REVIEW: Natural Antibodies to Glycans
N. V. Bovin
Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, ul. Miklukho-Maklaya 16/10, 117997 Moscow, Russia; fax: (495) 330-5592; E-mail: bovin@carb.ibch.ru
Received February 4, 2013; Revision received February 19, 2013
A wide variety of so-called natural antibodies (nAbs), i.e. immunoglobulins generated by B-1 cells, are directed to glycans. nAbs to glycans can be divided in three groups: 1) conservative nAbs, i.e. practically the same in all healthy donors with respect to their epitope specificity and level in blood; 2) allo-antibodies to blood group antigens; 3) plastic antibodies related to the first or the second group but discussed separately because their level changes considerably during diseases and some temporary conditions, in particular inflammation and pregnancy. Antibodies from the third group proved to be prospective markers of a number of diseases, whereas their unusual level (below or above the norm) is not necessarily the consequence of disease/state. Modern microarrays allowed the determination of the human repertoire, which proved to be unexpectedly broad. It was observed that the content of some nAbs reaches about 0.1% of total immunoglobulins. Immunoglobulins of M class dominate for most nAbs, constituting up to 80-90%. Their affinity (to a monovalent glycan, in KD terms) were found to be within the range 10–4-10–6 M. Antibodies to Galβ1–3GlcNAc (LeC), 4-HSO3Galβ1–4GalNAc (4′-O-SuLN), Fucα1–3GlcNAc, Fucα1–4GlcNAc, GalNAcα1–3Gal (Adi), Galα1–4Galβ1–4Glc (Pk), Galα1–4Galβ1–4GlcNAc (P1), GlcNAcα-terminated glycans, and hyaluronic acid should be noted among the nAbs revealed and studied during the last decade. At the same time, a kind of “taboo” is observed for a number of glycans: antibodies to LeX and LeY, and almost all gangliosides have not been observed in healthy persons. Many of the revealed nAbs were directed to constrained inner (core) part of glycan, directly adjoined to lipid of cell membrane or protein. The biological function of these nAbs remains unclear; for anti-core antibodies, a role of surveillance on appearance of aberrant, especially cancer, antigens is supposed. The first data related to oncodiagnostics based on quantitation of anti-glycan nAbs are reported.
KEY WORDS: glycans, glycan array, microchip, natural antibodiesDOI: 10.1134/S0006297913070109
Abbreviations: ELISA, enzyme-linked immunosorbent assay; Ig, immunoglobulin; nAbs, natural antibodies; PGA, printed glycan array; SA, suspension array; Sia, sialic acid residue; Su, sulfate; TACA, tumor-associated carbohydrate antigen.
Natural antibodies (nAbs) are produced mainly by a population of long
living CD5+ B-1 lymphocytes comprising 20-35% of overall
number of B-cells [1]. B-1 cells mature much
earlier than B-2 cells, during fetal life and several weeks after
birth. These cells do not disappear until the end of an
individual’s life. B-1 cells can response to immunization but
very restrictedly because they are excluded from somatic hypermutation
and affinity maturation, which B-2 cells undergo [1, 2]. The repertoire and
reactivity of nAbs are conservative not only within a species, but also
between species [3]; gene segments VJC appeared
about 430 million years ago. It was demonstrated that some nAbs are
autoantibodies executing important functions, in particular
anti-idiotypic blocking of pathogenic (e.g. to DNA) antibodies or
promotion of phagocytosis of an organism’s own cells subject to
elimination.
The studies cited above were dedicated mostly to antibodies to protein antigens. There are many fewer papers on the study of nAbs to glycans. Activity in this field increased drastically due to development of microarray technique allowing attaching several hundred glycans on a microchip (25 × 75 mm) and identification of the corresponding antibodies [4-10]. This review summarizes and discusses the data on anti-glycan nAbs obtained chiefly during the last decade.
Study of repertoire of nAbs to glycans performed on large groups of donors allows us to divide nAbs into three groups. The first group includes conservative nAbs that can be observed in all (or practically all) healthy donors; variation of their content, affinity, and epitope specificity is low, which is consistent with the cited general data on particularities of natural antibodies. The second group includes, first of all, allo-nAbs, i.e. antibodies to blood group antigens that for an obvious reason are absent in a considerable number of people (e.g. anti-A antibodies in blood group A individuals); their level varies greatly even within persons with the same blood group. At least five human blood systems have purely carbohydrate nature: ABH1, Lewis, Pp, Ii, and Forssman. Lack of any antigen of a given blood system in an individual is usually accompanied by production of cognate allo-nAbs. When used in a comprehensive glycoarray (more than 300 glycans), at a glance, profiles of antibodies from different individuals look quite different; however, removal of blood group antibody signals produces practically identical profiles, or, at least similar ones.
1 According to recent classifications this system is divided in two separate ones, AB and H. Indeed, blood of any human contains antibodies directed to present and already eliminated pathogens, but these antibodies are out of the scope of our discussion. Nevertheless, it is necessary to take into consideration borderline cases that are difficult to unambiguously classify. The difficulties arise first because it is impossible to reckon an individual to healthy group as there are no definite criteria for this. Second, some conditions, like pregnancy, must be considered separately. Thus, it is necessary to select a third group of nAbs (referred as plastic ones), whereto we reckon both conservative and blood group-specific antibodies, whose status is considerably changed during non-infectious diseases and some temporary conditions like pregnancy and inflammation. Importantly, these antibodies are prospective markers of a number of diseases, especially oncological diseases, which are one of the subject matters of this article.
The following generalizations can be made for anti-glycan nAbs of all the three groups [11]:
– the content of each top rank nAb is high, reaching about 0.5% of total immunoglobulins of M class;
– variation of their content in blood serum is small, within the range 1-10 μg/ml;
– affinity (to monovalent hapten, in KD terms) was found to be within the range 10–4-10–6 M, i.e. affinity interval (two orders of magnitude) is broader than mass content range (only one order of magnitude). In other words, the difference in observed titer is due to different affinity rather than antibody concentration in blood.
Immunoglobulins of M class usually dominate, comprising up to about 80%. Some exceptions were observed where IgG share exceeded 50%. In contrast to concentration of all Ig of particular specificity and their affinity, the ratio IgG/IgM/IgA varies greatly between donors; IgA is prevailing in some individuals.
CONSERVATIVE nAbs
Structures of glycans to which the largest titers of conservative natural antibodies were observed are given in Table 1. Notably, most of these nAbs were not described before the appearance of the microchip technique. Several conservative nAbs were isolated using hapten-specific chromatography (when a specific glycan is attached to Sepharose) [12-16], followed by detailed characterization of their epitope specificity, determination of activity, and level in blood.
