 expression at the surface of gastric mucosa parietal cells anchored to the basolateral domain. Our results show that GRP94 is not an integral membrane protein since 50 mM Na2CO3 treatment dissociates part of it from the membrane. However, 100 mM Na2CO3 treatment did not extract all GRP94 from the membrane, which indicates that it is strongly associated with it. The presence of GRP94 in isolated plasma membrane was demonstrated by Western blotting and its functionality by radio-ligand binding experiments. Both the KD value obtained in saturation experiments with N-ethylcarboxamido-[3H]adenosine at 4°C, at the nanomolar range, and the inhibition constant of its binding by radicicol, the most specific GRP94 inhibitor, indicate that active receptor regions are exposed at the membrane surface. Western blotting of plasma membrane subfractions showed that GRP94 is mainly expressed in the basolateral membrane of gastric parietal cells, while its presence in the apical domain is negligible, thereby inferring a role for GRP94 in processes operating in this membrane domain.
KEY WORDS: gastric mucosa parietal cell, basolateral polarization, molecular chaperone, adenosine receptorDOI: 10.1134/S0006297914010027
Abbreviations: ER, endoplasmic reticulum; GRP94, glucose-regulated protein of 94 kDa; Hsp, heat shock protein; NECA, N-ethylcarboxamidoadenosine.
Parietal cells are responsible for the production and secretion of
gastric acid and, in species like humans or rabbits, also of
Castle’s intrinsic factor. Parietal cells undergo dramatic
structural changes in the transition from resting to secreting state.
In the secreting state they acquire a broad variety of functionally
coupled apical and basolateral ion transport proteins. Among others,
they are equipped with basolateral transporters for K+ and
Cl– uptake like
Na+-K+-Cl– cotransporter,
Cl–/HCO3– exchangers, and
Na+/K+-ATPase [1]. The
secretory activity of parietal cells is controlled by neural stimuli or
by several endocrine and paracrine agents acting on membrane receptors
of the parietal cells or through other cells located close to them in
gastric glands.
Glucose-regulated protein of 94 kDa (GRP94, also called gp96 and endoplasmin) is a member of the heat shock protein (Hsp) family consisting of chaperones regulated by ATP [2]. It is one of the most abundant proteins in the mammalian endoplasmic reticulum (ER) lumen and the paralog of cytosolic Hsp90 in that organelle [3]. In recent years, GRP94 has been detected on the surface of several cell types and also identified as a secretory protein. It is present on the hepatocyte surface and in bile canaliculi [4], on the surface of antigen presenting cells during immune response [5], on differentiating skeletal muscle myocytes [6], and in several cell lines [7], among others. Because of its sequence, GRP94 cannot be an integral membrane protein, and as a consequence its ectopic presence is presumably mediated by association with other membrane or secretory proteins [8]. For instance, GRP94 has been observed to be associated with bile salt-dependent lipase, also known as pancreatic cholesterol esterase. In the GRP94/bile salt-dependent lipase complex, GRP94 lacks a KDEL motif that is necessary for the retention of soluble proteins in rough ER, and the complex does not dissociate in ER and is instead secreted in pancreatic juice [9, 10].
Several biological functions have been recognized for GRP94. One of the better understood functions is its role in the folding and assembly of membrane proteins of the secretory pathway. The molecular chaperone activity of GRP94 is selective, with targets including integrins, toll-like receptors, insulin-like growth factors, thyroglobulin, the platelet glycoprotein Ib-IX-V complex, and others [11-13]. In the absence of GRP94, these proteins are targeted for ER-associated degradation. Interestingly, GRP94 also has a role in cell survival and defense as it is required for the cell surface export of cell receptors involved in the native immune response [6], and it is a regulator of antigen presenting cells during the immune response [5] and of calcium homeostasis [14]. Moreover, GRP94 is overexpressed in several cell types as a defense mechanism in situations in which a rapid elevation of cell protection is needed, e.g. in hypoxia [15].
Despite such a multiplicity of functions, the physiological role of GRP94 in the various cell types that form the gastric mucosa remains to be determined. Different research groups have detected an elevation of the GRP94 precursor levels in the gastric mucosa of human patients infected with Helicobacter pylori [16, 17]; it has been proposed that this elevation represents a cellular defense mechanism against oxidative stress. GRP94 induces the production of proinflammatory cytokines such as tumor necrosis factor α and interleukin-8 in human gastric mucosa macrophages [17]. In addition, GRP94 has been found overexpressed in gastric [18] and other digestive tract [19-21] carcinomas.
