Characterization of Iron–Sulfur Cluster Assembly Protein IscA from Acidithiobacillus ferrooxidans
Lin Qian, Chunli Zheng, and Jianshe Liu*
College of Environmental Science and Engineering, Donghua University, Shanghai, 201620, China; fax: 86-21-67792522; E-mail: Liujianshe@dhu.edu.cn; qianlin_2012@hotmail.com; zhengchunli1979@163.com* To whom correspondence should be addressed.
Received September 23, 2012; Revision received December 3, 2012
IscA is a key member of the iron–sulfur cluster assembly machinery found in bacteria and eukaryotes, but the mechanism of its function in the biogenesis of iron–sulfur cluster remains elusive. In this paper, we demonstrate that Acidithiobacillus ferrooxidans IscA is a [4Fe-4S] cluster binding protein, and it can bind iron in the presence of DTT with an apparent iron association constant of 4·1020 M–1. The iron binding in IscA can be promoted by oxygen through oxidizing ferrous iron to ferric iron. Furthermore, we show that the iron bound form of IscA can be converted to iron–sulfur cluster bound form in the presence of IscS and L-cysteine in vitro. Substitution of the invariant cysteine residues Cys35, Cys99, or Cys101 in IscA abolishes the iron binding activity of the protein; the IscA mutants that fail to bind iron are unable to assemble the iron–sulfur clusters. Further studies indicate that the iron-loaded IscA could act as an iron donor for the assembly of iron–sulfur clusters in the scaffold protein IscU in vitro. Taken together, these findings suggest that A. ferrooxidans IscA is not only an iron–sulfur protein, but also an iron binding protein that can act as an iron donor for biogenesis of iron–sulfur clusters.
KEY WORDS: Acidithiobacillus ferrooxidans, IscA, IscU, iron–sulfur clusterDOI: 10.1134/S000629791303005X
Iron–sulfur (Fe-S) clusters are one of the most ancient and ubiquitous protein prosthetic groups in nature [1]. Throughout evolution, iron–sulfur clusters exhibit diverse functions such as electron transport, redox and non-redox catalysis, DNA synthesis and repair, and the regulation of gene expression [2-4]. Iron and sulfide are toxic for cells in concentrations for in vivo iron–sulfur protein maturation. Therefore, cells have developed different mechanisms for cellular iron–sulfur cluster assembly. Three distinct biosynthesis systems have been found in bacteria: ISC, SUF, and NIF machineries. The ISC and SUF systems are widely distributed, where is ISC expressed in normal physiological conditions, and SUF is expressed in iron limitation and/or oxidation stress. The NIF system is used to assemble complex or specialized clusters for a specific enzyme [5, 6].
Three iscSUA operon genes iscS, iscU, and iscA have been targets of recent investigations. Biochemical studies indicated that IscS is a pyridoxal phosphate (PLP) dependent cysteine desulfurase, which catalyzes desulfurization of L-cysteine and transfers sulfur for iron–sulfur cluster assembly in scaffold proteins [7, 8]. IscU acts as an iron–sulfur cluster assembly scaffold protein and transfers the assembled iron–sulfur clusters to target proteins [9, 10]. The exact role of IscA in the biogenesis of iron–sulfur proteins remains controversial. Two hypotheses concerning the biochemical function of IscA have recently emerged. The first states that IscA is an alternative scaffold protein for iron–sulfur cluster assembly, which could host transient iron–sulfur cluster and transfer the assembled clusters to target proteins in vitro [11, 12]. The second hypothesis is that IscA acts as an iron chaperone that binds free iron, and the iron center in IscA could be mobilized by L-cysteine for iron–sulfur cluster assembly in IscU [13-15]. The relative iron binding or iron–sulfur cluster binding activities of IscA needs to be further investigated.