Table 1. Glycans to which natural antibodies
were observed in all or almost all individuals [17, 20]

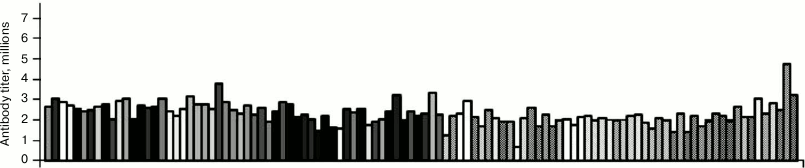
Antibodies to Galα1–6Glc are an example of minor titer variations. It can be seen in Fig. 1 that all 106 studied donors have these antibodies, and variation of their titers is small [17]. The use of the term titer should be explained. Titer is the product of antibody concentration multiplied by affinity, and the signal on a chip is proportional to this factor rather than to the antibody concentration.
Fig. 1. Relative titers of antibodies to disaccharide Galα1–6Glc. Data for 106 healthy donors are given; printed glycan array (PGA) [17].
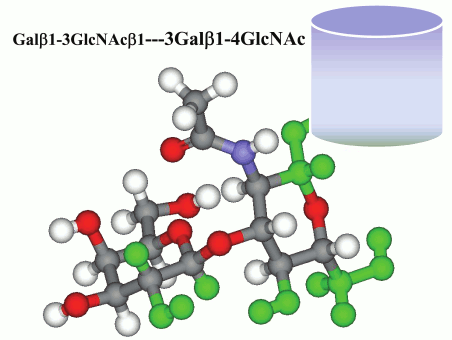
One of the highest binding levels was observed for antibodies to glycans LeC, Su-LeC, and SiaLeC. Detailed study demonstrated that in fact the same nAbs, insensitive to the nature of substituent R in 3-O-R-Galβ1–3GlcNAc, interacted with all the three glycans [12]. Interestingly, any long carbohydrate chain with a Galβ1–3GlcNAc fragment at the non-reducing end is totally inert to these nAbs (Fig. 2; see color insert).
Fig. 2. Molecular model of disaccharide LeC. Any “continuation” of the chain (shown as a semi-transparent cylinder) abolishes glycan interaction with anti-LeC nAbs.
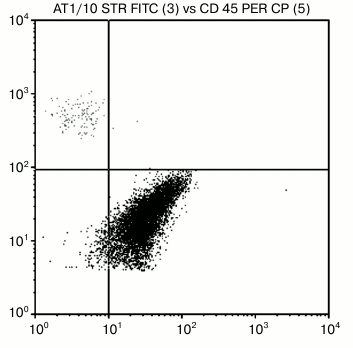
Therefore, these nAbs are incapable of interaction with normal natural carbohydrate chains where a LeC fragment is obligatorily attached to lactosamine core. At the same time, according to flow cytometry and histochemistry data these antibodies interact with breast cancer cells [11] (Fig. 3).
Fig. 3. Cytometric analysis of biotin-labeled human antibodies to disaccharide LeC demonstrates positive reaction with breast cancer cells (lower right quadrant) and lack of interaction with normal cells (upper left quadrant). The picture is taken from [11].
Though it remains unclear what binds with these antibodies in human tissues, the fact of discrimination between normal and tumor cells is notable as it opens up possibilities for diagnostics and therapy of cancer.
The nature of the true target for antibodies capable of binding to ligand 4-O-Su-Galβ1–4GlcNAc on glycochip is also unclear. A similar termination 4-O-Su-GalNAcβ1–4GlcNAc was observed in the composition of several human hormones [18], but affinity-isolated nAbs did not interact with 4-O-Su-GalNAcβ1–4GlcNAc. Thus an “extra” NAc group (underlined) abolishes the interaction. A relation of increased level of anti-4-O-Su-Galβ1–4GlcNAc with such diseases as systemic sclerosis and pulmonary hypertension was observed [19].
At a glance, human cells bear numerous targets for antibodies to disaccharides Fucα1–3GlcNAc and Fucα1–4GlcNAc, because Fucα1–3GlcNAc is a part of more complex glycans, such as SiaLeX, mediating leukocyte rolling during inflammation and some other processes of cell recognition. Nevertheless, these nAbs did not interact with either tetrasaccharide SiaLeX or trisaccharide LeX. According to their “track” of specificity, they could bind disaccharide fragment Fucα1–3GlcNAc only from the side where galactose residue is situated [20]. For the same reason, anti-Fucα1–4GlcNAc is incapable of interaction with either trisaccharide Lea or tetrasaccharide Leb.
The same explanation can be given for the fact of the lack of autoimmune reactions of the next four antibodies in Table 1, i.e. Adi, Bdi, “Galili”, and Hdi [20]. All four structures are regular motifs in the composition of blood group ABH antigens, i.e. they are the components of carbohydrate chains of all or at least large groups of people, but they do not interact with the discussed antibodies due to the finely programmed specificity of these nAbs. For example, specificity of anti-Adi antibodies allows interaction with free trisaccharide GalNAcα1–3(Fucα1–2)Gal, but in the case of the tetrasaccharide GalNAcα1–3(Fucα1–2)Gal-GlcNAc the GlcNAc residue prevents their access to the Adi-epitope [13].
It should be particularly noted that the level of antibodies specifically binding xeno-antigen Galα1–3Gal (and Galα1–3Galβ1–4GlcNAc) in human blood proved to be much lower than the values given in early original papers and numerous reviews and books referred to as 2% of total immunoglobulins. Exhaustive affinity isolation of these antibodies with thorough removal of all irrelevant similar specificities (many of them being present in human blood) gives the value of only 0.1%, i.e. the same as for many other anti-glycan nAbs [14] (see above).
Disaccharide fragment GlcAβ1–3GlcNAc with terminal glucuronic acid residue is the repeating unit of hyaluronic acid polysaccharide. However, whereas disaccharide GlcAβ1–3GlcNAc bound nAbs of all patients, interaction with its polymeric form was observed only in several individuals, so that anti-hyaluronic antibodies are related to the group referred herein as plastic nAbs. The real target for conservative antibodies found to bind GlcAβ1–3GlcNAc (and anti-GlcAβ1–6Gal) has not been revealed yet.
