Interaction of Tetraphenylphosphonium and Dodecyltriphenylphosphonium with Lipid Membranes and Mitochondria
T. A. Trendeleva1, A. G. Rogov1, D. A. Cherepanov2, E. I. Sukhanova1, T. M. Il’yasova3, I. I. Severina3, and R. A. Zvyagilskaya1*
1Bach Institute of Biochemistry, Russian Academy of Sciences, Leninsky pr. 33, 119071 Moscow, Russia; fax: (495) 954-2732; E-mail: renata_z@inbi.ras.ru2Frumkin Institute of Physical Chemistry and Electrochemistry, Russian Academy of Sciences, Leninsky pr. 31, 119991 Moscow, Russia; fax: (495) 952-5308
3Belozersky Institute of Physico-Chemical Biology, Lomonosov Moscow State University, 119991 Moscow, Russia; fax: (495) 939-0338
* To whom correspondence should be addressed.
Received May 28, 2012
The permeability of a planar lipid membrane (composed of diphytanoylphosphatidylcholine) for tetraphenylphosphonium (TPP) was investigated. The observed level of the diffusion potential generated as a function of the TPP concentration gradient differed from the theoretically expected value, possibly due to proton leakage of the membrane mediated by the traces of fatty acids in the phospholipid forming the membrane. Using the molecular dynamics approach to study movement of TPP and dodecyltriphenylphosphonium (C12TPP) with different affinity to the lipid bilayer through a bilayer lipid membrane, it was found that C12TPP has a greater affinity to the membrane surface than TPP. However, the two cations have the same activation energy for transmembrane transfer. Interaction of TPP and C12TPP with tightly-coupled mitochondria from the yeast Yarrowia lipolytica was also investigated. At low, micromolar concentrations, both cations are “relatively weak, mild uncouplers”, do not shunt electron transfer along the respiratory chain, do not disturb (damage) the inner mitochondrial membrane, and profoundly promote the uncoupling effect of fatty acids. At higher concentrations they inhibit respiration in state 3, and at much higher concentrations they induce swelling of mitochondria, possibly due to their detergent action.
KEY WORDS: TPP, C12TPP, diffusion potential, movement through lipid bilayer, yeast, Yarrowia lipolytica, mitochondria, fatty acidsDOI: 10.1134/S000629791209009X
Abbreviations: Ala, alamethicin; CCCP, carbonyl cyanide m-chlorophenylhydrazone; C12TPP, dodecyltriphenylphosphonium; MES, 2-(N-morpholino)ethanesulfonic acid; ROS, reactive oxygen species; TPP, tetraphenylphosphonium; Δψ, transmembrane electric potential difference.
Artificial (not naturally occurring) penetrating ions were originally
synthesized and successfully used by E. A. Liberman and V. P. Skulachev
[1-3] to demonstrate the
validity of Mitchell’s chemiosmotic hypothesis. Cations with
delocalized charge depolarized the inner mitochondrial membrane
(decreased the membrane potential) using the transmembrane electric
potential difference generated by the respiratory chain as a driving
force, precisely in conformity with predictions of Mitchell’s
hypothesis. Later, the penetrating ions were dubbed “Skulachev
ions” by David E. Green [4].
There is now ample evidence that mitochondria are the principle generators of reactive oxygen species (ROS) in cells and that many pathologies and aging arise from oxidative damage to key cellular compounds (DNA, especially mitochondrial DNA (mDNA), lipids, and proteins) [5]. The idea arose to employ penetrating cations with delocalized charge as “electric locomotive molecules” targeting antioxidants conjugated to these ions to the cellular and mitochondrial interior [6-10]. Mitochondria-targeted (i.e. being accumulated predominantly, if not exclusively into mitochondria) lipophilic antioxidants offer advantages over conventional water-soluble antioxidants as they are transported into and accumulated within cells and mitochondria in conformity with the membrane potential generated on the cytoplasmic or mitochondrial membrane, respectively. As a result, their concentrations in mitochondria increase several orders of magnitude in comparison with initial low, nontoxic nanomolar and sub-nanomolar concentrations. Moreover, if the cations are conjugated with antioxidants via a long aliphatic chain, they gain the additional capacity to be accumulated in the lipid bilayer of the inner mitochondrial membrane in conformity with their distribution in the octanol–water system, which increases their concentrations in the lipid phase of mitochondria by two to three orders of magnitude. Finally, some of (but not all) lipophilic antioxidants, once accumulated in the lipid bilayer of the inner mitochondrial membrane, can be reduced (regenerated) by components of the respiratory chain, which ensures their repetitive functions.