In our previous studies, we have revealed that purified Acidithiobacillus ferrooxidans IscA can be overexpressed and purified with a [4Fe-4S] cluster in E. coli, and the apo-IscA could serve as a scaffold protein that recruits intracellular iron and sulfur to host a [4Fe-4S] cluster in vitro [16]. Here we report A. ferrooxidans IscA is an iron binding protein with an apparent iron association constant of 4·1020 M–1, and that the iron binding in IscA can be promoted by oxygen through oxidizing ferrous iron to ferric iron. Furthermore, we show that the iron bound form IscA can be converted to iron–sulfur cluster bound form in the presence of IscS and L-cysteine in vitro. Additional studies reveal that the iron-loaded IscA can provide iron for the iron–sulfur cluster assembly in the scaffold protein IscU in vitro. These results suggest that A. ferrooxidans IscA is not only an iron–sulfur protein, but also an iron binding protein that may act as an iron donor for biogenesis of iron–sulfur clusters.
MATERIALS AND METHODS
Protein preparation. Recombinant A. ferrooxidans IscA [16], IscU [17], and IscS [18] were produced as described previously. In short, they were expressed as fusion proteins with a His-tag and purified by affinity chromatography using Ni-NTA agarose gel (Qiagen, Germany). Apo-IscA was prepared by incubating purified IscA with EDTA (10 mM) and L-cysteine (2 mM) at 37°C for 60 min, followed by passing through a HiTrap desalting column (GE Healthcare, USA). The purity of all the purified proteins was greater than 95% as judged by electrophoretic analysis on a 15% (w/v) polyacrylamide gel containing SDS followed by staining with Coomassie Blue. The amounts of the acid-labile iron and sulfide in the protein samples were analyzed according to Fischer’s method [19] and Siegel’s method [20], respectively.
Iron binding analysis in IscA. Apo-IscA (100 µM) was incubated with various concentrations of ferrous iron or ferric iron in the presence of 2 mM DTT in open-to-air microtubes at room temperature (25°C) for 30 min. The protein samples then passed through a HiTrap desalting column to remove unbound iron and DTT. For anaerobic conditions, all reaction solutions were purged with pure argon gas before use. The relative iron binding in IscA was measured by the absorption amplitude at 315 nm [21].
Reconstitution of iron–sulfur cluster in IscA. Iron-loaded IscA (100 µM) was incubated with 1 μM IscS and 2 mM DTT in buffer containing 500 mM NaCl and 20 mM Tris-HCl (pH 8.0). The reaction mixtures were purged with pure argon gas and preincubated at 37°C for 5 min before 1 mM L-cysteine was added to initiate the iron–sulfur cluster assembly reaction under anaerobic conditions. IscA was re-purified from the incubation solutions and subjected to UV-visible absorption measurements. The amount of the iron–sulfur clusters assembled in IscA was monitored at 315 and 415 nm.
Iron–sulfur cluster assembly of IscU in vitro. Purified IscU (50 μM) was incubated anaerobically with 1 μM IscS, 2 mM DTT, and 1 mM L-cysteine in buffer containing 500 mM NaCl and 20 mM Tris-HCl (pH 8.0). Either Fe(NH4)2(SO4)2 or iron-loaded IscA was added as iron source for the assembly of iron–sulfur cluster in IscU. The amount of iron–sulfur clusters assembled in IscU was monitored at 456 nm [22].
UV-visible scanning and EPR spectra. UV-visible spectra were scanned at 25°C in a Beckman DU-640 UV-visible spectrometer equipped with a temperature controller. EPR spectra were recorded at 100 K on a JEOL JES-FEIXG spectrometer. The routine EPR conditions were: microwave frequency, 9.154 GHz; microwave power, 4 mW; modulation frequency, 100 kHz; modulation amplitude, 0.63 mT; sample temperature, 4.0 K; and receive gain, 1×105.