All oligosaccharides with a peripheral GlcNAcα residue demonstrated high levels of binding. Carbohydrate chains of this type are very rare in humans and other mammals, being found only in gastric mucin, seemingly only in the chain GlcNAcα1–4Galβ1–4GlcNAcβ1–6(GlcNAcα1–4Galβ1–3)GalNAcα-Ser/Thr [21]. Whether the blood nAbs are capable of recognizing just this glycan (or its essential fragment GlcNAcα1–4Galβ) remains unclear because only the fragments with structure motif GlcNAcα1–3(or 6)Galβ were present on the chip.
Antibodies to LacdiNAc, i.e. the disaccharide GalNAcβ1–4GlcNAc, seem to execute the classic for pre-existing antibodies protective function: this glycan is typical for a wide range of parasites, being extremely rare in human carbohydrate chains.
At a glance, the next three nAbs, i.e. interacting with Fs-2, P1, and Pk, should be placed in Table 2 rather than Table 1. In fact, it is incorrect to relate them to blood group-specific ones because all the studied individuals were found to have these antibodies. In the case of the Fs system, the pentasaccharide but not disaccharide is the true antigen [22]. Indeed, when studying a group of donors using an array containing both di- and pentasaccharide, it was observed that not all individuals had antibodies to pentasaccharide, which is typical for blood group antigens [20]. As pentasaccharide Fs has a unique compact conformation, all the five monosaccharide residues are included directly or mediately to its antigenic determinant, so disaccharide is by no means the equivalent of complete pentasaccharide despite its terminal position. Possibly, a similar situation is observed for P1 antigen, which is a pentasaccharide on the cell surface, whereas conservative antibodies are observed to its trisaccharide fragment (glycans of Pp group are discussed in more detail below).
CONSERVATIVE nAbs DIRECTED TO THE INNER PART OF GLYCANS
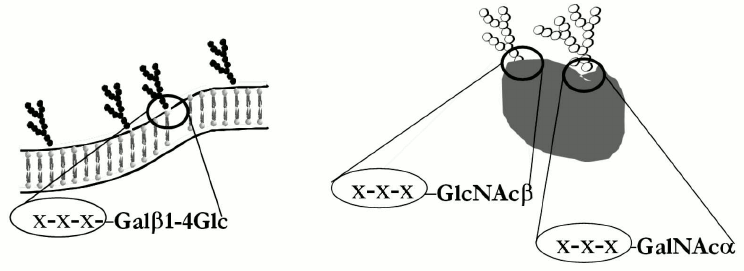
Despite considerable differences, the next ten glycans in Table 1 have a common structure motif, a lactose fragment at the reducing end (underlined in formulas). Some of the antibodies from this group were isolated using hapten-specific chromatography; epitope specificity of isolated material was studied in detail [20, 23]. It was found that the nAbs were tolerant to lactose core elongation, i.e. they interacted with a number of glycans having the formula R-Galβ1–4Glc even if substituent R was a large oligosaccharide. Nevertheless, there are cases when an R substituent prevents binding of nAbs to lactose fragment – first, when glycan conformation is compact as in case of globo-series of glycosphingolipids, and second, in the case of gangliosides where a sialic acid residue is the masking factor. It should be noted that these unusual antibodies are capable of recognizing the inner fragment of glycan only in cell-free (artificial) test systems. Figure 4 demonstrates a scheme of glycosphingolipid inserted in the membrane of a normal cell; its fragment –Galβ1–4Glc is completely masked, and possibly this is a reason why anti-core nAbs do not induce autoimmune reaction.
Fig. 4. Schematic presentation of glycosphingolipid regions in the composition of plasma membrane (left) and glycoproteins (right), to which “anti-core” antibodies are observed in human blood. Monosaccharide residues (shown as X-X-X) attached to core do not prevent the interaction of these unusual antibodies with core fragment.
The lower part of Table 1 (marked with gray) includes rather similar nAbs, i.e. antibodies to the inner residue GalNAcα of glycoprotein O-chains. There is no doubt that most antibodies capable of binding with Tn antigen (in particular, of being affinity isolated) are sensitive to substitution of GalNAc moiety. Thus, inhibition of affinity-isolated anti-Tn antibodies with disaccharides TF and SiaTn proceeds only to a limited extent [24]. Nevertheless, such antibodies exist; they are also found between monoclonal anti-Tn [25].
IS THERE ANY SIMILARITY BETWEEN THE GLYCANS TO WHICH ANTIBODIES
ARE NOT OBSERVED?
About half of PGA glycans demonstrate a negative result with human blood sera [17], this making a false impression of the total lack of antibodies to a number of glycans. Nevertheless, it would be incorrect to speak about absolute “veto” on some glycans as assay data are averaged. Remember that we are discussing the entire population rather than separate individuals. For example one of 106 donors had a notable (higher than the selected cut-off) level of antibodies to LeY [17]. So, it is more correct to speak about glycans to which most people do not have antibodies, or their level is very low. Moreover the concept of “healthy person” is a very illusive one, as a rule “healthy control” includes donors without the studied disease, not more. Table 2 includes the glycans to which the level of antibodies in the cohort of 106 donors was lower than the cut-off value (selected as 5% of the maximum observed binding on chip). Below, the term “antibodies are absent” will be used as follows: average value of signal across the cohort does not exceed the selected threshold (cut-off) value.
Table 2. Glycans to which antibody level (in
healthy donors) is close to background [17, 20]

Some generalizations can be made on the base of data from Table 2. Antibodies to glycans with core 2 including oligosaccharides Lex, SiaLex, and H (type 2) are absent in humans. These glycans are widely present on human endothelial cells, so antibodies against them appear only in pathological conditions, such as autoimmunity and oncological diseases. Interestingly, Table 2 includes even tetrasaccharide αGalLex, i.e. Galα1–3Galβ1–4(Fucα1–3)GlcNAc, despite the presence of xenoreactive epitope Galα1–3Gal in its structure.
Another regularity is observed: the lack of antibodies to the most sialylated glycans, both with 2–3 and 2–6, and 2–8 bonds. It follows from Table 1 that only two glycans are antibody-binding, namely, SiaLeC, where a sialic residue is not included in the epitope recognized by the natural antibodies (see above), and tumor-associated glycan SiaTn discussed below. An example is the pair GM1/asialoGM1, where the glycan of the ganglioside falls in the group of weakly binding ones (close to cut-off value), whereas glycan of the corresponding neutral glycosphingolipid is in the group of strongly binding entities. Thus Neu5Ac residue plays an antigen-masking role. Though antibodies to ganglioside GM1 have been revealed in healthy donors using ELISA and immuno-TLC [26], this is not contradictory to the data of chip assay, where a rather high cut-off value was selected.