Among “locomotive cations” targeting antioxidants to mitochondria, the most popular is triphenylphosphonium conjugated with ubiquinone (a component of the respiratory chain) [7, 8], plastoquinone (a component of the photosynthetic electron-transport chain) [5, 9-11], and vitamin E [6]. Conjugates of triphenylphosphonium with plastoquinone (SkQ1) and dodecyltriphenylphosphonium (C12TPP) were found to depolarize the inner mitochondrial membrane of animal mitochondria, to stimulate cell respiration, acting as “mild uncouplers”, and to promote transport of fatty acid anions through artificial lipid and mitochondrial membranes [11].
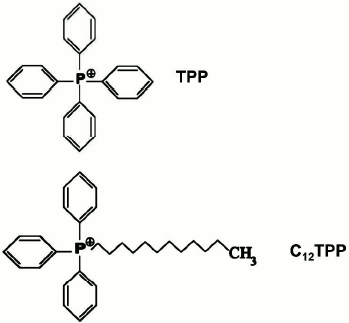
In this paper, we focus on the interaction of tetraphenylphosphonium (TPP) and lipophilic dodecyltriphenylphosphonium (C12TPP) (both are penetrating cations with delocalized charge) with yeast mitochondria. The structures of these compounds are presented in Fig. 1. The need for this approach stems from the fact that the interaction of TPP with animal mitochondria was investigated many years ago, and studies on yeast mitochondria remain scarce. Besides, for close examination of the effects of biologically active compounds at the mitochondrial level, tightly-coupled yeast mitochondria have some advantages over animal mitochondria as they have lower rate of endogenous respiration (which permits the investigation of the oxidation of individual substrates), respire with exogenous substrates with very high rates, and, as we showed [12-15], lack Ca2+/Pi-dependent pores (which facilitates interpretation of the resulting data).
Fig. 1. Chemical structures of TPP and C12TPP.
MATERIALS AND METHODS
Reagents. ATP, pyruvate, malate, succinate, palmitate, fatty acid-free BSA, alamethicin, oligomycin, safranine O, carbonyl cyanide m-chlorophenylhydrazone (CCCP), EDTA, EGTA, Tris, sorbitol, and mannitol were purchased from Sigma-Aldrich (USA); Coomassie G-250 and Zymolyase 20T were from MP Biomedicals (USA); yeast extract was from Difco (USA); diphytanoylphosphatidylcholine was from Avanti Polar Lipids (USA). Other reagents of the highest quality available were obtained from domestic suppliers.
Permeability of planar lipid membrane. This was evaluated as described earlier [16] with minor modifications. The bilayer lipid membrane (BLM) was formed on a 1-mm aperture in a Teflon septum separating the experimental chamber into two compartments. To form the BLM, we used synthetic diphytanoylphosphatidylcholine dissolved in decane (20 mg phospholipid in 1 ml decane). Electric potential difference across the BLM was measured with AgCl electrodes submerged in each compartment into medium containing 10 mM Tris, 10 mM Mes, 10 mM KCl, and 0.1 mM TPP, pH 7.0, using a Keithley 6517 amplifier (Cleveland, USA). The dependence of the diffusion potential on the TPP concentration, which was varied only in one compartment, was measured.
Molecular dynamics modeling. The free energy of cations C12TPP and TPP within a homogeneous lipid bilayer was studied by molecular dynamics modeling. The lipid vesicle was modeled as a bilayer consisting of 100 palmitoyloleoylphosphatidylcholine (POPC) molecules (50 per leaflet) surrounded by 5450 water molecules. This system is large enough to ensure that ions in the middle of the membrane patch do not perturb the general membrane structure. The net positive charge of the membrane was neutralized by Cl–. The system included ~30,000 atoms in total. Orthogonal periodic boundaries with x,y-translation length and height of 5.8 and 8.8 nm on average, respectively, were used. Simulations were performed with the Gromacs program [17] using the CHARMM36 force field [18] and TIP3P water model [19]. The electrostatics were calculated using the particle mesh Ewald method, and all bonds were maintained with the LINCS routine. Molecular dynamics simulations were carried out at a constant number of particles (N), constant pressure (P = 1 atm), and constant temperature (T = 313 K) (NPT ensemble). Three-dimensional periodic boundary conditions were applied with the z-axis lying along a direction normal to the bilayer. The pressure was controlled semi-isotropically so that the x-y and z sizes of the simulation box were allowed to fluctuate independently from each other, keeping the total pressure constant. The averaged area per lipid was 0.67 nm2. The force field parameters for C12TPP and TPP were taken from [11]. All molecular dynamics calculations were performed at the Lomonosov supercomputer in the Computation Center of Moscow State University.