RESULTS
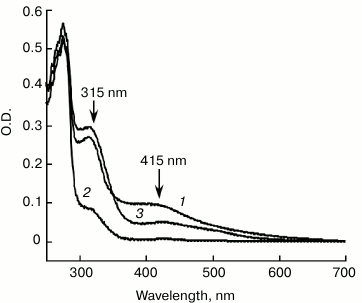
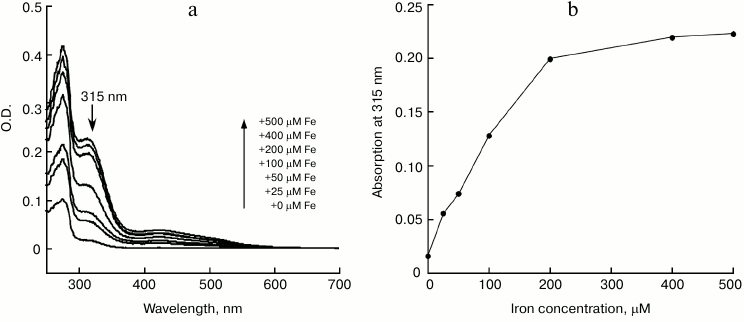
IscA acts as an iron-binding protein. The purified IscA was brown in color, with a clear absorption peak at 315 nm and a shoulder at 415 nm (Fig. 1, spectrum 1), indicating iron–sulfur cluster binding in the protein. Analysis of total iron and sulfide contents in purified IscA showed that there were 3.56 ± 0.10 Fe and 3.78 ± 0.12 S atoms per IscA monomer. This result suggested that IscA contained a single [4Fe-4S] cluster per monomer. When purified IscA was incubated with L-cysteine and the iron chelator EDTA, the bright color disappeared quickly. Analysis of the prepared apo-IscA showed that the protein had little absorption at 315 and 415 nm (Fig. 1, spectrum 2) and containing less than 0.2 Fe and S atoms per apo-IscA monomer. However, when the apo-IscA was re-incubated with ferrous iron in the presence of DTT, the absorption peak at 315 nm was restored (Fig. 1, spectrum 3). To check the iron binding activity of IscA, the apo-IscA was incubated with various concentrations of ferrous iron in the presence of DTT, followed by re-purification of IscA. Figure 2 shows the iron-binding curve of apo-IscA. As the iron concentration in the incubation solution was increased, the absorption peak at 315 nm of IscA was proportionally increased and apparently saturated when the iron concentration was about 4 times of the IscA concentration. Analysis of total iron and sulfide contents in iron-loaded IscA showed that there were 3.81 ± 0.13 Fe and less that 0.2 S atoms per iron-loaded IscA monomer. These results suggested that iron, not iron–sulfur cluster, was binding in IscA after incubation apo-IscA with ferrous iron and DTT. Using sodium citrate as a competing iron chelator, we found that IscA had an apparent iron association constant of 4.0·1020 M–1, which is higher than that of E. coli IscA (3.0·1019 M–1) [22, 23] and human IscA (2.0·1019 M–1) [14].
Fig. 1. UV-visible spectra of IscA: 1) the wild-type IscA (100 μM) was purified from E. coli cells grown in LB medium; 2) the apo-IscA (100 μM) was prepared by incubating purified IscA with EDTA (10 mM) and L-cysteine (2 mM) at 37°C for 30 min; 3) the iron-loaded IscA (100 µM) was prepared by incubating apo-IscA with 10-fold molar excess of ferrous ammonium sulfate and 2 mM DTT. The protein was re-purified by passing through a HiTrap desalting column and subjected to UV-visible absorption measurements.
Fig. 2. Iron binding in apo-IscA. a) UV-visible absorption spectra of apo-IscA after reconstitution. Apo-IscA (50 μM) was incubated with Fe(NH4)2(SO4)2 (0-500 μM) in the presence of DTT (2 mM) in open-to-air microtubes at room temperature (25°C) for 30 min. The protein was re-purified by passing through a HiTrap desalting column. b) Iron binding curve of apo-IscA. Amplitudes of the absorption peak at 315 nm of apo-IscA were obtained from the spectra in (a) and plotted as a function of the ferrous iron concentration in the incubation solution.