Based on preliminary results, we suspected that there was a binding site for N-ethylcarboxamidoadenosine (NECA) in the plasma membrane of gastric parietal cells in rabbits, which, however, did not show the expected behavior on cAMP homeostasis [22]. The substituted adenosine derivative NECA is a non-selective agonist for the adenosine receptor subtypes [23]. For years it was speculated that a binding site for NECA did not fulfill all the characteristics of a true adenosine receptor; this was called adenotin. The KD of the binding of this protein to NECA was in the nanomolar range, and it was later concluded that this high-affinity NECA-binding site corresponded to GRP94 [24]. Our work focused on investigating the presence of GRP94 in the plasma membrane of parietal cells from rabbit gastric mucosa. For our study, we performed density-gradient fractionation of cells, selective extraction of membrane proteins, Western blotting, and saturation assays with [3H]NECA and competitive binding assays with NECA and radicicol (which potently inhibits GRP94 from binding NECA) as competitors. We provide for the first time evidence of functional GRP94 expression in parietal cells of gastric mucosa, anchored to the basolateral plasma membrane domain.
MATERIALS AND METHODS
Isolation of parietal cells from rabbit gastric mucosa. Male and female New Zealand rabbits weighing 2.5-4 kg were used. Housing and experimental procedures were in accordance with the Spanish (RD 1201/2005) and European (2003/65/CE Directive and 2007/526/CE Recommendation) guidelines for the use of laboratory animals.
The rabbits were euthanized by the administration of an anesthetic preparation of pentobarbital. The gastric mucosa was isolated as described by Berglindh and Obrink [25]. Briefly, the stomach was perfused with phosphate buffered saline, and the mucosa was separated by blunt dissection. Gastric mucosa cells were isolated following the method described by Fryklund et al. [26] as modified by Ainz et al. [27]. Briefly, the mucosa was incubated with 15 U/ml pronase and 90 U/ml type I collagenase, and centrifugal elutriation was applied to obtain a fraction enriched with parietal cells, as described elsewhere (Sanders and Soll [28] modified by Ainz et al. [27]). The purity of the fraction was assessed by differential staining (Polysciences) and direct observation by phase-contrast optical microscopy. Cell viability was measured by the trypan blue exclusion test and averaged 90-95%. Cell functionality was monitored in every preparation by measuring the hydrochloric acid secretion in response to 10–3 to 10–7 M histamine stimulation as a surrogate index (data not shown).
Isolation of plasma membranes from parietal cells. We followed the method by Muallem et al. [29] with slight modifications. Parietal cell suspension was centrifuged (177g, 5 min, 24°C), and the pellet was resuspended in homogenization buffer (20 mM Tris-HCl, pH 7.4, 250 mM sucrose, 0.5 mM EDTA, 0.54 mM dithiothreitol (DTT), 5 µg/ml leupeptin, 15.7 µg/ml benzamidine) with a final concentration of 60⋅106 cells/ml, and homogenization was performed in a Potter–Elvehjem homogenizer (2000 rpm, 4°C). The homogenate was centrifuged (700g, 10 min, 4°C), and the supernatant was collected.
To obtain unfractioned plasma membranes, a 47% sucrose solution was topped by the 700g supernatant and ultracentrifuged in a swinging rotor (100,000g, 45 min, 4°C). Plasma membranes were collected from the interphase. Plasma membrane subfractions were isolated in a discontinuous sucrose gradient; a 47% sucrose phase was laid in a centrifugation tube, which was subsequently topped by a 40% sucrose phase and the 700g supernatant. Ultracentrifugation was performed in a swinging rotor (100,000g, 45 min, 4°C), and two plasma membrane fractions were obtained: apical membranes were aspirated from the supernatant/40% sucrose interphase and basolateral membranes from the interphase between the sucrose phases. After harvesting, fractions were resuspended in 50 mM Tris-HCl, pH 7.4, washed once (120,000g, 20 min, 4°C), incubated with 2 U/ml adenosine deaminase (1 h, room temperature) to metabolize endogenous adenosine, and washed again. The pellets were finally resuspended in 50 mM Tris-HCl, pH 7.4, and stored at –80°C until used.
Along this sequential procedure, we measured the enrichment of activity of the following marker proteins: 5′-nucleotidase activity was used as indicator of the recovery of plasma membrane [30], H+/K+-ATPase activity as apical membrane marker [31], Na+/K+-ATPase activity as basolateral membrane marker [32], and glucose 6-phosphatase activity as ER marker [30]. No enhancement of glucose 6-phosphatase specific activity was observed in any of the fractions, which indicated that the membrane fractions were enriched in plasma membrane and not in ER.
Protein concentration was measured following the colorimetric method by Bradford [33].