The free energy of an ion within the membrane relative to its value in solution W(z) was calculated as the integral of the mean force acting on average on the ion during its slow movement across the membrane (the potential of mean force, PMF). According to the method of umbrella sampling [20], the movement of the ion was driven by a harmonic biasing potential:
where F(ζ) is the instantaneous force acting on the transferred ion at a chosen coordinate, ζ.
Organism, cultivation conditions, and isolation of mitochondria. In this work we used the Yarrowia lipolytica strain isolated as a pure isolate from epiphytic microflora of salt-excreting arid plants (central Negev Desert of Israel) and identified on the basis of its morphology, biochemical-physiological characteristics, and quantitative chemotaxonomic and molecular marker analyses as Yarrowia lipolytica (Wick.) van der Walt and Arx [21]. For bioenergetics this strain could be interesting as possessing under “normal” conditions (in the absence of respiratory inhibitors) typical for yeasts respiratory chain with all four respiratory complexes and the alternative oxidase. Besides, this strain with its unique capacity to grow at alkaline pH values (to pH as high as 9.7) is so far the only model for studying “Na+ bioenergetics” in eukaryotes, including Na+-coupled transport of nutrients [22-26], as well as for revealing extremes at which the mitochondrial mode of energy conservation is still operative [27].
The yeast cells were cultivated at 28°C in agitated (220 rpm) 750-ml Erlenmeyer flasks in 100 ml of a semi-synthetic medium [28] containing 1% succinate as the sole source of carbon and energy and harvested at late exponential growth phase. Cultivation on succinate as the sole source of carbon and energy should be accompanied by constant functioning of reverse electron transport in the respiratory chain to produce NADH and then NADPH (by using NADH-kinase) required for anabolic reactions. Therefore, it would provide the constant functioning of complex I in the respiratory chain and reduce to a minimum the activity of the terminal alternative oxidase that is not coupled to energy conservation.
Mitochondria were isolated from Y. lipolytica by the methods designed in our laboratory and described earlier [14]. Mitochondrial preparations thus obtained were fully active for at least 4 h when kept on ice. In some experiments, mitochondria were isolated in BSA-free medium.
Oxygen consumption was monitored polarographically with a closed Clark-type oxygen electrode in a 1-ml cell in basal medium containing 0.6 M mannitol, 0.2 mM Tris-phosphate buffer, pH 7.2-7.4, 20 mM pyruvate, 5 mM malate, and mitochondrial protein (0.5 mg/ml). Respiratory control and ADP/O ratios were calculated as recommended by Chance and Williams [29].
Mitochondrial transmembrane potential (Δψ) generated on the inner mitochondrial membrane was measured with a Beckman Coulter DU-650 spectrophotometer (USA) at the wavelength pair (511-533 nm) with safranine O as a Δψ-sensitive probe [30]. The incubation medium contained 0.6 M mannitol, 0.2 mM Tris-phosphate buffer, pH 7.2-7.3, 20 mM succinate, and mitochondrial protein (0.5 mg/ml).
Mitochondrial swelling was monitored with a Hitachi-557 spectrophotometer (Japan) by recording changes in apparent absorbance at 540 nm.
Mitochondrial protein was assayed by the Bradford method [31] with BSA as the standard.
All data shown in figures are representative of at least four replicates.
RESULTS AND DISCUSSION
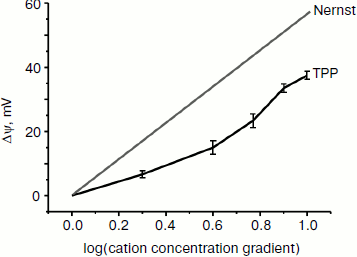
Interaction of TPP with planar lipid membrane. Figure 2 shows the diffusion potential of H+ ions created between the two BLM-separated compartments as a function of increasing TPP concentrations. In the case of an “ideal” singly charged cation penetrating through the lipid membrane but not disturbing it, a 10-fold gradient of its concentration should, as predicted by the Nernst equation, generate Δψ equal to 60 mV. As seen in Fig. 2, the experimental curve did not coincide with the theoretical one. The discrepancy may be either due to some damaging effect of TPP on the planar lipid membrane, or, most probably, due to a proton “leakage” across the membrane mediated by the presence of traces of fatty acids in the membrane-forming phospholipid. When fatty acid-free phospholipid was used, the experimental and theoretical curves coincided (personal communication from T. M. Il’yasova).