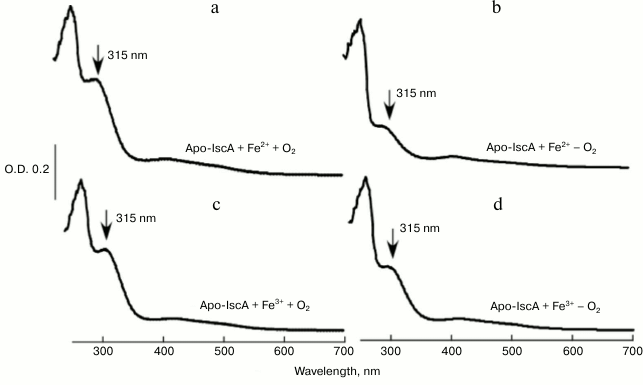
Oxygen promotes iron binding of IscA by oxidizing ferrous iron to ferric iron. To obtain deeper insight into the physiological iron binding activity of IscA, 50 μM apo-IscA was incubated anaerobically with 10-fold molar excess of Fe(NH4)2(SO4)2 in the presence of DTT; the reaction solutions were purged with pure argon gas separately and mixed together. The reaction solutions were incubated at room temperature (25°C) for 30 min followed by re-purification of IscA. Figure 3 shows that compared with aerobic conditions (Fig. 3a), there was a small absorption peak at 315 nm under anaerobic conditions (Fig. 3b), indicating that ferrous iron binding of IscA was greatly inhibited under anaerobic conditions. The data suggests that oxygen plays a crucial role in iron binding of IscA. To test our hypothesis, we incubated 50 μM apo-IscA with 10-fold molar excess of FeCl3 in the presence of DTT as described previously, and we found that after incubation the apo-IscA was converted to iron-loaded IscA both under aerobic (Fig. 3c) and anaerobic conditions (Fig. 3d). Therefore, we propose that oxygen could promote the iron binding of IscA by oxidizing ferrous iron to ferric iron in the iron-binding site of IscA [21].
Fig. 3. Iron-binding activity of IscA in vitro under aerobic (a, c) and anaerobic (b, d) conditions. a) Apo-IscA (50 μM) was incubated with 10-fold molar excess of Fe(NH4)2(SO4)2 and DTT (2 mM) under aerobic conditions at 25°C for 30 min, and IscA was re-purified from the incubation solutions and subjected to UV-visible absorption measurements; b) same as in (a) with the exception that the protein was incubated under anaerobic conditions; c) same as in (a) with the exception that FeCl3 was used instead of Fe(NH4)2(SO4)2; d) same as in (a), except that FeCl3 was used instead of Fe(NH4)2(SO4)2, and the protein was incubated under anaerobic conditions.
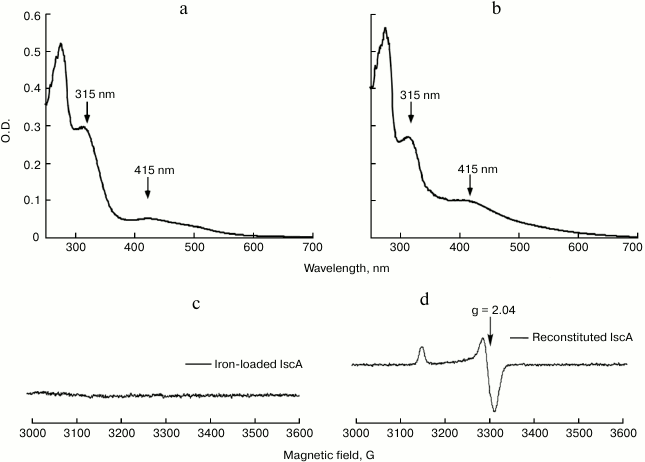
The iron bound form IscA could convert to iron–sulfur cluster bound form in vitro. When iron-loaded IscA was incubated with 1 mM L-cysteine and 1 μM IscS in buffer containing 2 mM DTT anaerobically at 37°C, the solution slowly turned from red to brownish. After 30 min incubation, the final UV-visible spectrum is similar to that of the wild type IscA containing a [4Fe-4S] cluster with one major absorption peak at 415 nm (Figs. 4a and 4b). Additionally, the iron-loaded IscA exhibited no EPR activity (Fig. 4c), while the reconstituted IscA gave a typical EPR signal at S = 1/2, with the g value around 2.04 (Fig. 4d), indicating the presences of the [4Fe-4S] cluster [15]. Furthermore, analysis of the reconstituted IscA showed that there were 3.73 ± 0.09 Fe and 3.82 ± 0.12 S atoms per IscA monomer, these results being in good agreement with those of wild type IscA. These results strongly suggested that the iron bound form of IscA was successfully converted to the iron–sulfur cluster bound form in vitro.