Measurement of radio-ligand binding to GRP94 in isolated cell membranes. Measurements were carried out as described by Casado et al. [34]. Membranes (0.5 mg protein/ml) in saturation assays were incubated (1 h, 4°C) with 20 to 2560 nM [3H]NECA (850 nCi/nmol) (Amersham, Sweden) in 50 mM Tris-HCl, pH 7.4, and free and bound ligands were quickly separated by vacuum filtration through Whatman GF/C filters previously washed for 2 h in 0.3% polyethyleneimine, pH 10. After filtration, the filters were bathed in 10 ml Tris-HCl, pH 7.4, and placed afterwards in 5 ml scintillation fluid Biogreen 3 (Scharlau) for radioactivity measurement. All binding assays were performed in triplicate. Nonspecific binding, measured in the presence of saturating 100 µM NECA (Sigma-Aldrich, USA), was subtracted from the total binding data to calculate the specific binding. Nonspecific binding represented ~5% of total binding.
In competitive binding assays, unlabeled NECA was used as competitor in some cases and radicicol (Sigma-Aldrich) in others. Radicicol is an antibiotic and is considered to be one of the most potent inhibitors of Hsp90s [35], among which is GRP94. Membranes were incubated with 200 nM [3H]NECA for 1 h, and afterwards 10–12 to 10–3 M NECA or 10–9 to 3⋅10–5 M radicicol were added as displacing agents.
Analysis of association of GRP94 with membranes. We analyzed the nature of association of GRP94 with isolated plasma membranes and, in addition, with microsomes isolated from rabbit parietal cells and from HepG2 human hepatoma cells as a control. For isolating microsomes, cells were resuspended in six volumes of homogenization buffer (20 mM Tris-HCl, pH 7.4, 250 mM sucrose, 0.5 mM EDTA, 0.54 mM DTT, 5 µg/ml leupeptin, 15.7 µg/ml benzamidine) and homogenized in a Potter–Elvehjem homogenizer (20 strokes, 1400 rpm, 4°C). After centrifugation (16,000g, 15 min, 4°C), the supernatant was collected and centrifuged again under the same conditions, and the upper 2/3 of the supernatant volume was collected to be subsequently centrifuged (105,000g, 1 h, 4°C). The resulting pellet contained the microsomes.
Proteins were selectively solubilized by treating membranes with either 0.3% 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonic acid (CHAPS), which completely solubilizes membrane proteins [36], 100 mM Na2CO3, pH 11.5, which solubilizes proteins with a strong association to the membrane, or 50 mM Na2CO3, pH 11.5, which releases peripheral proteins [37]. In all cases the plasma membranes or microsomes (2 mg protein) were resuspended in 500 µl of the solution of interest (50 or 100 mM Na2CO3, pH 11.5, or 0.3% CHAPS), incubated for 30 min at 4°C, and ultracentrifuged (230,000g, 1 h, 4°C). Supernatants containing solubilized products were collected, and pellets were resuspended in 400 µl of homogenization buffer. GRP94 was detected in the originating membranes, solubilized products, and pellets by Western blotting.
Analysis of GRP94 protein expression by Western blotting. Equal amounts of proteins (20 or 50 µg) were separated by 8% reducing SDS-PAGE [38] together with commercially available preparations of molecular weight standards (Bio-Rad, USA) and transferred to Immobilon P membranes (Millipore, USA). The membranes were blocked with 3% BSA and subsequently Western blotted. Chicken anti-rat GRP94 (1 : 1000 dilution, overnight, 4°C) and goat anti-chicken IgG-HRP (1 : 2000 dilution, 1 h, room temperature), both from Calbiochem (USA), were used as primary and secondary antibodies, respectively. Enhanced chemiluminescence reagent (Amersham) was used to visualize the protein.
Lack of cross-contamination of basolateral with apical membranes was revealed by Western blotting of the apical membrane marker protein H+/K+-ATPase [31]. The procedure was identical to that described above, using mouse monoclonal anti-hog H+/K+-ATPase beta (1 : 1000 dilution, overnight, 4°C), from Thermo Scientific (USA), and goat anti-mouse IgG-HRP (1 : 2000 dilution, 1 h, room temperature), from Sigma (USA), as primary and secondary antibodies, respectively.
Calculations. GraphPad Prism 2 was used to compare binding results with models of one or more binding sites, to draw graphs, and to calculate the parameters that define the binding: maximum binding capacity (Bmax), radio-ligand–receptor dissociation constant in equilibrium (KD) in saturation assays, and IC50 value and inhibition constant (Ki) in competitive binding assays. These parameters were calculated directly from nonlinear regression curves.
