Development of Approaches to Creation of Experimental Test System for Evaluation of Antigenic Activity of Synthetic Oligosaccharide Ligands Related to Fragments of the Streptococcus pneumoniae Type 14 Capsular Polysaccharide
E. A. Kurbatova1*, D. S. Vorobiov1, I. B. Semenova1, E. V. Sukhova2, D. V. Yashunsky2,3, Y. E. Tsvetkov2, and N. E. Nifantiev2
1Mechnikov Research Institute of Vaccines and Sera, Russian Academy of Medical Sciences, Malyi Kazennyi Pereulok 5a, 105064 Moscow, Russia; fax: (495) 917-4900; E-mail: kurbatova6162@yandex.ru2Laboratory of Glycoconjugate Chemistry, N. D. Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Leninsky pr. 47, 119991 Moscow, Russia; fax: (499) 135-8784; E-mail: nen@ioc.ac.ru
3Orekhovich Research Institute of Biomedical Chemistry, Russian Academy of Medical Sciences, Pogodinskaya ul. 10/8, 119121 Moscow, Russia
* To whom correspondence should be addressed.
Received December 22, 2012; Revision received January 26, 2013
A conjugate of a synthetic hexasaccharide fragment of the Streptococcus pneumoniae type 14 capsular polysaccharide with bovine serum albumin (BSA) has been prepared. The antigenic activity and specificity of this conjugate are comparable with those of natural antigens of S. pneumoniae type 14. The data suggest that the resulting synthetic conjugate can be used as a coating antigen in an experimental test system (based on enzyme immunoassay) for evaluating the antigenic activity and specificity of synthetic oligosaccharide ligands and for testing specimens of natural capsular polysaccharides and immune sera.
KEY WORDS: Streptococcus pneumoniae type 14, capsular polysaccharide, synthetic oligosaccharide fragment, oligosaccharide–BSA conjugate, antigenic activityDOI: 10.1134/S0006297913070122
Bacteria of the Streptococcus pneumoniae species are causative agents of severe inflammatory diseases of the respiratory tract, meningitis, otitis, bacteremia, etc. [1-3] that sometimes have lethal outcome [4, 5]. The majority of pneumococcal strains have a polysaccharide capsule that is an important virulence factor of this microorganism [6, 7]. Based on antigenic features of the polysaccharide capsule, 46 serogroups of S. pneumoniae have been described, and among them more than 90 serotypes have been determined [8-10]. The spectrum of prevailing types of the capsule varies depending on age, time factor, and geographical region, but the most frequent serotypes are identified everywhere. But only about 20 serotypes of S. pneumoniae are associated with more than 80% of invasive pneumonias [11].
During the last two decades, S. pneumoniae type 14 has been recorded as one of the most widely distributed S. pneumoniae serotypes [11-16], and its natural capsular polysaccharide is a component of all commercial pneumococcal vaccines.
The capsular polysaccharide of S. pneumoniae type 14 consists of tetrasaccharide repeating units [17] as its minimal structural component with immunological activity. The structure of the S. pneumoniae type 14 capsular polysaccharide is well known, and this makes it possible to chemically synthesize its oligosaccharide fragments with different length. Conjugates of synthetic oligosaccharide fragments of the S. pneumoniae type 14 capsular polysaccharide with a protein carrier are known to induce formation of IgG antibodies specific to the same type native capsular polysaccharide [18]. Such synthetic immunologically active conjugates are more advantageous than natural capsular polysaccharides, which frequently contain admixtures of protein, teichoic acids (C-polysaccharide), nucleic acids, and other microbial antigens whose separation is difficult.
To develop approaches for creating conjugated pneumococcal vaccines based on synthetic oligosaccharide ligands, it was necessary to develop a method for evaluation the antigenic activity of these ligands. The purpose of this work was to study the possibility of creating an experimental test system (based on enzyme immunoassay) with a synthetic hexasaccharide related to the fragment of the S. pneumoniae type 14 capsular polysaccharide chain used as a coating antigen.