Fig. 4. The iron bound form of IscA could convert to iron–sulfur cluster bound form in vitro. Iron-loaded IscA (100 µM) was incubated anaerobically with 1 μM IscS, 1 mM L-cysteine, 2 mM DTT in buffer containing 500 mM NaCl and 20 mM Tris-HCl (pH 8.0). The amplitudes of the absorption peaks at 315 and 415 nm were used to monitor the formation of iron–sulfur cluster in IscA. a) UV-visible spectrum of iron-loaded IscA; b) UV-visible spectrum of reconstituted IscA; c) EPR spectrum of iron-loaded IscA; d) EPR spectrum of reconstituted IscA.
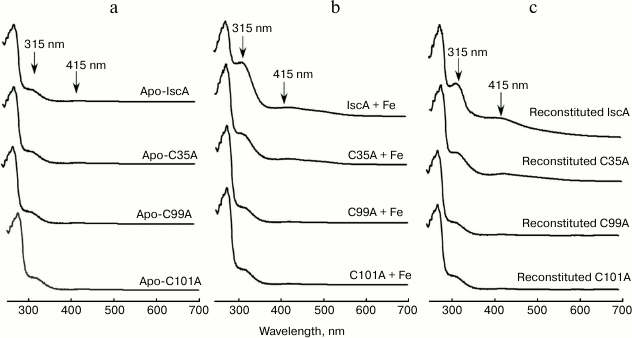
IscA is highly conserved from bacteria to humans with three invariant cysteine residues (Cys35, Cys99, and Cys101). To determine whether the iron bound form IscA converting to iron–sulfur cluster form is specific, we used the IscA mutant in which the Cys35, Cys99, and Cys101 were replaced with alanine. Figure 5 shows the UV-visible absorption spectra of the apo-IscA variants before (a) and after (b) incubation with 10-fold molar excess of Fe(NH4)2(SO4)2 in the presence of DTT. Whereas replacing Cys35 with alanine significantly decreased the iron binding activity of IscA, replacing Cys99 or Cys101 completely abolished the iron binding in the protein. Consequently, the IscA variants, which could not bind iron, also lost the iron–sulfur cluster binding activity (Fig. 5c). These results lead us to speculate that during the biogenesis of iron–sulfur cluster assembly in IscA, apo-IscA recruits intracellular free iron and forms an iron-loaded IscA, and the iron-loaded IscA is an intermediate state and can be converted to [4Fe-4S] cluster-loaded IscA.
Fig. 5. The conserved cysteine residues in IscA are essential for its specific iron and iron–sulfur cluster binding activity. a) The UV-visible absorption spectra of apo-IscA and the apo-IscA variants (C35A, C99A, and C101A); b) apo-IscA and the variants (50 μM) incubated with 500 μM Fe(NH4)2(SO4)2 in the presence of 2 mM DTT; c) apo-IscA and the variants (50 μM) were incubated with 500 μM Fe(NH4)2(SO4)2, 1 μM IscS, 1 mM L-cysteine, and 2 mM DTT at 37°C for 30 min. The protein was re-purified by passing through a Hi-Trap desalting column and subjected to UV-visible absorption measurements.