RESULTS
Binding of radio-ligands to GRP94 in parietal cell membranes from rabbit gastric mucosa. We first investigated whether the membranes isolated from parietal cells exhibited binding sites of GRP94 for the adenosine derivative NECA. In parallel experiments, we confirmed that the originating parietal cells were functional and readily activated by histamine as they displayed a dose-response increased secretion of hydrochloric acid that reached around 15-fold when exposed to 10–5, 10–4, or 10–3 M histamine (data not shown).
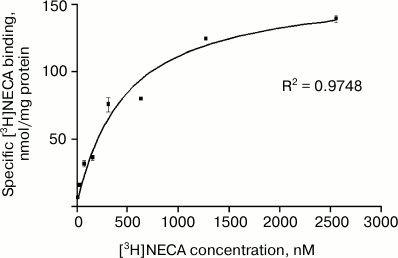
The kinetics of binding of [3H]NECA to membranes of gastric parietal cells is shown in Fig. 1. Increasing concentrations of the radio-ligand were used (from 20 to 2560 nM), and the binding turned out to be saturable. The curve fit best to a unique binding site model. Binding site density, indicated by Bmax, was 163.4 nmol/mg protein, and the affinity of the receptor to the radio-ligand, indicated by the dissociation constant in equilibrium, KD, was 466.7 nM. This KD is in the range described for purified GRP94 and corresponds to the affinity observed when the assay is performed at 4°C [22, 24].
Fig. 1. Saturation of [3H]NECA binding to parietal cell plasma membranes from rabbit gastric mucosa. Membranes (0.5 mg protein/ml) were incubated at 4°C for 1 h with 20 to 2560 nM [3H]NECA. Specific binding was calculated subtracting nonspecific binding (determined in presence of saturating, 100 µM NECA) from total binding. The Bmax value obtained was 163.4 nmol/mg protein and KD value 466.7 nM. Data represent the mean ± SEM, n = 3 samples from six animals.
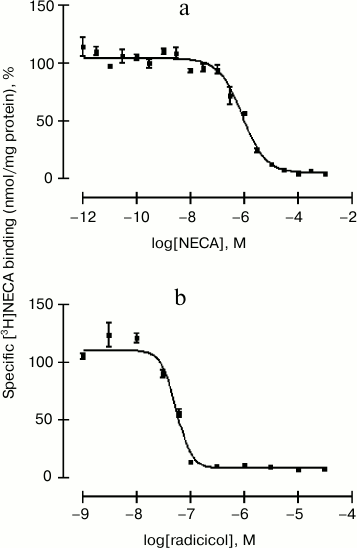
Two distinct assays were performed to study the displacement of [3H]NECA from GRP94. NECA was used as competitor in the first assay. After membrane saturation with [3H]NECA, 10–12 to 10–3 M unlabeled NECA was added. The competition curve was monophasic, as shown in Fig. 2a. The binding constant of the competitor ligand, Ki, was 328 nM, and the concentration at which the inhibition is 50% of maximum, IC50, was 848 nM. These values are in the range of nanomolar and correspond to the binding to GRP94.
Fig. 2. Displacement of the specific binding of [3H]NECA to parietal cell plasma membranes from rabbit gastric mucosa by NECA (a) and radicicol (b). Concentrations 10–12 to 10–3 M NECA (a) or 10–9 to 3⋅10–5 M radicicol (b) were added after saturation with 200 nM [3H]NECA to membranes (0.5 mg protein/ml) and incubated at 4°C for 1 h. NECA showed a Ki value of 328 nM and an IC50 value of 848 nM, and in the case of radicicol those values were 18.9 and 48.9 nM, respectively. Data represent the mean ± SEM, n = 3 samples from six animals, normalized with the maximal binding of [3H]NECA.
In the second competition assay, 10–9 to 3⋅10–5 M radicicol was used to displace [3H]NECA from GRP94. The competition curve fit to a unique binding site model (Fig. 2b). Radicicol bound to GRP94 with a Ki of 18.9 nM and displaced [3H]NECA with an IC50 of 48.9 nM. The Ki value coincides with the value described by Roe et al. for the inhibition of GRP94 by radicicol [39]. These results indicate that the inhibition by radicicol is more potent than the inhibition by NECA, as expected considering the binding dynamics of both competitors to GRP94. This chaperone normally exists exclusively as a homodimer composed by two identical monomers. Radicicol is able to completely occupy the two nucleotide-binding sites present in the native GRP94, while other ligands, such as NECA, occupy one binding site and induce a conformational change that hinders the subsequent binding of other ligands [24]. This set of radio-ligand binding experiments demonstrate that GRP94 is inserted into the membrane in a potentially active manner, as it is able to bind both the adenosine receptor agonist NECA and displacers with affinity characteristics reported by others [24].
