3-O-Methoxyimino Group Inhibits Interactions between Progestins and Blood Transcortin
A. V. Polikarpova1*, I. S. Levina2, and A. N. Smirnov1#
1Biological Faculty, Lomonosov Moscow State University, Leninsky Gory 1/12, 119899 Moscow, Russia; fax: (495) 939-4309; E-mail: kairo911@gmail.com2Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Leninsky pr. 47, 117913 Moscow, Russia; fax: (499) 135-5328; E-mail: li@ioc.ac.ru
* To whom correspondence should be addressed.
# Deceased.
Received May 17, 2013
The interactions between E- and Z-isomers of 3-O-methoxyimino-pregn-4-ene-20-one and its 17α-hydroxy derivative and transcortin from human blood were investigated. The substitution of the progesterone 3-oxo group for a 3-O-methoxyimino group was shown to diminish the affinity of the steroid for transcortin by approximately one order of magnitude irrespective of the substituent’s orientation. The data suggests that progesterone derivatives substituted thereby must have higher bioavailability compared to progesterone and must not significantly affect the biodynamics of glucocorticoid in vivo.
KEY WORDS: selective progesterone analogs, transcortinDOI: 10.1134/S0006297913100076
Abbreviations: mPR, membrane progesterone receptor; RCA, relative competitive activity.
The search for the structural determinants of progestins determining
their selective interaction with membrane progesterone receptor (mPR)
alpha has shown that the substitution of a 3-keto group of pentacyclic
progesterone derivatives for a 3-O-methoxyimino group is a
highly efficient method of discrimination of the interaction of
compounds with the membrane and nuclear progesterone receptors [1]. However, when thereby modified progesterone
derivatives are used in vivo to elucidate the biological role of
mPR, one should take into consideration the possibility of their
interactions with other steroid-binding proteins. The main part of the
blood plasma progesterone forms a complex with corticosteroid-binding
globulin, or transcortin, and the affinity of progesterone to the
latter is comparable with the affinity of glucocorticoids [2]. According to the data of X-ray structure analysis,
the 3-keto group of glucocorticoids is not involved in polar
interactions with transcortin [3]. Therefore, one
could anticipate that the substitution of this group in progesterone
for a 3-O-methoxyimino group would have no significant influence
on transcortin binding. However, surprisingly it has turned out that
such substitution drastically reduces the affinity of steroids for
protein.
MATERIALS AND METHODS
3-O-Methoxyimino-pregn-4-ene-20-one and 3-O-methoxyimino-pregn-4-ene-17α-ol-20-one were synthesized from progesterone and 17α-hydroxyprogesterone, respectively, in three stages by way of selective protection of the 20-oxo group by ethylene ketal, the treatment of derived ketal with O-methylhydroxylamine hydrochloride, and elimination of the protection by treatment with p-TsOH. Individual E- and Z-isomers of 3-O-methoxyimino-pregn-4-ene-20-ones were isolated by chromatography. The compared steroids (progesterone and hydrocortisone) were obtained from Sigma (USA). [1,2-3H]Progesterone with a specific radioactivity of 40 Ci/mmol was synthesized by V. P. Shevchenko at the Institute of Molecular Genetics (Russian Academy of Sciences).
Blood serum was obtained by incubation of whole donor (female) blood at room temperature for 30 min and then at 4ºC for 15 min, followed by centrifugation at 1500g at 4ºC for 10 min. The resultant serum was treated with 2% dextran-coated activated charcoal for 15 min at 40ºC under continuous shaking to remove endogenous steroids. After precipitation of the charcoal by centrifugation at 1500g for 5 min at 4ºC, the serum was divided into aliquots and stored at –20ºC. Before use, the serum was thawed and diluted 40 times with 50 mM Tris-HCl buffer (pH 7.5) containing 10 mM KCl and 1 mM EDTA.
The interaction between progesterone derivatives and transcortin was analyzed by incubating 100 µl of diluted serum for 2 h at 4ºC with 100 µl of the mixture of [3H]progesterone (final concentration of 6-7 nM) and unlabeled competitor (final concentrations of 3.2-6320 nM) in the same buffer. [3H]Progesterone that was not bound to protein was removed by treating the incubation mixture with 100 µl of 2% suspension of dextran-coated activated charcoal for 5 min at 4ºC and precipitating the charcoal by centrifugation at 1500g for 5 min at 4ºC. Radioactivity was measured in the supernatant.
The equilibrium dissociation constant (Kd) and progesterone binding site concentrations (Bmax) were determined by selecting the values of these parameters that would provide the maximum correspondence of experimental data to the one-protein-one-ligand kinetic model. The Kd values for competitors were obtained for the one-protein-two-ligand model by analogy [4]. The values of relative competitive activity (RCA) were calculated as a ratio of progesterone and competitor Kd values in each experiment, followed by calculation of the mean and standard error. The differences in RCA values were estimated by the Mann–Whitney nonparametric criterion in Statistica 8.0.
RESULTS AND DISCUSSION
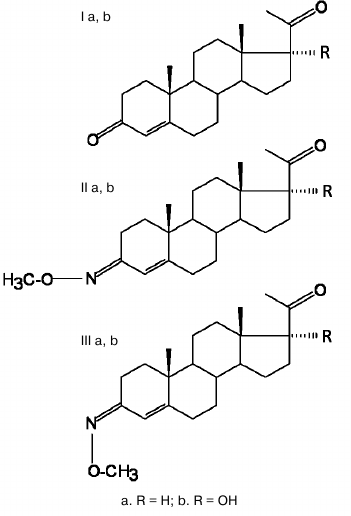
Figure 1 shows the structures of the analyzed compounds and the initial progesterone and its 17α-hydroxy derivative.