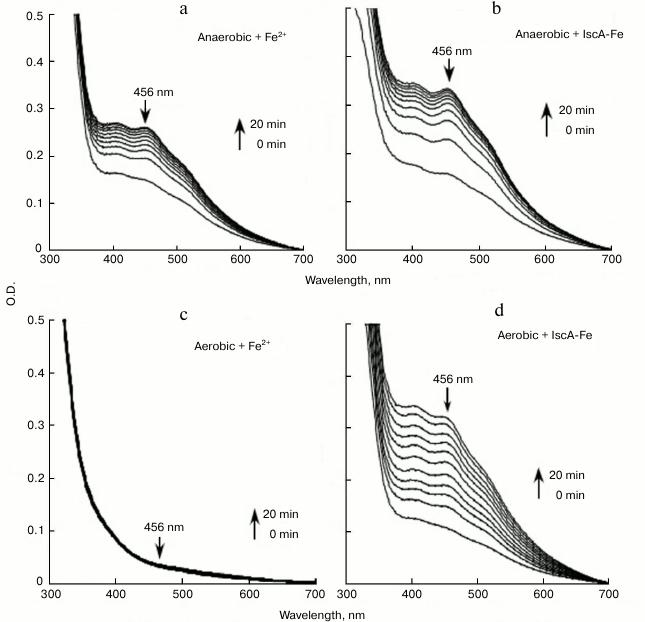
The iron-loaded IscA is able to provide iron for iron–sulfur cluster assembly in IscU in vitro. The finding that IscA is able to bind iron in a stable form prompted us to speculate that the iron-loaded IscA can behave as an iron donor for the assembly of iron–sulfur clusters. For that purpose, the scaffold protein IscU was chosen as a target. When 50 μM IscU was incubated anaerobically with 1 μM IscS, 2 mM DTT, and 200 μM Fe(NH4)2(SO4)2 in buffer containing 20 mM Tris-HCl (pH 8.0) and 500 mM NaCl at 37°C, an absorption peak at 456 nm that was attributed to the IscU [2Fe-2S] cluster gradually appeared and reached its maximum after 20-min incubation (Fig. 6a). Under the same anaerobic conditions, when iron-loaded IscA instead of ferrous iron was added to the reaction solution, the same absorption peak at 456 nm was apparent (Fig. 6b), indicating that the iron-loaded IscA had similar activity in providing iron for the iron–sulfur cluster in IscU.
Fig. 6. Iron-loaded IscA promotes the iron–sulfur cluster assembly in IscU in vitro. Purified IscU (50 μM) was incubated anaerobically with 1 μM IscS, 2 mM DTT, and different iron sources in buffer containing 20 mM Tris-HCl (pH 8.0) and 500 mM NaCl at 37°C for 5 min. Spectra were obtained every 2 min for 20 min after addition of 1 mM L-cysteine to initiate the iron–sulfur cluster assembly reaction. The amplitudes of the absorption peak at 456 nm were used to monitor the formation of iron–sulfur cluster in IscU. a) Fe(NH4)2(SO4)2 (200 μM) was used for the iron–sulfur cluster assembly in IscU under anaerobic conditions; b) same as in (a) except that 50 μM iron-loaded IscA was used instead of 200 μM Fe(NH4)2(SO4)2; c) 200 µM (NH4)2(SO4)2 was preincubated at 37°C for 3 h under aerobic conditions before being used for the iron–sulfur cluster assembly in IscU; d) same as in (c) except that 50 μM iron-loaded IscA was used instead of 200 μM Fe(NH4)2(SO4)2.
Under aerobic conditions, the ferrous iron was fully oxidized to ferric iron with the production of ferric hydroxide precipitate after exposure to air for 3 h. No absorption peak at 456 nm appeared (Fig. 6c), suggesting that there was no iron–sulfur cluster assembly in IscU. However, the same IscU [2Fe-2S] cluster absorption peak at 456 nm was apparent, indicating that the iron-loaded IscA could still provide iron for iron–sulfur cluster assembly in IscU after the same preincubation under aerobic conditions (Fig. 6d). The results suggest that the iron center in iron-loaded IscA is very stable and resistant to oxygen, which can prevent the formation of the biologically inaccessible ferric hydroxide and provide iron for iron–sulfur cluster assembly under aerobic conditions [21].