Antibodies to N-chains of glycoproteins were not detected in blood [20]. Decoration of N-chain either with bisecting residue GlcNAc or core residue Fucα1–6 did not led to appearance of antigenicity, the corresponding examples being given in Table 2. Increase of antenna number and the presence of lactosamine repeats do not lead to antigenic structures. Moreover, even the shortened variants (this was demonstrated for bi-antennary chains), such as desialylated ones and even having terminal GlcNAc or Man, did not show binding with blood serum. The lack of binding on the array in this case does not prove strictly the lack of antibodies, because shortened bi-antennary chains are in the composition of all IgG species, and therefore can inhibit nAbs potentially capable of binding to similar bi-antennary chains on the array [17].
nAbs TO BLOOD GROUP ANTIGENS AND STRUCTURALLY RELATED
OLIGOSACCHARIDES
Visual evidence of the difference between nAb profiles from the first (conservative) and second (allo-) groups can be given by comparison of Fig. 1 and Fig. 5 (see color insert).
Fig. 5. Relative titers of typical allo-antibodies; data for each of 106 healthy donors are given [17]. a) Binding to blood group tetrasaccharide A (type 2). b) Binding to blood group tetrasaccharide i. c) Binding with blood group hexasaccharide I.
It is well known that the number of persons having blood group antigen A (i.e. the persons with phenotypes A and AB) on one hand and those not having this antigen (i.e. phenotypes B and O) differs insignificantly. As seen from Fig. 5a, the proportion of persons with and without anti-A antibodies (tall vs. short bars) is also comparable; intermediate cases can be seen as well, when antibodies are revealed but their titer is low.
In the case of the Ii system, erythrocytes of most donors bear branched chains (phenotype I), whereas linear chain variants (phenotype i) are very rare. Due to this distribution one can suppose the presence of nAbs anti-I in most donors, but it follows from Fig. 5 (b and c) that this is not the case. Persons of phenotype I rarely have nAbs anti-i for the reason [20] that the linear chain variant (i) is normally exposed on human embryo cells, so their presence should lead to maternal–fetal incompatibility.
Table 3 lists not only tetrasaccharides A and B of type 2, which in fact are allo-antigens of erythrocytes, but also corresponding trisaccharides. Until recently trisaccharides were considered as antigenic determinants of blood group antigens A and B because sera of blood group A individuals bind well trisaccharide B and vice versa. However it was revealed in [13] that tetra- rather than trisaccharides were the true antigenic determinants, and that the population of anti-A antibodies contained both nAbs specific to tetrasaccharide and trisaccharide. Only the first were capable of agglutinating erythrocytes, whereas anti-Atri nAbs were found to circulate in blood of both blood group B and (paradoxically) blood group A individuals. Notably, anti-A vs. anti-B appear to be “asymmetrical” antibodies: epitope specificity of anti-A is wider than of anti-B, and their titer is much lower, being close to the cut-off value of PGA. Low anti-A titer looks unexpected taking into account well-known avidity (ability for agglutination) of these antibodies towards erythrocytes.
Table 3. Human blood group antigens (and
related glycans) to which natural antibodies were observed [17, 20]
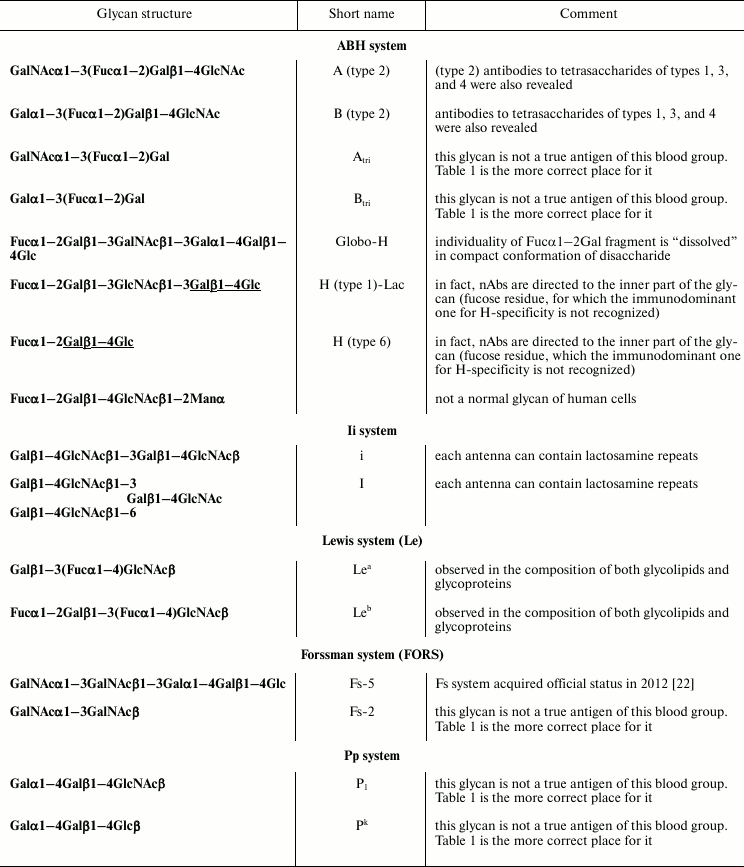
The same part of Table 3 lists glycans having a common structure motif Fucα1–2Gal, i.e. glycans of blood group H. It appeared that considerable level of nAbs binding was observed for a number of H-glycans – notably higher than that for A/B-tetrasaccharides. From general considerations, one can expect the presence of antibodies to H antigen is impossible, H antigens are widely expressed on all erythrocytes independently of blood group; they are known as biosynthetic precursors of antigens A and B, the biosynthesis never proceeds completely. The lack of autoimmunity reaction of revealed anti-H antibodies with autologous cells can be explained by their restricted epitope specificity. Indeed, glycan Fucα1–2Galβ1–3GalNAcβ1–3Galα1–4Galβ1–4Glc (globo-H) known to have a unique compact conformation (like all the other glycans of globo-series [27]) where the H-specific motif is “alloyed”, i.e. canonical anti-H antibodies are incapable of recognizing it; revealed in blood antibodies to globo-H recognize the whole hexasaccharide, analogously to described above true anti-Fs nAbs recognizing only Fs pentasaccharide. The next two structures in Table 3 have in their composition inner core fragment Galβ1–4Glc (Lac), allowing us to conclude that anti-core antibodies described above interact in the corresponding spots on the array rather than anti-H, which are indeed absent in humans. Finally, H-trisaccharide is attached to the mannose residue in glycan Fucα1–2Galβ1–4GlcNAcβ1–2Man, which does not occur in human glycoproteins; possibly antibodies revealed on the array recognize a unique compact epitope that is not an H-antigen – similar to the globo-H case.