MATERIALS AND METHODS
Strains of microorganisms. Strains of S. pneumoniae types 14, 19A, and 19F from the collection of the Mechnikov Research Institute of Vaccines and Sera, Russian Academy of Medical Sciences, were used for preparation of diagnostic antimicrobial sera through repeated immunization of mice with increasing doses of the living culture.
Obtaining natural antigenic preparations from S. pneumoniae type 14. Microbial cells were cultured in a brain–heart broth for 24 h at 37°C, the cells were separated by centrifugation, and the polysaccharide–protein complex was isolated from the supernatant by precipitation with acetone. The resulting preparation was used as a reference preparation.
Preparation from S. pneumoniae type 14 antigens was obtained from acetone-dried microbial cells by three sequential extractions with water [19]. The resulting preparation, which contained carbohydrates (5%), protein (53.5%), and nucleic acids (28%), was used as a reference preparation.
Pneumo 23 pneumococcal polyvalent polysaccharide vaccine (Sanofi Pasteur, France). One dose of the vaccine (0.5 ml) contains purified natural capsular polysaccharides of 23 types of the S. pneumoniae, including types 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F, and 33F (25 µg of each type in a buffer solution which contained phenol, NaCl, Na2HPO4·2H2O, NaH2PO4·2H2O, and 0.5 ml of water for injections). The vaccine was used for comparing the antigenic activity of the preparations under study.
Preparation of synthetic S. pneumoniae type 14 capsular polysaccharide has been described by Nifantiev, Backinowsky, and Kochetkov [20, 21].
Synthesis of the hexasaccharide fragment of S. pneumoniae type 14 (1) capsular polysaccharide will be described separately.
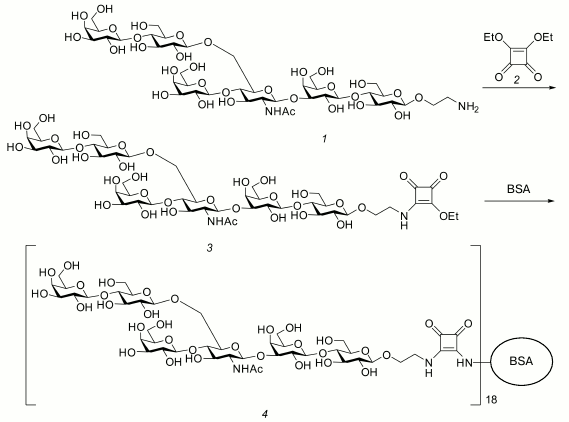
Preparation of hexasaccharide 1 conjugate with BSA (4) by a squarate approach [22, 23] is presented in Fig. 1. Diethyl squarate 2 (1.6 µl, 11 µmol) and Et3N (1.5 µl) were added to solution of hexasaccharide 1 (9.3 mg, 8.7 µmol) in 50% aqueous ethanol (800 µl), and the mixture was kept for 4 h at room temperature and then concentrated. The residue was dissolved in water and applied on a Sep-Pak C-18 cartridge (Waters, USA). The cartridge was washed with water (10 ml), and then the product was eluted with 2-ml portions of aqueous MeOH with the increasing methanol concentration from 5 to 20%. As a result of concentrating and subsequent lyophilization of the eluate, compound 3 (9 mg, 95%) was obtained from water as a white amorphous powder. High-resolution mass-spectrometry found m/z 1221.4015 [M+Na]+ (calculated for C46H74N2NaO34, m/z 1221.3982).
Compound 3 (9 mg, 7.5 µmol) and BSA (25 mg, 0.380 µmol) were kept at room temperature for 48 h in 190 µl of buffer solution (350 mM KHCO3 and 70 mM Na2B4O7·10H2O, pH 9). The resulting mixture was subjected to gel chromatography on a Sephadex G-15 column (350 ´ 25 mm) in water, and upon lyophilization conjugate 4 (34 mg, 98%) was obtained as an amorphous white powder. In the MALDI-TOF mass-spectrum, a wide peak was present with maximum at m/z 84342, which corresponds to inclusion into the conjugate of 18 hexasaccharide residues on average.
Fig. 1. Preparation of the glycoconjugate 4.
Chemical composition of the preparations. The content of protein in the preparations was determined spectrophotometrically at 750 nm by the Lowry method [24], of carbohydrates at 490 nm by the Dubois method [25], and of nucleic acids at 260 nm.