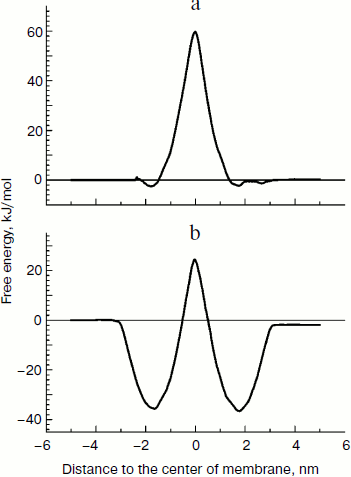
Free energy of lipophilic ions in the membrane. We applied the molecular dynamics (MD) approach to study movement of the two cations with different affinity to the lipid bilayer, C12TPP and TPP, through a lipid membrane. At the starting point of simulations, we placed the ions in the polar part of the lipid leaflet at one side of the membrane, and after 100 nsec equilibration, the biasing potential (Eq. (1)) was applied to the C12TPP or TPP, respectively, starting from the ion equilibrium position z0 in two opposite directions, so the ions crossed the whole membrane from one side to the other with a constant rate, the total simulation time for each ion being ~1 μsec. The free energy profiles calculated by the potential of mean force (PMF) are plotted in Fig. 3.Fig. 2. Dependence of diffusion potential generated on a planar lipid membrane on TPP concentration.
The free energy profile could be characterized by two parameters: the free energy of ion binding to the membrane surface ΔGbound (the minimum depth relative the water phase) and the activation energy of the transmembrane transfer ΔG# (the energy barrier height in the hydrophobic part of the membrane). Comparing the results, we conclude that C12TPP has greater affinity to the membrane surface than C12TPP (the binding energy being –36 and –2 kJ/mol, respectively). However, the two ions have the similar activation energy for the transmembrane transfer (~61 kJ/mol). The greater affinity of C12TPP to the membrane apparently could be explained by the longer hydrocarbon “tail”, so one carbon atom contributes ~3 kJ/mol to ΔGbound.
Interaction of TPP and C12TPP with yeast mitochondria. The yeast mitochondrial preparations used in this series of experiments exhibited a high degree of intactness as inferred from high (~5) respiratory control ratios [29] (indicative of tight coupling of respiration and phosphorylation) upon oxidation of NAD-dependent substrates and ADP/O ratios (indicative of efficiency of oxidative phosphorylation) that were close to the theoretically expected maxima.Fig. 3. Free energy profiles for transmembrane transfer of TPP (a) and C12TPP (b) along the normal to the lipid membrane plane, the results of molecular dynamics simulation.
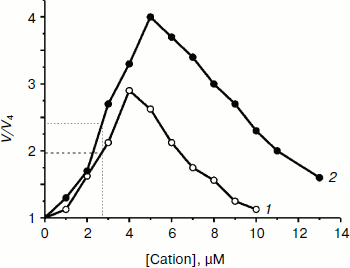
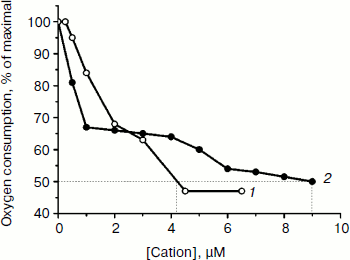
Both TPP and C12TPP taken at low concentrations substantially stimulated oxidation of NAD-dependent substrates (pyruvate + malate) in state 4 respiration (Fig. 4), causing a half-maximal effect (C50) at 2.8 µM, which was indicative of their uncoupling effect. Under these conditions neither cation shunted the electron flow from complex I (Fig. 5) and from complex II (data not shown). Both compounds were partially inhibitory for respiration in state 3 if added in excess (Fig. 6). The more profound inhibitory action of C12TPP was likely related to its higher affinity to the membrane (see above) and higher hydrophobicity. It is known that complex I of the respiratory chain is inhibited by hydrophobic compounds of different composition, which are suggested [32] to compete with piericidin/rotenone (inhibitors of the complex) for one of the sites accessible for hydrophobic compounds.
Fig. 4. TPP (1) and C12TPP (2) at low concentrations activate respiration of yeast mitochondria in state 4. The incubation medium contained 0.6 M mannitol, 0.2 mM Tris-phosphate, pH 7.2-7.4, 0.5 mM EGTA, 20 mM Tris-pyruvate, 5 mM malate, and mitochondria (0.5 mg/ml protein).