Blood group system Lewis (two antigens, Lea and Leb) is similar to the Ii system by the level and distribution of antibodies in blood; only a few antibody-positive individuals were revealed in the cohort of 106 studied donors, and their titers are low. This mode of distribution is explained by low occurrence of phenotypes Lea(–) and Leb(–), so most individuals are carriers of one or even both antigens.
The most unexpected results were observed for antigens of the Pp system. Namely, all the donors of the studied cohort have antibodies of medium level titer [17] to trisaccharides Pk and P1, the pattern being similar to that given in Fig. 1. This looks strange because erythrocytes and endothelial cells of about 75% of humans express these antigens [28]. Antibodies isolated using hapten-specific chromatography on the corresponding trisaccharide-hapten proved to be specific exactly to trisaccharide, no interaction with Lac was observed, so they were not antibodies to core as described above. Intriguingly, the antibodies did not interact with a variety of natural glycolipids Gb3 where Pk trisaccharide is attached to different ceramides. Possibly, these unusual nAbs are identical to the so-called NOR antibodies of the first type that bind epitope Galα1–4Galβ1–3Gal-R in the composition of NOR glycosphingolipid Galα1–4GalNAcβ1–3Galα1–4Galβ1–4Glc [29]. Presumably, analogously to antibodies to LeC described above, anti-Pk nAbs capable of recognizing their cognate epitope from the “reducing” side are obscured by the cell membrane massive.
Anti-P1 do not bind human erythrocytes for apparently another reason. As in case of globo-H, a unique conformation of full-size glycan is important: trisaccharide is not a true antigenic determinant of P1-antigen, which is presented as a pentasaccharide on the cell surface. Low presented true anti-P1 are highly specific, i.e. they bind only pentasaccharide epitope. The biological significance of the presence of nAbs to trisaccharide P1 in all humans can be deduced from the recent data on their tumor-association [30], possibly antibodies to trisaccharide P1 have a function of surveillance on appearance of tumor cells.
Sometimes high variability between normal donors is observed for non-blood group specific antibodies, e.g. for nAbs to trisaccharide 3′SL, i.e. the glycan part of ganglioside GM3 presented in all humans. A surprise is that these formal antibodies are found in two thirds of healthy individuals [17].
Are there autoantibodies between anti-glycan nAbs? A specific publication was dedicated to discussion of this matter [20], describing antibodies producing high or medium signal on PGA (Table 1). As the result, a conclusion was made that due to various items we do not have sufficient reasons to relate any of them to autoantibodies; discussed above nAbs to disaccharides LeC, Fucα1–3GlcNAc, Fucα1–2Gal, GalNAcα1–3Gal, and core fragments of glycans belongs to this series. The key arguments are as follows: epitope specificity of the discussed human anti-glycan nAbs does not allow them to bind short (di-, trisaccharide) fragments, which are spatially masked in composition of natural more complicated molecules or molecular ensembles. Thus the results of analysis of human antibodies using PGA or other analytical approaches based on the use of small oligosaccharides (all the more monosaccharides) should be interpreted with caution taking into consideration real, natural context of a particular oligosaccharide. At the same time, a rather considerable group of antibodies can be related to auto-nAbs, which will be characterized below.
Anti-TACA
This section describes a group of antibodies related to plastic ones according to our classification (see introductory section). The ten lower lines in Table 1 are occupied by glycans related to tumor-associated carbohydrate antigens (TACA), associated rather than specific because they can be found in normal tissues though in another amount and another environment [31-33]. More than 50 TACA are mentioned in the literature that have been identified chemically or immunochemically in various tumors. About a dozen TACA are in “negative” Table 2, this meaning that antibodies to them are not observed in healthy (more exactly individuals without identified cancer) donors or observed sporadically in low titer. It should be noted that this is the case for cancer patients as well [34, 35]. At the same time, natural antibodies to some TACA have been observed and often in high titers [31], especially to core fragments of glycoprotein O-chains (marked with gray in Table 1). Interestingly, antibodies to them are formed not due to tumor progression, but they exist during the whole lifetime, so they are obviously related to nAbs. Particularly interesting is the fact that the level of nAbs to disaccharide TF and some other TACA in tumor patients is lower than in healthy individuals [31]. Data on the level and incidence of these nAbs in healthy donors obtained using PGA is basically consistent with the literature data. There is still no unambiguous answer whether it is possible to ascribe them to autoantibodies. On one hand, these antibodies interact with glycoprotein O-chains observed in normal tissues. On the other hand, their titer (affinity, concentration) can be insufficient for interaction with normal cells, whereas due to drastically increased concentration of the second component, antigen, in a tumor cell a threshold can be achieved when these antibodies become able to execute their function.
Immunological presentation of TACA on tumor cells, i.e. the molecular context, which can be considerably different for the same O-chain in normal versus transformed cells, remains almost unenlightened in the literature. The above data about fine-tuned epitope specificity of numerous anti-glycan nAbs is believed to be in concert with ideas about importance of immunological presentation of glycan antigens in tumor cells for their recognition/non-recognition by those nAbs. Yet, the presence of preexisting antibodies to some TACA assumes the possibility of diagnostics and prognostics of cancer by quantitation of the antibodies. Recent practice demonstrated the fruitfulness of this approach, discussed below.
CANCER DIAGNOSTICS BASED ON DETERMINATION OF nAbs LEVEL
Molecular diagnostics of cancer is very complicated for several reasons. First, a tumor is heterogeneous and can rapidly evolve. Second, patients are genetically non-identical, this affecting the nature of some tumor markers. An example is the classic glycoprotein marker of pancreatic cancer CA19.9, whose antigenic determinant is sialylated glycan LeA, not produced by individuals with Lea(–)Leb(–) phenotype. Third, tumor markers are not exclusively tumor-specific, their level changing during other diseases, this increasing the risk of false-positive tests. As a result, diagnostic sensitivity and specificity are inferior to those in infection diagnostics. The mentioned problems can be solved by the use of an approach based on simultaneous determination of several (up to ten and more) independent markers, the so-called “signature”. Revealing signatures looks promising, but it is not practical because determination of n markers requires n specific test systems. A solution is the use of anti-glycan nAbs as markers [36]; their potential number is large, and they are revealed by the same reagents under the same conditions, this making the use of Ig-signatures practical. It should be noted that the search for highly efficient diagnostic signatures is performed on large cohorts of patients by analysis of binding with several hundred glycans, so such investigation requires serious mathematics during the search study [37].