Fig. 1. Structure of progesterone (R = H) and 17α-hydroxyprogesterone (R = OH) (Ia,b), E-isomers (IIa,b), and Z-isomers (IIIa,b) of 3-O-methoxyimino-pregn-4-ene-20-one (R = H) and 3-O-methoxyimino-pregn-4-ene-17α-ol-20-one (R = OH).
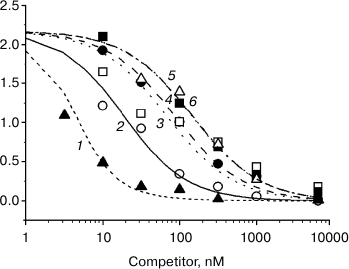
Figure 2 presents the results of a typical experiment. One can see that hydrocortisone displaces [3H]progesterone from protein complexes more effectively than progesterone, while 3-O-methoxyiminic progesterone derivatives displace it less effectively.
Fig. 2. Curves of displacement by unlabeled steroids of [3H]progesterone from complexes with serum protein: 1) hydrocortisone; 2) progesterone; 3) (Z)3-O-methoxyimino-pregn-4-ene-20-one; 4) (E)3-O-methoxyimino-pregn-4-ene-20-one; 5) (Z)3-O-methoxyimino-pregn-4-ene-17α-ol-20-one; 6) (E)3-O-methoxyimino-pregn-4-ene-17α-ol-20-one.
The quantitative characteristics of ligand–protein interaction are given in the table. One can see that the affinity of hydrocortisone to the serum protein exceeds 5-fold the affinity of progesterone. The affinity of three out of four 3-O-methoxyiminic progesterone derivatives is approximately 10 times lower than the affinity of progesterone. No reliable differences were observed between 3-O-methoxyiminic derivatives in the affinity to the serum protein.
Relative competitive activity (RCA) of analyzed steroids in
[3H]progesterone displacement from complexes with serum
protein

Note: The number of measurements is given in parentheses. Asterisk
(*) denotes significant differences from 100%. The Kd
value for progesterone binding was 10.4 ± 6.4 (4) nM.
The higher affinity of hydrocortisone to the serum protein compared to progesterone (table) and good correspondence of the experimental data for all steroids of the one-protein-two-ligand kinetic model (Fig. 2) indicate that transcortin is the only protein that specifically binds [3H]progesterone in the serum.
Transcortin–steroid ligand complexes are formed, according to the data of X-ray structure analysis, with the involvement of polar (direct and water molecule mediated) and hydrophobic interactions. In contrast to the nuclear receptors of steroid hormones (receptors of gluco- and mineral corticoids, progesterone, and androgens), which form a complex network of hydrogen bonds with the 3-keto group of their ligands [5, 6], the 3-keto group of steroids is not involved in transcortin binding. However, it is located very close to the amino acid chain of the N-terminus of the protein molecule [3]. It seems that the larger 3-O-methoxyiminic substituent of progesterone derivatives investigated in this work becomes a steric obstacle for high affinity steroid–protein interaction just because of this close proximity. The absence of substantial differences between E- and Z-isomers in their competitive activity is indicative of the limited size of the cavity of the transcortin ligand-binding pocket adjacent to the steroid A-ring in both equatorial and zodiac direction.
The results suggest that 3-O-methoxyiminic progesterone derivatives, when introduced in vivo, must have a shorter (compared to progesterone) half-life in the blood stream due to accelerated diffusion into the intercellular space of organs and tissues, i.e. they must have higher bioavailability. In addition, the low affinity to transcortin suggests that introduction in vivo of compounds of this group even in significant, pharmacological amounts must not be accompanied by significant disturbances in the bioavailability and biodynamics of glucocorticoids.
REFERENCES
1.Lisanova, O. V., Shchelkunova, T. A., Morozov, I.
A., Rubtsov, P. M., Levina, I. S., Kulikova, L. E., and Smirnov, A. N.
(2013) Biochemistry (Moscow), 78, 236-243.
2.Westphal, U. (1970) in Steroid Assay by Protein
Binding (Diczfalusy, E., ed.) Karolinska Symposia on Research
Methods in Reproductive Endocrinology, Stockholm, pp. 122-140.
3.Klieber, M. A., Underhill, C., Hammond, G. L., and
Muller, Y. A. (2007) J. Biol. Chem., 282,
29594-29603.
4.Smirnov, A. N., Pokrovskaya, E. V., Kogteva,
G. S., Shevchenko, V. P., Levina, I. S., Kulikova, L. E., and
Kamernitzky, A. V. (2000) Steroids, 65, 163-170.
5.Williams, S. P., and Sigler, P. B. (1998)
Nature, 393, 392-396.
6.Bledsoe, R. K., Montana, V. G., Stanley, T. B.,
Delves, C. J., Apolito, C. J., McKee, D. D., Consler, T. G., Parks, D.
J., Stewart, E. L., Willson, T. M., Lambert, M. H., Moore, J. T.,
Pearce, K. H., and Xu, H. E. (2002) Cell, 110,
93-105.