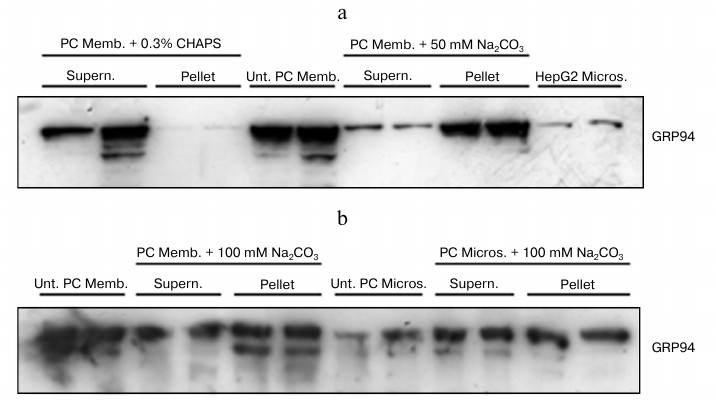
Immunodetection of GRP94 by Western blotting and analysis of its association to membranes. Based on the findings obtained in the radio-ligand binding experiments, we performed immunodetection assays to confirm the presence of GRP94 in membranes of gastric parietal cells and selective solubilization followed by Western blotting assays to assess its association degree to the membrane. We used microsomes isolated from both parietal cells isolated from rabbit gastric mucosa and from HepG2 cells as a positive control. Membranes were subjected to treatment with (i) 0.3% CHAPS, which completely solubilizes membrane proteins, or (ii) 100 mM Na2CO3, which extracts strongly membrane-associated proteins, or (iii) 50 mM Na2CO3, which releases peripheral proteins. GRP94 was afterwards immunodetected in the original membrane preparations (memb.) and the pellets (pellet) and supernatant (supern.) fractions.
Figure 3a shows that GRP94 was completely solubilized from plasma membrane by CHAPS, while the protein remaining in membranes was nearly negligible. The proportion of protein solubilized by 50 mM Na2CO3 was small, and most of it remained associated with the plasma membrane. Figure 3b shows that 100 mM Na2CO3 extracted much more protein than 50 mM Na2CO3 but, even so, in both microsomes and plasma membrane GRP94 is detectable in pellets and also in supernatants. We conclude that GRP94 is not an integral protein, since a part of it is extracted by 50 mM Na2CO3, but its association with the lipid bilayer and neighbor proteins is strong since a substantial proportion of GRP94 remains associated with the membranes after extraction with 100 mM Na2CO3.
Fig. 3. Extraction of GRP94 from plasma membranes and microsomes from rabbit gastric parietal cells. Parietal cell (PC) plasma membranes (memb.), and microsomes (micros.) were treated with 0.3% CHAPS, or 50 mM (a) or 100 mM Na2CO3 (b), or remained untreated (Unt.) (a, b). After ultracentrifugation (230,000g, 1 h) proteins attached to membranes remained in pellets (pellet), while supernatants (supern.) contained the released proteins. In (a), HepG2 microsomes were included as positive controls for GRP94 immunodetection. Twenty micrograms of total protein were fractionated on 8% SDS-PAGE and Western blotted for GRP94. Blots shown are representative of three independent experiments.
The main anti-GRP94 reactivity corresponded to a band of 94 kDa. There were also bands of lower but non-negligible intensity of a molecular weight around 60 kDa (data not shown). Those might be the dissociated monomers of this chaperone or also the consequence of certain heterogeneity in the endogenous GRP94 reservoir as a result of different glycosylation degrees. Whereas other authors have observed the presence of GRP94 oligomers [40], no immunoreactive bands of molecular weight above 94 kDa were detected in our samples, excluding the existence of interactions, in vivo or in vitro, leading to oligomerization.
GRP94 is expressed in the basolateral membrane of gastric parietal cells. As mentioned in the introduction, the plasma membrane domains of parietal cells participate in a formidably specific way in gastric secretion and other cells functions. Having proved that GRP94 is anchored to the plasma membrane of gastric parietal cells, we aimed to determine if GRP94 associates to the same extent with the apical and to the basolateral membrane or if, on the contrary, its distribution is asymmetric.