Preparation of hyperimmune mouse sera to S. pneumoniae type 14. To determine the specific activity of synthetic and natural preparations of the S. pneumoniae type 14 capsular polysaccharide, diagnostic hyperimmune sera were prepared to microbial cells of S. pneumoniae types 14, 19A, and 19F. The two latter sera were used as a positive control for possible cross-reactions with S. pneumoniae type 14. The immunizing dose of the living pneumococcus culture was chosen in preliminary experiments on infecting mice. The immunization was performed using no less than five doses of the microbial culture. For the first immunization, we used a dose of 106 microbial cells, which was not lethal for the animals on intraperitoneal injection. Subsequent immunization was performed with 10-fold increasing doses of living microbial culture with fortnight interval. The blood serum was obtained two weeks after the last immunization.
Enzyme-linked immunosorbent assay (ELISA) was performed to determine the titer of antibodies to the repeating hexasaccharide fragment of the S. pneumoniae type 14 capsular polysaccharide chain. The synthetic glycoconjugate 4 dissolved in phosphate-buffered saline (PBS) (Sigma, USA) was sorbed (0.2 µg/well) on flat-bottom polystyrene plates (Medpolymer, Russia) for 2 h at 37°C and left overnight at 4°C. Upon sorption, the antimicrobial serum (100 µl/well) was introduced into the wells in twofold dilutions starting from 1 : 100. The sera were diluted with PBS supplemented with 0.05% Tween 20 (Panreac Synthesis, Spain) and incubated for 1 h at 37°C in a thermostatted chamber. The serum was washed thrice with PBS containing Tween 20 (200 µl/well), and the working solution of the conjugate in 100 µl was added. As the conjugate, we used peroxidase-labeled dry (antispecies) diagnostic antibodies to IgG (H + L) of white mice produced by Medgamal (Gamaleya Institute of Experimental Immunology, Russia). The serum was washed thrice in PBS supplemented with Tween 20 (200 µl/well), and then the developing reagent tetramethylbenzidine (TMB; BioTest Sistemy, Russia) was added (100 µl/well). After 20 min, the reaction was stopped with 1 M solution of H2SO4 (Sigma). The optical density was determined with an ELISA reader (iMark, Japan) at 450 nm. Sera of intact (non-immunized) mice were used for control.
Competitive ELISA. To determine the antigenic activity of hexasaccharide 1, a working dilution of the serum in PBS supplemented with Tween 20 was added (90 µl/well) into all wells coated with conjugate 4. In the first two rows, 10 µl of PBS was added to the serum, and into all other experimental wells the studied antigens were added in PBS at the concentration of 10 µg/well. The plates were incubated at 37°C for 1 h. Then the wells were washed thrice with PBS supplemented with Tween 20 (200 µl/well), and the working dilution of the conjugate was added into the wells, which were then incubated for 45 min at 37°C and washed thrice with PBS supplemented with Tween 20 (200 µl/well). Then 100 µl of TMB was added as a developing reagent, and the reaction was stopped after 15 min with 1 M solution of H2SO4. The optical density was determined at 450 nm. The inhibition was calculated using the formula: 100% — x, where 100% was the optical density of the serum (OD450) before the antigen addition, and x was the serum activity (%) after the antigen addition.
The data were processed statistically using the Microsoft Excel 2007 and Biostat computer programs.
RESULTS
To determine in the homologous antimicrobial mouse serum the titer of specific antibodies to the S. pneumoniae type 14 capsular polysaccharide, twofold dilutions of conjugate 4 were sorbed on the solid phase, from 4 to 0.125 µg/well. In the serum the titer of IgG-antibodies (IgG-Ab) specific to the used synthetic hexasaccharide representing the capsular polysaccharide fragment was 1 : 400-1 : 800, which was significantly different from the antibody titer in the non-immunized control animals (1 : 100).