This approach proved to be prospective in diagnostics of malignancies such as breast cancer [36], cervix cancer [38], ovarian cancer [30], colorectal cancer [39, 40], and others [34, 35]. Unexpectedly, well-known TACA did not dominate between the glycans selected for diagnostic signatures. During the studies on the signature search just discussed, it was expected that the most suitable for diagnostics would be the following: 1) unique antibodies; 2) antibodies with low titer in healthy individuals, and 3) antibodies whose level increased during cancer progression. In fact, nAbs from a conservative group often become diagnostically valuable, e.g. anti-P1 [30], anti-Adi [38], anti-LeC [11], and anti-Bdi [41], which are detected in all healthy and diseased individuals in high titer. Moreover, about half of the diagnostically valuable nAbs demonstrate decreased rather than increased level in cancer patients. In total, these data allows us to suppose that some nAbs to glycans play a surveillance role, taking part in constant elimination of malignant cells [17, 42]. At least in the case of breast cancer, it was found that the level of antibodies to LeC in blood was inverse to the amount of the corresponding antigen revealed with isolated nAbs in the same group of patients [11].
The reasons and mechanisms for different antibody repertoire in oncological patients are poorly studied, and therefore their investigation is of obvious interest. We suppose that one of the reasons is failure in production of nAbs (IgM), whose level in patients was found to be reduced compared to norm. These nAbs possibly execute the function of surveillance on sporadically formed transformed aberrantly glycosylated cells. The second reason is classic immune response to appearing of TACA; it can be supposed that antibodies both promoting and preventing tumor development are of diagnostic value [43]. The third possible reason is the recently found process of synthesis and secretion by tumor cells of non-classic IgG capable of protecting the tumor from the immune system [44].
METHODS OF nAbs STUDY
Several methods are used for the study of the antibody repertoire in blood. Besides two-dimensional glycochip PGA [5] cited most often in this review, three methods must be mentioned: 3D-glycochip [45], suspension array (SA) [46], and ELISA [47]. Each of these methods has its own application niche only partially covered with other methods; publication [48] is dedicated to direct experimental comparison of three of them. Obviously, PGA is most suitable for screening experiments allowing us in its latest version to study binding of blood serum with 600 glycans simultaneously [9]. The “capacity” of three other methods is inferior to this. Another advantage of PGA is low background, allowing working with even undiluted sera [49]. Finally PGA has a wide dynamic range, i.e. it is possible to quantitate both low and high antibody titers. A unique feature of 3D-chip is spatial (in hydrogel) arrangement of glycan resulting in orders of magnitude higher signal intensity and increased precision, so it is possible to decrease the number of repeats on a chip. Additionally, using a 3D-chip it is possible to immobilize glycomolecules of different nature, such as oligosaccharides, polysaccharides, glycoproteins, and neoglycoconjugates in the same conditions and with the same efficiency.
In contrast to other methods, glycans in SA are immobilized on microparticles; this method and ELISA are convenient because of the possibility of easy reconfiguration of the test system. The main advantage of SA is small volume of test sample, this being particularly valuable during work with blood sera of infants or small laboratory animals.
ELISA is inferior to chips and SA in dynamic range and background value, but in contrast to other approaches it is still an available routine method. However, even in when other methods are available, ELISA is on demand because it allows easy performance of experiments on inhibition, dose dependence, and solution of other problems requiring good flexibility of the method.
Another important aspect of method selection is the ratio (stoichiometry) antigen/antibody. In most cases a researcher does not take this in mind using the conditions recommended by a device manufacturer or prior literature and optimizing the conditions empirically. This is wrongful in some cases because during the work with polyclonal antibodies heterogeneous by epitope specificity, affinity, and class (IgM/IgG/IgA), there is a risk of incorrect interpretation of the result without consideration of stoichiometry. Particularly in the case of considerable antigen excess, all antibodies of a given specificity have the possibility of binding with cognate antigen. As a result, the determined IgG/IgM ratio is close to reality, but information about affinity is lost under these conditions. Oppositely, in the case of great excess of antibodies only those with the highest affinity are determined due to competition, but IgG/IgM ratio becomes distorted. For this reason the results of nAbs (remember, polyclonal, heterogeneous ones) analysis by different methods can either coincide or not coincide [48]. PGA conditions include low amount of antigen and low dilution. As the result when working with blood serum the antibody/cognate glycan ratio is close to equivalence, or there are more antibodies. Not surprisingly, in the case of different serum dilutions in PGA the ratio of some signals changes, namely of those glycans to which immunoglobulins of G class are prevailing [49]. In the case of the other three methods (SA, 3D-chip, and ELISA), especially for the latter, the antigen/antibody ratio is the inverse, i.e. in favor of antigen.
Risk of incorrect interpretation of results. Risk of incorrect interpretation of results arises during determination of polyclonal antibody specificity and, particularly, during work with blood serum independently of the method used. It was already mentioned that a great number of the studied nAbs was directed to the inner part of a glycan, i.e. it was capable of binding with a small part of a large molecule. Taking this into consideration, it is possible to explain some seemingly paradoxical results. For example, analysis of a large cohort of donors demonstrates that nAbs to the tetrasaccharide Fucα1–2Galβ1–4(Fucα1–3)GlcNAc (LeY) were absent in humans, but, surprisingly, considerable binding with hexasaccharide LeY, i.e. Fucα1–2Galβ1–4(Fucα1–3)GlcNAcβ1–3Galβ1–4Glc, was observed in most donors [17]. After isolation on a column with immobilized hexasaccharide and application of the isolated antibodies, on PGA interaction not only with hexasaccharide but also with its inner fragment Galβ1–4Glc was revealed. The lessons of hapten-specific chromatography on a number of adsorbents [12, 23, 50] give evidence that this situation is typical. Thus, primary data on blood serum interaction with glycans on PGA must be interpreted taking into consideration quite possible “false positive” binding of competing antibodies.
Can false-negative interpretation of results occur? At least one example of this can be given [51]. As mentioned before, the glycochip did not reveal antibodies to most glycosides including Neu5Acα2–8Neu5Acα2–3Galβ1–4Glc, a glycan of ganglioside GD3, when the binding level is less than the cut-off value. However, in the case of analysis of intravenous immunoglobulin preparation (IvIg is total IgG isolated from plasma of thousands or more healthy donors) the signal of tetrasaccharide GD3 on a chip appears at the top of the list. It seems that antibodies to GD3 are present in blood serum, but they are blocked with ganglioside, and thus cannot be detected on PGA. During IvIg purification, namely, precipitation with ethanol, the complex is decomposed and antibodies to GD3 become unmasked.