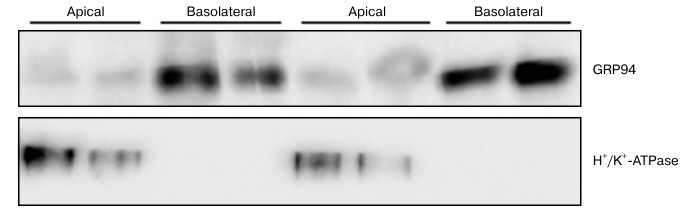
After exhaustive evaluation of purity of the cell membrane subfractions, we detected by Western blotting an intense expression of GRP94 in basolateral membranes, while its presence in apical membranes is dramatically low (Fig. 4). Absolute lack of cross-contamination of basolateral with apical membranes is shown in Fig. 4, as the apical membrane marker H+/K+-ATPase protein [31] band signal was absent in the corresponding lanes.
Fig. 4. Immunodetection of GRP94 in plasma membrane basolateral and apical subfractions from parietal cells isolated from rabbit gastric mucosa. Membrane subfractions were isolated and characterized as described in “Materials and Methods”. Fifty micrograms of total protein were fractionated on 8% SDS-PAGE and Western blotted for GRP94 and H+/K+-ATPase. Blots shown are representative of three independent experiments.
DISCUSSION
Parietal cells secrete concentrated hydrochloric acid into the gastric lumen. To fulfill this task, they are equipped with a broad variety of functionally coupled apical and basolateral ion transporters as well as of receptors to ensure tightly regulated acid secretion. Parietal cells are thus endowed with basolateral transporters for K+ and Cl– uptake, Cl–/HCO3–exchangers, and Na+/K+-ATPase [1], to name some, and with an array of receptors [41]. In this study, we show that GRP94 is mainly expressed in the basolateral domain of gastric mucosa parietal cells, and that it binds radicicol and the adenosine receptor agonist NECA eliciting affinity characteristics that fit with those of a proper receptor.
GRP94 is one of the most abundant proteins in the lumen of the mammalian ER and is normally confined to it, but GRP94 has also been shown to escape the KDEL-mediated retention system of the ER to be expressed at the plasma membrane. Several functions have been assigned to GRP94, some of them general and others cell type-specific. The most relevant ones are those derived from its interaction with proteins and peptides in the ER [42], where GRP94 plays an essential role in structural maturation and/or in trafficking of proteins and peptides targeted to cell surface [43], but, to the best of our knowledge, the significance of surface GRP94 expression and the mechanisms through which it acts are not known and might well differ from those occurring at the ER level.
Past investigations on the identification and characterization of adenosine receptors performed with NECA identified GRP94 as a high-affinity NECA-binding protein [22] and radicicol as a potent and specific competitor of NECA binding to GRP94 (Ki = 19 nM) [24], whereas GRP94 was not able to bind to N-6 substituted adenosine derivatives or xanthines [36]. With respect to the radio-ligand binding assays shown here, the KD obtained in saturation assays for [3H]NECA (Fig. 1) is in the nanomolar range (466.7 nM) (Fig. 1). When we inhibited the binding of [3H]NECA, Ki was calculated to be 328 nM for NECA and 18.9 nM for radicicol (Fig. 2). These values are in close agreement with those described for purified GRP94 [24] and suggest comparable ligand interactions in the isolated and the parietal cell membrane-embedded protein, indicating that active receptor regions are exposed at the membrane surface. This concept is also supported by our finding that 200 µM R-PIA (an N-6 substituted adenosine analog) was not able to displace NECA binding to GRP94 (unpublished observation).
Whether the high Bmax obtained in saturation assays with [3H]NECA could be the consequence of a high concentration of GRP94 in parietal cell membranes is an open question. In studies using solubilized GRP94 from human placenta [44] or human platelets [45], tissues in which GRP94 is abundant, Bmax values were in the nanomolar range even though they used longer incubation times than us. Here we show that GRP94 is easily detectable in gastric mucosa parietal cell membranes of adult rabbits. A high concentration of GRP94 in the plasma membrane of parietal cells suggests that its function is likely to be different to that in ER and related to the adenosine regulatory function. Two points are immediately evident from our data, collectively taken. First, a role for cell surface GRP94 as an adenosine nucleotide receptor given that NECA binds to the conserved adenosine nucleotide-binding domain of GRP94 and other heat shock proteins. Second, and as far as GRP94 is also able to bind adenosine [36], the endogenous purine nucleoside that modulates many physiological processes, that surface GRP94 might serve to mediate endogenous adenosine binding and regulatory activity.