The findings show that synthetic hexasaccharide ligand 1 representing a fragment of the S. pneumoniae type 14 capsular polysaccharide chain specifically binds with antibodies to the capsular polysaccharide in the antimicrobial serum, which proves the antigenic activity of this hexasaccharide. Optimal sorption doses of the preparation were in the range from 0.125 to 1 µg/well. As a result, the sorption dose of 0.2 µg/well at dilution 1 : 2000-1 : 4000 of the antispecies conjugate with peroxidase-labeled diagnostic antibodies against mouse IgG (H + L) was chosen for further studies.
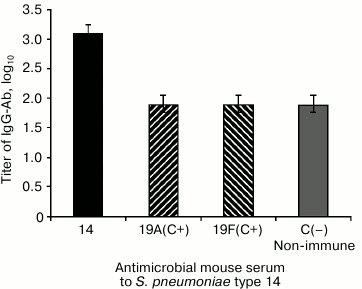
To reveal the specificity of the antigen—antibody interaction, i.e. the absence of possible cross-reactions with other capsular polysaccharides of S. pneumoniae, we used antimicrobial pneumococcal sera to the 19A and 19F types (Fig. 2). The antibodies presenting in the antimicrobial serum to S. pneumoniae type 14 bound with the coating antigen – conjugate 4 (the antibody titer expressed in log10 was 3.1), whereas antibodies presenting in the antimicrobial sera 19A and 19F reacted with this antigen on the control level, i.e. so as with the non-immune serum (the antibody titer expressed in log10 was 1.9).
Fig. 2. Study on cross-reactions of conjugate 4 with antimicrobial sera to S. pneumoniae type 19A and 19F capsular polysaccharides. Conjugate 4 (2 µg/well) was sorbed on the solid phase. The antimicrobial sera were prepared on the 14th day after the fivefold immunization of mice with increasing doses of living cultures of S. pneumoniae type 14, 19A, and 19F. The serum pool from 10 mice was studied. C, control.
These data confirmed the high specificity of the IgG-Ab reaction with hexasaccharide 1 on the solid phase and showed the absence of cross-reactions of IgG-Ab to S. pneumoniae type 14 capsular polysaccharide with those of 19A and 19F types.
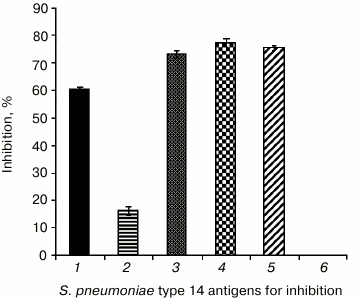
Studies on the antigenic activity of hexasaccharide 1 and conjugate 4 were continued using competitive ELISA. Conjugate 4 (0.2 µg/well) was sorbed on the solid phase. The immune antimicrobial serum to S. pneumoniae type 14 in working dilution 1 : 400 was used as a control, and the mean value of its optical density was taken as 100%. To inhibit ELISA, some antigens of S. pneumoniae type 14 were used at concentration 10 µg/well (Fig. 3).
Fig. 3. Specific interaction of conjugate 4 with IgG-Ab to S. pneumoniae type 14 capsular polysaccharide in competitive ELISA. 1) S. pneumoniae type 14 synthetic capsular polysaccharide; 2) hexasaccharide 1; 3) glycoconjugate 4; 4) commercial 23-valent pneumococcal vaccine containing type 14 natural capsular polysaccharide; 5) natural polysaccharide–protein complex; 6) S. pneumoniae type 14 microbial cell antigens. Conjugate 4 sorption dose in the plate was 0.2 µg/well; antispecies conjugate (peroxidase-labeled antibodies to mouse IgG) dilution was 1 : 2000; antimicrobial serum was prepared 14 days after fivefold immunization of mice with increasing doses of S. pneumoniae type 14 living culture; inhibition was performed with antigen dose of 10 µg/well in PBS; in the control, 10 µl of PBS without antigen was used.
On binding of serum IgG-Ab to capsular polysaccharide with the antigen in solution, the quantity of antibodies decreased, and this decreased quantity bound with the solid phase, which was manifested by a decrease in optical density. The IgG-Ab to the S. pneumoniae type 14 capsular polysaccharide presenting in the antimicrobial serum recognized with high specificity the studied pneumococcal antigens (% of the inhibition): synthetic capsular polysaccharide – 60%; hexasaccharide 1 – 16.3%; conjugate 4 – 73.1%; commercial 23-valent pneumococcal vaccine containing type 14 natural polysaccharide – 77.3%; natural polysaccharide–protein complex – 75.4%; microbial cell antigens – 0%.