DISCUSSION
It has been shown that formation of intracellular iron–sulfur cluster is not a spontaneous process; it requires complex biosynthetic machinery [24]. Previous studies have shown that sulfur in iron–sulfur clusters is provided by IscS [25], whereas the iron donor of iron–sulfur cluster assembly is not really known. Iron toxicity implies that within cells iron must be stored and transported by specific proteins from which it can be delivered to the assembly machinery [26]. The fact that iron–sulfur clusters can be assembled with free iron in vitro without accessory proteins strongly suggested that the iron donor protein in the biosynthesis process is more in relation with its ability to solubilize, transport, and sequester iron than with an essential catalytic function for the reaction [27]. So far, these proteins are still unknown.
IscA is a putative iron donor of iron–sulfur cluster assembly [13-15]. In our previous studies, we have revealed that A. ferrooxidans IscA is an iron–sulfur protein, and the apo-IscA can serve as a scaffold to host a [4Fe-4S] cluster in vitro [16]. Here we show that the A. ferrooxidans IscA is indeed found to bind iron and to be able to provide it to IscU for iron–sulfur cluster synthesis in the presence of IscS and L-cysteine in vitro. At this time, it is worth pointing out that the recombinant IscAs from E. coli [26], Azotobacter vinelandii [11], and Synechocystis PCC 6803 [12] were purified as apoprotein without the iron–sulfur cluster, but the [2Fe-2S] or [4Fe-4S] could be reconstituted in vitro using sulfide and ferrous iron.
The crystal structure of E. coli IscA revealed that IscA can exist in a tetrameric form, and that the three conserved cysteine residues from each IscA monomer are predicted to form a “cysteine pocket” in a channel formed by association of the monomers [28]. Site-directed mutagenesis of A. ferrooxidans IscA showed that Cys35, Cys99, and Cys101 are crucial residues for its iron and iron–sulfur cluster binding, as removal of the sulfhydryl group of the cysteines abolished the iron binding activity and resulted in [4Fe-4S] cluster loss. These results are consistent with the crystal structure of IscA in which the conserved “cysteine pocket” can accommodate an iron center or an iron–sulfur cluster without significant re-arrangements of the protein structure [22].
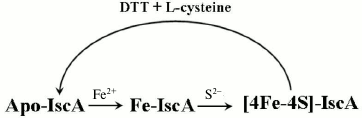
The work described here also shows that different forms of IscA represent different stages in the process of iron–sulfur cluster assembly in A. ferrooxidans IscA. These forms include apo-IscA, iron-loaded IscA, and [4Fe-4S] cluster-loaded IscA. First, apo-IscA recruits free iron and forms the iron-loaded IscA. The iron binding in IscA is an intermediate state during assembly of iron–sulfur clusters in IscA. It can convert to [4Fe-4S] cluster-loaded IscA in the presence of IscS and L-cysteine (Fig. 7). Additionally, iron-loaded IscA can act as iron donor for biogenesis of iron–sulfur cluster. The iron center in IscA can be mobilized by L-cysteine and transferred for the iron–sulfur cluster assembly in IscU. The interactions of IscA with IscS and IscU and how iron–sulfur cluster is transferred is currently under investigation.
Fig. 7. Proposed model for biogenesis of iron–sulfur cluster in IscA. Apo-IscA recruits free iron and forms iron-loaded IscA. Iron binding in IscA is an intermediate state during the assembly of iron–sulfur clusters. It can convert to [4Fe-4S] cluster-loaded IscA in the presence of IscS and L-cysteine.
We thank Dr. H. Ding at Louisiana State University (USA) for his kind help and advice; some of the experiments were performed in his laboratory.
This work was supported by the National Science Foundation of China (No. 50374075, 41073060) and the Research Fund for the Doctoral Program of Higher Education of China (20100075110010).
REFERENCES
1.Beinert, H., Holm, R. H., and Munck, E. (1997)
Science, 277, 653-659.