We have also demonstrated the presence of GRP94 in the plasma membrane of parietal cells isolated from rabbit gastric mucosa by immunodetection. By combining Western blotting and the selective extraction of proteins from isolated membranes we identified GRP94 as a non-integral protein that is strongly associated with the plasma membrane of the gastric mucosa parietal cell (Fig. 3). These data agree with findings by us of physicochemical and structural predictive analyses of the rabbit GRP94 protein sequence (GI:2581793, accession AF001631.1) carried out with bioinformatics tools (ExPASy, unpublished observation). When membranes were treated with different Na2CO3 concentrations, the proportion of GRP94 remaining in the membranes was significant, although most of it had been released.
Given that parietal cells are a paradigm of structurally and functionally polarized cells, we asked whether cell-surface GRP94 expression may be different in apical and basolateral domains. In comparing the expression of this chaperone in apical and basolateral membrane subfractions purified by centrifugation in sucrose concentration gradient by Western blotting, clear differences were noted. The results revealed intense GRP94 expression in basolateral membrane, while its presence in apical membrane was relatively negligible (Fig. 4). It is tempting to speculate that this difference in the location of GRP94 is related to the function of the protein, possibly in association with one of the receptors or other proteins present in the basolateral domain of plasma membrane. GRP94 in basolateral membrane is likely to come from ER, where it is present as observed in microsomes from parietal cells (Fig. 3). The function of GRP94 in gastric parietal cells is unknown, but it should be achieved in the resting state of parietal cells when hydrochloric acid secretion takes place at basal rates.
In conclusion, we can formally confirm that GRP94 is tightly associated with the cell surface of parietal cells from rabbit gastric mucosa, where it exhibits high-affinity binding for the adenosine receptor agonist NECA and specific inhibitor radicicol with affinity characteristics of a proper receptor, and identify GRP94 particularly expressed in the basolateral membrane domain. Whether GRP94 can be functionally interacting with a protein of or taking part in some of the mechanisms operating preferentially in this plasma membrane domain is an open question that needs complementary analysis.
We thank Dr. Chico for her invaluable help in Western blotting assays and Dr. Fresnedo with in silico GRP94 sequence analysis.
This study was funded by the University of the Basque Country to RMA (NUPV 08/10) and the Basque Government to BO and YR (IT366-10).
REFERENCES
1.Kopic, S., Murek, M., and Geibel, J. P. (2010)
Am. J. Physiol. Cell. Physiol., 298, C1-C10.
2.Immormino, R. M., Dollins, D. E., Shaffer, P. L.,
Soldano, K. L., Walker, M. A., and Gewirth, D. T. (2004) J. Biol.
Chem., 279, 46162-46171.
3.Soldano, K. L., Jivan, A., Nicchitta, C. V., and
Gewirth, D. T. (2003) J. Biol. Chem., 278,
48330-48338.
4.De Crom, R., van Haperen, R., Janssens, R., Visser,
P., Willemsen, R., Grosveld, F., and van der Kamp, A. (1999)
Biochim. Biophys. Acta, 1437, 378-392.
5.Liu, B., Dai, J., Zheng, H., Stoilova, D., Sun, S.,
and Li, Z. (2003) Proc. Natl. Acad. Sci. USA, 100,
15824-15829.
6.Frasson, M., Vitadello, M., Brunati, A. M., La
Rocca, N., Tibaldi, E., Pinna, L. A., Gorza, L., and Donella-Deana, A.
(2009) Biochim. Biophys. Acta, 1793, 239-252.
7.Altmeyer, A., Maki, R. G., Feldweg, A. M., Heike,
M., Protopopov, V. P., Masur, S. K., and Srivastava, P. K. (1996)
Int. J. Cancer, 69, 340-349.
8.Gorza, L., and Vitadello, M. (2000) FASEB
J., 14, 461-475.
9.Bruneau, N., Lombardo, D., Levy, E., and Bendayan,
M. (2000) Microsc. Res. Tech., 49, 329-345.
10.Nganga, A., Bruneau, N., Sbarra, V., Lombardo,
D., and Le Petit-Thevenin, J. (2000) Biochem. J., 352,
865-874.
11.McLaughlin, M., and Vandenbroeck, K. (2011)
Br. J. Pharmacol., 162, 328-345.
12.Weekes, M. P., Antrobus, R., Talbot, S., Hor, S.,
Simecek, N., Smith, D. L., Bloor, S., Randow, F., and Lehner, P. J.
(2012) J. Proteome Res., 11, 1475-1484.
13.Marzec, M., Eletto, D., and Argon, Y. (2012)
Biochim. Biophys. Acta, 1823, 774-787.
14.Pizzo, P., Scapin, C., Vitadello, M., Florean,
C., and Gorza, L. (2010) J. Cell. Mol. Med., 14,
970-981.