Our findings show that synthetic hexasaccharide 1 representing the fragment of S. pneumoniae type 14 capsular polysaccharide within conjugate 4 possesses high antigenic activity comparable with the activity of natural antigens of S. pneumoniae type 14. This can be used for preparation of highly sensitive ELISA test systems, and prospectively also for preparation of semisynthetic conjugated vaccines against this type of pneumococcus. Note that studies on the potential of structurally related oligosaccharides representing different fragments of S. pneumoniae type 14 capsular polysaccharide revealed that just the oligosaccharide chains, similarly to the case of hexasaccharide 1, seem to be the most promising for creating conjugated carbohydrate vaccines against the considered pathogen [26]. Just this determined our choice of compound 1 for the present work.
DISCUSSION
The antigenic activity of synthetic hexasaccharide 1 structurally related to S. pneumoniae type 14 capsular polysaccharide and including the tetrasaccharide repeating unit of the polysaccharide has been studied. IgG-Ab of antimicrobial serum to S. pneumoniae type 14 recognized hexasaccharide 1 on the solid phase, whereas in the liquid phase hexasaccharide 1 poorly bound with IgG-Ab to the natural capsular polysaccharide of the antimicrobial serum. As a result, the reaction was inhibited only by 16.3%. On the contrary, conjugate 4 containing 18 hexasaccharide ligands joined to the protein carrier BSA actively bound with IgG-Ab in the liquid phase, and this increased the reaction inhibition to 73.1%, which was comparable to the antigenic activity of the synthetic and natural capsular polysaccharides. Most probably, the difference in the binding of monomeric oligosaccharide 1 and the oligosaccharide within conjugate 4 is due to the cluster effect [27] and/or to oligodentate binding with immunoglobulins in the sera.
Although the findings presented are still insufficiently used in practice, nevertheless we can determine the main lines for using the experimental ELISA test system based on conjugate 4. The ELISA test system can be used for evaluation of antigenic activity and specificity of synthetic oligosaccharide fragments of the capsular polysaccharide and their conjugates with protein carriers, for determination of titer of antibodies to S. pneumoniae type 14 in subjects immunized with pneumococcal vaccines to assess immunity level, for testing experimental specimens of pneumococcal capsular polysaccharides to specify their belonging to type 14, for determination of S. pneumoniae type 14 distribution in patients with pneumococcus-induced diseases by the level of pneumococcal IgG-Ab, for evaluation of S. pneumoniae type 14 circulation in populations of different regions and in different age groups, and also for other laboratory and clinical studies.
Diagnostic pneumococcal test systems based on the pneumococcus synthetic capsular polysaccharide or on its fragments can be useful for avoiding shortcomings of natural polysaccharides, such as presence of admixtures, using living cultures of microorganisms, expensive methods of isolation and purification of antigens, and will result in creation of qualitatively new modern ELISA test systems.
The findings can be used for development of synthetic and semisynthetic vaccines against pneumococcus and other socially important microorganisms possessing a polysaccharide capsule.
This work was supported by the Russian Foundation for Basic Research, project No. 11-04-01187.
REFERENCES
1.Tatochenko, V. K. (2008) Zh. Detsk.
Infekts., 2, 13-17.
2.Tatochenko, V. K. (2010) Zh. Mikrobiol.,
5, 90-98.
3.Vishnyakova, L. A. (1993) Pulmonologiya,
3, 17-20.
4.O’Brien, K. L., Wolfson, L. J., Watt, J. P.,
Henkle, E., Deloria-Knoll, M., McCall, N., Lee, E., Mulholland, K.,
Levine, O. S., and Cherian, T. (2009) Lancet, 374,
893-902.
5.Weinberger, D. M., Harboe, Z. B., Sanders, E. A.,
Ndiritu, M., Klugman, K. P., Ruckinger, S., Dagan, R., Adegbola, R.,
Cutts, F., Johnson, H. L., O’Brien, K. L., Scott, J. A., and
Lipsitch, M. (2010) Clin. Infect. Dis., 51, 692-699.