The study of natural anti-glycan antibodies is at a stage of rapid progress. In outline, it is already possible to discuss the profile of the whole population of human antibodies. A large number of specificities were revealed, completely obscured several years ago; a class of unusual antibodies directed to the inner (core) part of carbohydrate chain was discovered; the application of the obtained basic knowledge has been started for cancer diagnostics. Some of the performed studies remain out of the scope of this review, as the data has not been considered enough. This includes the study of antibody dynamics in infants during the first moths of life [52], comparison of nAbs profiles in humans and laboratory animals, and their change after transplantation [53]. At the same time, many questions are still open, first of all about biological importance of the identified natural antibodies.
The author would like to express his sincerest acknowledgements to N. V. Shilova for valuable discussion of the material of this publication and I. M. Belyanchikov for the help with manuscript preparation.
This work was supported in part by the Russian Academy of Sciences Presidium Program “Molecular and Cell Biology”.
REFERENCES
1.George, J., and Shoenfeld, Y. (1996) in
Autoantibodies (Peter, J. B., and Shoenfeld, Y., eds.) Elsevier,
pp. 534-539.
2.Kaveri, S. V., Silverman, G. J., and Bayry, J.
(2012) J. Immunol., 188, 939-945.
3.Avrameas, S. (1991) Immunol. Today,
12, 154-159.
4.Galanina, O. E., Mecklenburg, M., Nifantiev, N. E.,
Pazynina, G. V., and Bovin, N. V. (2003) Lab. Chip, 3,
260-265.
5.Blixt, O., Head, S., Mondala, T., Scanlan, C.,
Huflejt, M. E., Alvarez, R., Bryan, M. C., Fazio, F., Calarese, D.,
Stevens, J., Razi, N., Stevens, D. J., Skehel, J. J., van Die, I.,
Burton, D. R., Wilson, I. A., Cummings, R., Bovin, N., Wong, C.-H., and
Paulson, J. C. (2004) Proc. Natl. Acad. Sci. USA, 101,
17033-17038.
6.Pochechueva, T., Jacob, F., Goldstein, D. R.,
Huflejt, M. E., Chinarev, A., Caduff, R., Fink, D., Hacker, N., Bovin,
N. V., and Heinzelmann-Schwarz, V. (2011) Glycoconj. J.,
8-9, 507-517; PMID: 21948103.
7.Alvarez, R. A., and Blixt, O. (2006) Methods
Enzymol., 415, 292-310.
8.Shilova, N. V., Navakouski, M. J., Huflejt, M.,
Kuehn, A., Grunow, R., Blixt, O., and Bovin, N. V. (2011)
Biochemistry (Moscow), 76, 862-866.
9.The Functional Glycomics Gateway, www.functionalglycomics.org
10.Park, S., Gildersleeve, J. C., Blixt, O., and
Shin, I. (2013) Chem. Soc. Rev., DOI: 10.1039/C2CS35401B.
11.Tupitsyn, N. N., Udalova, Y. A., Galanina, O. E.,
Kadagidze, Z. G., Borovkova, N. B., Podolsky, V. V., Shinkarev, S. A.,
Gadetskaya, N. A., Letyagin, V. P., Obukhova, P. S., Shilova, N. V.,
Subbotina, A. A., and Bovin, N. V. (2009) Hematopoiesis
Immunol., 2, 45-54.
12.Obukhova, P., Piskarev, V., Severov, V.,
Pazynina, G., Tuzikov, A., Navakouski, M., Shilova, N., and Bovin, N.
(2011) Glycoconj. J., 8-9, 501-505; PMID: 22057658.
13.Obukhova, P., Korchagina, E., Henry, S., and
Bovin, N. (2011) Transfusion, 52, 860-869; PMID:
21981750.
14.Obukhova, P., Rieben, R., and Bovin, N. (2007)
Xenotransplantation, 14, 627-635.
15.Korchagina, E. Y., Pochechueva, T. V., Obukhova,
P. S., Formanovsky, A. A., Imberty, A., Rieben, R., and Bovin, N. V.
(2005) Glycoconj. J., 22, 127-133.
16.Smorodin, E. P., Jansson, B., Milyukhina, L.,
Paaski, G., Bovin, N. V., Ovchinnikova, T. V., and Kurtenkov, O. (1997)
Rus. J. Bioorg. Chem., 23, 718-721.
17.Huflejt, M. E., Vuskovic, M., Vasiliu, D., Xu,
H., Obukhova, P., Shilova, N., Tuzikov, A., Galanina, O., Arun, B., Lu,
K., and Bovin, N. (2009) Mol. Immunol., 46,
3037-3049.
18.Frete, D., Srivastava, V., Hindsgaul, O., and
Baenziger, J. U. (1991) Cell, 67, 1103-1110.
19.Grader-Beck, T., Boin, F., von Gunten,
S., Smith, D., Rosen, A., and Bochner, B. S. (2011) Ann.
Rheum. Dis., 70, 2218-2224.
20.Bovin, N., Obukhova, P., Shilova, N., Rapoport,
E., Popova, I., Navakouski, M., Unverzagt, C., Vuskovic, M., and
Huflejt, M. (2012) Biochim. Biophys. Acta, Gen. Subj.,
1820, 1373-1382; DOI: 10.1016/j.bbagen.2012.02.005.
21.Ishihara, K., Kurihara, M., Goso, Y., Urata, T.,
Ota, H., Katsuyama, T., and Hotta, K. (1996) Biochem. J.,
318, 409-416.
22.Svensson, L., Hult, A. K., Stamps,
R., Angstrom, J., Teneberg, S., Storry, J.
R., Jorgensen, R., Rydberg, L., Henry, S. M.,
and Olsson, M. L. (2012) Blood, Published ahead of print
December 18, 2012, doi: 10.1182/blood-2012-10-455055.
23.Obukhova, P. S. (2012) Specificity of Natural
Human Anti-carbohydrate Antibodies in the Norm: Candidate of
Science Thesis [in Russian], Shemyakin Institute of Bioorganic
Chemistry, RAS, Moscow.
24.Smorodin, E. P., Kurtenkov, O. A., Sergeyev, B.
L., Pazynina, G. V., and Bovin, N. V. (2004) Glycoconj. J.,
20, 83-89.
25.Blixt, O., Lavrova, O. I., Mazurov, D. V., Clo,
E., Kracun, S. K., Bovin, N. V., and Filatov, A. V. (2011)
Glycobiology, 22, 529-542.