2.Johnson, D. C., Dean, D. R., Smith, A. D., and
Johnson, M. K. (2005) Annu. Rev. Biochem., 74,
247-281.
3.Xu, X. M., and Moller, S. G. (2011) Antioxid.
Redox. Signaling, 15, 271-307.
4.Fontecave, M. (2006) Nat. Chem. Biol.,
2, 171-174.
5.Ayala-Castro, C., Saini, A., and Outten, F. W.
(2008) Microbiol. Mol. Biol. R, 72, 110-125.
6.Lill, R., and Muhlenhoff, U. (2006) Annu. Rev.
Cell. Dev. Biol., 22, 457-486.
7.Wu, G. F., Li, P., and Wu, X. C. (2008) Biochem.
Biophys. Res. Commun., 374, 399-404.
8.Li, K. Y., Tong, W. H., Hughes, R. M., and Rouault,
T. A. (2006) J. Biol. Chem., 281, 12344-12351.
9.Chandramouli, K., Unciuleac, M. C., Naik, S., Dean,
D. R., Huynh, B. H., and Johnson, M. K. (2007) Biochemistry,
46, 6804-6811.
10.Wu, G. F., and Li, L. F. (2012) Biochemistry
(Moscow), 77, 135-142.
11.Bonomi, F., Lametti, S., Morleom, A., Ta, D., and
Vickery, L. E. (2011) Biochemistry, 50, 9641-9650.
12.Krebs, C., Agar, J. N., Smith, A. D., Frazzon,
J., Dean, D. R., Huynh, R. H., and Johnson, M. K. (2001)
Biochemistry, 40, 14069-14080.
13.Wollenberg, M., Berndt, C., Bill, E., Schwenn, J.
D., and Seidler, A. (2003) Eur. J. Biochem., 270,
1662-1671.
14.Lu, J., Bitoun, J. P., Tan, G., Wang, W., Min,
W., and Ding, H. (2010) Biochem. J., 428, 125-131.
15.Ding, H., Yang, J., Cloeman, L. C., and Yeung, S.
(2007) J. Biol. Chem., 282, 7997-8004.
16.Zeng, J., Geng, M., Jiang, H., Liu, Y., Liu, J.,
and Qiu, G. (2007) Arch. Biochem. Biophys., 463,
237-244.
17.Zeng, J., Zhao, W., Liu, Y., Xia, L., Liu, J.,
and Qiu, G. (2007) Biotechnol. Lett., 29, 1965-1972.
18.Zeng, J., Zhang, Y., Liu, Y., Zhang, X., Xia, L.,
Liu, J., and Qiu, G. (2007) Biotechnol. Lett., 29,
1983-1990.
19.Fischer, D. C. (1967) Clin. Chem.,
13, 6-11.
20.Siegel, L. M. (1965) Anal. Biochem.,
11, 126-132.
21.Wang, W., Huang, H., Tan, G., Si, F., Min, W.,
Landry, A. P., Lu, J., and Ding, H. (2010) Biochem. J.,
432, 429-436.
22.Yang, J., Bitoun, J. P., and Ding, H. (2006)
J. Biol. Chem., 281, 27956-27963.
23.Ding, H., Harrison, K., and Lu, J. (2005) J.
Biol. Chem., 280, 30432-30437.
24.Lill, R. (2009) Nature, 460,
831-838.
25.Schwartz, C. J., Djaman, O., Imlay, J. A., and
Kiley, P. J. (2000) Proc. Natl. Acad. Sci. USA, 97,
9009-9014.
26.Fontecave, M., and Ollagnier-de-Choudens, S.
(2008) Arch. Biochem. Biophys., 474, 226-237.
27.Layer, G., Ollagnier-de Choudens, S., Sanakis,
Y., and Fontecave, M. (2006) J. Biol. Chem., 281,
16256-16263.
28.Cupp-Vickery, J. R., Silberg, J. J., Ta, D. T.,
and Vickery, L. E. (2004) J. Mol. Biol., 338,
127-137.