6.Melin, M., Trzcinski, K., Antonio, M., Meri, S.,
Adegbola, R., Kaijalainen, T., Kayhty, H., and Vakevainen, M. (2010)
Infect. Immun., 78, 5252-5261.
7.Hyams, C., Yuste, J., Bax, K., Camberlein, E.,
Weiser, J. N., and Brown, J. S. (2010) Infect. Immun.,
78, 716-725.
8.Kamerling, J. P. (2000) in Streptococcus
Pneumoniae Molecular Biology and Mechanism of Disease
(Tomasz, A., ed.) Mary Ann Libert, pp. 81-114.
9.Prevention of Pneumococcal Disease among Infants
and Children – Use of 13-Valent Pneumococcal Conjugate
Vaccine and 23-Valent Pneumococcal Polysaccharide Vaccine (2010)
MMWR Recomm. Rep., 59 (RR-11), 1-18.
10.Kozlov, P. S. (2010) MAK/MAX,
25.
11.Kozlov, P. S., Chagaryan, A. N., Kozlova, L. V.,
and Muraviev, A. A. (2011) Klin. Mikrobiol. Antimikrob.
Khimioter., 13, 177-187.
12.Coffey, T. J., Berron, S., Daniels, M.,
Garcia-Leoni, M. E., Cercenado, E., Bouza, E., Fenoll, A., and Spratt,
B. G. (1996) Microbiology, 142, 2747-2757.
13.Rusen, I. D., and Fraser-Poberts, M. D. (1997)
Ped. Infect. Dis., 16, 656-662.
14.Jansen, W. T., and Snippe, H. (2004) Ind. J.
Med. Res., 119 (Suppl.), 7-12.
15.Reinter, R. R., Kaufhold, A., Kuhnemund, O., and
Luttichen, R. (1994) Int. J. Med. Microbiol. Virol. Parasitol.
Infect. Dis., 281, 481-490.
16.Hausdorf, W. P., Bryant, J., Paradiso, P. R., and
Siber, G. R. (2000) Clin. Infect. Dis., 30, 100-131.
17.Lindberg, B., Lonngren, J., and Powel, D. A.
(1977) Carbohydr. Res., 58, 177-186.
18.Safari, D., Dekker, H. A. T., and Joosten, A. F.
(2008) Infect. Immun., 76, 4615-4623.
19.Egorova, N. B., Kurbatova, E. A., Miroshnichenko,
I. V., Mashilova, G. M., Gladus, M. A., Lazareva, E. S., Kiseleva, B.
S., and Vaneeva, N. P. (1983) Zh. Mikrobiol., 2,
96-100.
20.Kochetkov, N. K., Nifant’ev, N. E., and
Backinowsky, L. V. (1987) Tetrahedron, 43, 3109-3121.
21.Nifant’ev, N. E., Backinowsky, L. V., and
Kochetkov, N. K. (1988) Carbohydr. Res., 174, 61-72.
22.Chernyak, A., Karavanov, A., Ogawa, Y., and
Kovac, P. (2001) Carbohydr. Res., 330, 479-486.
23.Hou, S.-J., Saksena, R., and Kovac, P. (2008)
Carbohydr. Res., 343, 196-210.
24.Lowry, O. H., Rosebrough, N. J., Farr, A. L., and
Randall, R. J. (1951) J. Biol. Chem., 193, 265-275.
25.Dubois, M., Gilles, K. A., Hamilton, J. K.,
Rebers, P. A., and Smith, F. (1951) Nature, 168, 167.
26.Safari, D., Dekker, H. A. T., Joosten, J. A. F.,
Michalik, D., Carvalho de Souza, A., Adamo, R., Lahmann, M., Sundgren,
A., Oscarson, S., Kamerling, J. P., and Snippe, H. (2008) Infect.
Immun., 76, 4615-4623.
27.Mammen, M., Choi, S.-K., and Whitesides, G. M.
(1998) Angew. Chem. Int. Ed., 37, 2754-2794.