26.Mizutamari, R. K., Wiegandt, H., and
Nores, G. A. (1994) J. Neuroimmunol., 50, 215-220.
27.Grounberg, G., Nilsson, U., Bock, K., and
Magnusson, G. (1994) Carbohydr. Res., 257, 35-54.
28.Bailly, P., and Bouhours, J.-F. (1995) in
Blood Cell Biochemistry, Vol. 6 (Cartron, J.-P., and Rouger, P.,
eds.) Plenum Press, New York, pp. 299-329.
29.Duk, M., Kuznierz-Alejska, G., Korchagina, E. Y.,
Bovin, N. V., Bochenek, S., and Lisowska, E. (2005)
Glycobiology, 15, 109-118.
30.Jacob, F., Goldstein, D. R., Huflejt, M., Bovin,
N., Pochechueva, T., Spengler, M., Caduff, R., Fink, D., and
Heinzelmann-Schwarz, V. (2012) Int. J. Cancer, 130,
138-146; DOI: 10.1002/ijc.26002.
31.Springer, G. F. (1984) Science,
224, 1198-1206.
32.Hakomori, S. (1984) Annu. Rev. Immunol.,
2, 103-126.
33.Lloyd, K. O. (1991) Semin. Cancer Biol.,
2, 421-431.
34.Huflejt, M. E., Blixt, O., Vuskovic, M., Xu, H.,
Shaw, L., Reuben, J. M., Kuerer, H., and Cristofanilli, M. (2005)
Br. Cancer Res. Treat., 94 (Suppl. 1), S85.
35.Arun, B., Vuskovic, M., Vasiliu, D., Xu, H.,
Atchley, D., Chambers, J., Bovin, N. V., Sneige, N., Hortobagyi, G. N.,
and Huflejt, M. E. (2007) Br. Cancer Res. Treat., 106
(Suppl. 1), S180.
36.Bovin, N. V., and Huflejt, M. E. (2008) Trends
Glycosci. Glycotechnol., 20, 245-258.
37.Vuskovic, M., Xu, H., Bovin, N., Pass, H., and
Huflejt, M. (2011) Int. J. Bioinformatics Res. Appl., 7,
402-426.
38.Li, Q., Anver, M. R., Li, Z., Butcher, D. O., and
Gildersleeve, J. C. (2010) Int. J. Cancer, 126,
459-468.
39.Pedersen, J. W., Blixt, O., Bennett, E. P., Tarp,
M. A., Dar, I., Mandel, U., Poulsen, S. S., Pedersen, A. E., Rasmussen,
S., Jess, P., Clausen, H., and Wandall, H. H. (2011) Int. J.
Cancer, 128, 1860-1871; DOI: 10.1002/ijc.25778.
40.Butvilovskaya, V. I., Popletaeva, S. B.,
Chechetkin, V. R., Zubtsova, Z. I., Tsybulskaya, M. V., Filippova, M.
A., Samokhina, L. O., Vinnitskii, L. I., Ragimov, A. A., Grigoryeva, G.
A., Meshalkina, N. Yu., Golysheva, S. V., Shilova, N. V., Bovin, N. V.,
Zasedatelev, A. S., and Rubina, A. Y. (2013), in preparation.
41.Smorodin, J. P., Kurtenkov, O. A., Miljukhina, L.
M., Sergeyev, B. L., Hint, E. K., Bovin, N. V., Lipping, A. A., and
Chuzhmarov, V. J. (1997) Exp. Oncol., 4, 338-342.
42.Vollmers, H. P., and Brandlein, S. (2007) J.
Autoimmun., 29, 295-302.
43.Newsom-Davis, T. E., Wang, D., Steinman, L.,
Chen, P. F., Wang, L. X., Simon, A. K., and Screaton, G. R. (2009)
Cancer Res., 69, 2018-2025.
44.Qiu, X., Zhu, X., Zhang, L., Mao,
Y., Zhang, J., Hao, P., Li, G., Lu, P., Li,
Z., Sun, X., Wu, L., Zheng, J., Deng, Y., Hou,
C., Tang, P., Zhang, S., and Zhang, Y. (2003) Cancer
Res., 63, 6488-6495.
45.Dyukova, V. I., Dementieva, E. I., Zubtsov, D.
A., Galanina, O. E., Bovin, N. V., and Rubina, A. Y. (2005) Anal.
Biochem., 347, 94-105.
46.Pochechueva, T., Chinarev, A., Spengler, M.,
Korchagina, E., Heinzelmann-Schwarz, V., Bovin, N., and Rieben, R.
(2011) Analyst, 136, 560-569.
47.Shilova, N. V., Galanina, O. E., Pochechueva, T.
V., Chinarev, A. A., Kadykov, V. A., Tuzikov, A. B., and Bovin, N. V.
(2005) Glycoconj. J., 22, 43-51.
48.Pochechueva, T., Jacob, F., Goldstein, D. R.,
Huflejt, M. E., Chinarev, A., Caduff, R., Fink, D., Hacker, N., Bovin,
N. V., and Heinzelmann-Schwarz, V. (2011) Glycoconj. J.,
8-9, 507-517.
49.Shilova, N. V., Navakouski, M. J., Khasbiullina,
N., Blixt, O., and Bovin, N. V. (2012) Glycoconj. J., 29,
87-91; DOI: 10.1007/s10719-011-9368-8; PMID: 22258790.
50.Butschak, G., and Karsten, U. (2002) Tumour
Biol., 23, 113-122.
51.Shilova, N., Huflejt, M. E., Vuskovic, M.,
Obukhova, P., Navakouski, M., Khasbiullina, N., Pazynina, G., Galanina,
O., Bazhenov, A., and Bovin, N. (2013) in Topics Curr. Chem.
Sialoglyco Chemistry and Biology (Gerardy-Schahn, R., Dellanoy,
P., and von Itzstein, M., eds.) Springer, in press.
52.Khasbiullina, N. R., Shilova, N. V., Navakouski,
M. J., Timofeeva, L. A., Kumar, S., Schwartz-Albiez, R., Sorensen, R.
U., Inostroza, J., Bovin, N. V., and Blixt, O. (2012) in 5th Baltic
Meet. on Microbial Carbohydrates, Program & Abstracts,
Institute of Organic Chemistry, Moscow, p. O26.
53.Blixt, O., Kumagai-Braesch, M., Tibell, A.,
Groth, C. G., and Holgersson, J. (2009) Am. J. Transplant.,
9, 83-90.