15.Bando, Y., Katayama, T., Kasai, K., Taniguchi,
M., Tamatani, M., and Tohyama, M. (2003) Eur. J. Neurosci.,
18, 829-840.
16.Baek, H. Y., Lim, J. W., Kim, H., Kim, J. M.,
Kim, J. S., Jung, H. C., and Kim, K. H. (2004) Biochem. J.,
379, 291-299.
17.Cho, S. O., Lim, J. W., Jun, J. H., Kim, K. H.,
and Kim, H. (2010) Dig. Dis. Sci., 55, 1550-1564.
18.Zheng, H. C., Takahashi, H., Li, X. H., Hara, T.,
Masuda, S., Guan, Y. F., and Takano, Y. (2008) Hum. Pathol.,
39, 1042-1049.
19.Chen, X., Ding, Y., Liu, C. G., Mikhail, S., and
Yang, C. S. (2002) Carcinogenesis, 23, 123-130.
20.Langer, R., Feith, M., Siewert, J. R., Wester, H.
J., and Hoefler, H. (2008) BMC Cancer, 8, 70.
21.Takahashi, H., Wang, J. P., Zheng, H. C., Masuda,
S., and Takano, Y. (2011) Histol. Histopathol., 26,
663-671.
22.Hutchison, K. A., and Fox, I. H. (1989) J.
Biol. Chem., 264, 19898-19903.
23.Fredholm, B. B., Ijzerman, A. P., Jacobson, K.
A., Linden, J., and Muller, C. E. (2011) Pharmacol. Rev.,
63, 1-34.
24.Rosser, M. F., and Nicchitta, C. V. (2000) J.
Biol. Chem., 275, 22798-22805.
25.Berglindh, T., and Obrink, K. J. (1976) Acta
Physiol. Scand., 96, 150-159.
26.Fryklund, J., Wallmark, B., Larsson, H., and
Helander, H. F. (1984) Biochem. Pharmacol., 33,
273-280.
27.Ainz, L. F., Salgado, C., Gandarias, J. M.,
Gomez, R., Vallejo, A., and Gil-Rodrigo, C. E. (1993) Pharmacol.
Res., 27, 319-334.
28.Sanders, M. J., and Soll, A. H. (1989) Methods
Enzymol., 171, 482-497.
29.Muallem, S., Burnham, C., Blissard, D.,
Berglindh, T., and Sachs, G. (1985) J. Biol. Chem., 260,
6641-6653.
30.Evans, W. H. (1978) Preparation and
Characterization of Mammalian Plasma Membranes, North-Holland
Publishing Company, Amsterdam.
31.Saccomani, G., Helander, H. F., Crago, S., Chang,
H. H., Dailey, D. W., and Sachs, G. (1979) J. Cell Biol.,
83, 271-283.
32.Culp, D. J., and Forte, J. G. (1981) J. Membr.
Biol., 59, 135-142.
33.Bradford, M. M. (1976) Anal.
Biochem., 72, 248-254.
34.Casad, V., Casillas, T., Mallol, J., Canela, E.
I., Lluis, C., and Franco, R. (1992) J. Neurochem., 59,
425-431.
35.Moulin, E., Zoete, V., Barluenga, S., Karplus,
M., and Winssinger, N. (2005) J. Am. Chem. Soc., 127,
6999-7004.
36.Lorenzen, A., Engelhardt, J., Kerst, B., and
Schwabe, U. (1998) Biochem. Pharmacol., 55, 455-464.
37.Fujiki, Y., Hubbard, A. L., Fowler, S., and
Lazarow, P. B. (1982) J. Cell Biol., 93, 97-102.
38.Laemmli, U. K. (1970) Nature, 227,
680-685.
39.Roe, S. M., Prodromou, C., O’Brien, R.,
Ladbury, J. E., Piper, P. W., and Pearl, L. H. (1999) J. Med.
Chem., 42, 260-266.
40.Nemoto, T. K., Ono, T., and Tanaka, K. (2001)
Biochem. J., 354, 663-670.
41.Urushidani, T., and Forte, J. G. (1997) J.
Membr. Biol., 159, 99-111.
42.Eletto, D., Dersh, D., and Argon, Y. (2010)
Semin. Cell Dev. Biol., 21, 479-485.
43.Ying, M., and Flatmark, T. (2006) FEBS J.,
273, 513-522.
44.Hutchison, K. A., Nevins, B., Perini, F., and
Fox, I. H. (1990) Biochemistry, 29, 5138-5144.
45.Fein, T., Schulze, E., Bar, J., and Schwabe, U.
(1994) Naunyn Schmiedebergs Arch. Pharmacol., 349,
374-380.