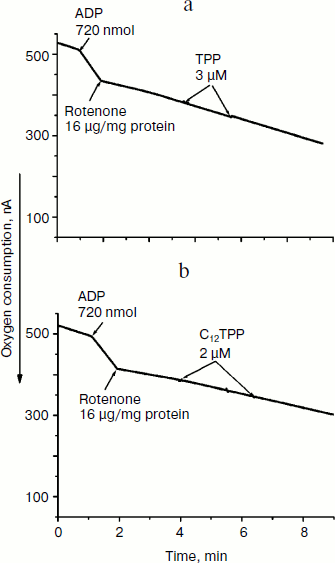
Fig. 5. TPP (a) and C12TPP (b) at low concentrations do not shunt electron flow from complex I of the respiratory chain. Incubation medium as in Fig. 4.
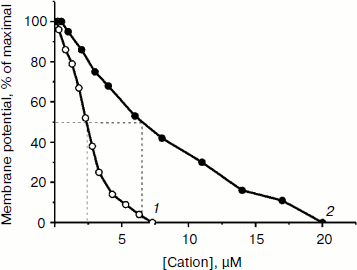
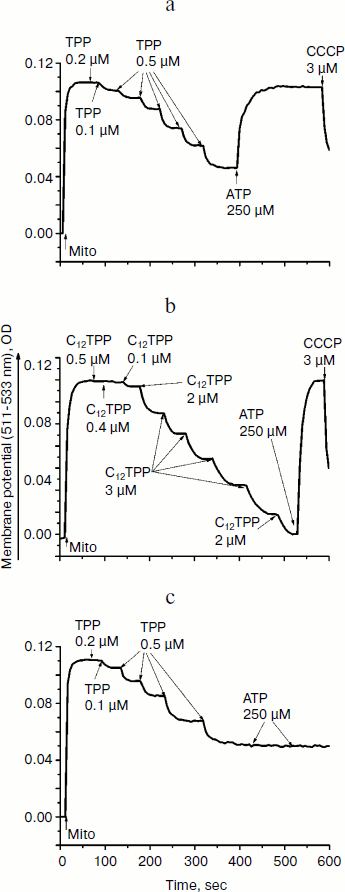
The ability of low concentrations of TPP and C12TPP to uncouple respiration from phosphorylation was supported by monitoring the membrane potential. Both TPP and C12TPP depolarized the mitochondrial membrane (diminished Δψ) (Fig. 7). Importantly, neither TPP nor C12TPP damaged the mitochondrial membrane, as inferred from the fact that added ATP totally restored the decreased membrane potential to the initial level (Fig. 8, a and b). The recoupling effect of ATP was entirely prevented by oligomycin (Fig. 8c) (an inhibitor of the ATP-synthase complex), indicating that the energization of mitochondria was presumably due to ATP hydrolysis by H+-ATPase. We suggest using this test (recovering of the membrane potential to the initial level after ATP addition) when studying the interaction of mitochondria with other physiologically important substances.Fig. 6. TPP (1) and C12TPP (2) at relatively high concentrations partially inhibit respiration of yeast mitochondria in state 3. Incubation medium as in Fig. 4.
Fig. 7. Membrane depolarization by TPP (1) and C12TPP (2). The incubation medium contained 0.6 M mannitol, 0.2 mM Tris-phosphate, pH 7.2-7.4, 0.5 mM EGTA, 20 mM Tris-succinate, 10.5 µM safranine O, and mitochondria (0.5 mg/ml protein).
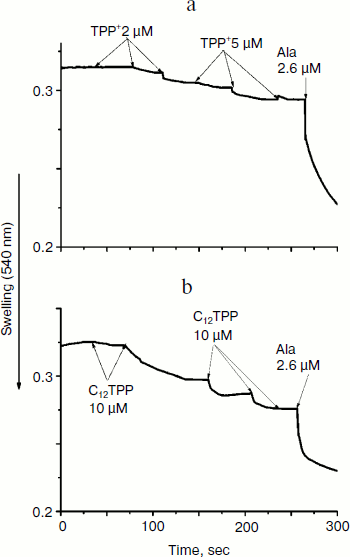
Consistent with the above-described recoupling effect of ATP, the yeast mitochondria did not swell in iso-osmotic mannitol-based medium supplemented with 40 mM KCl and a respiratory substrate when exposed to low concentrations of TPP or C12TPP (Fig. 9). The absence of response in yeast mitochondria to TPP and C12TPP was not simply due to structural limitations, since large amplitude swelling occurred in the presence of alamethicin (Ala), which being incorporated into mitochondrial membranes forms channels with diameter of 1 nm that are permeable to divalent cations and low molecular weight compounds. Higher concentrations of TPP and C12TPP induced swelling of the yeast mitochondria, presumably due to their detergent action.Fig. 8. TPP (a, c) and C12TPP (b) do not disturb the mitochondrial membrane. a, b) The membrane potential decrease by the cations was totally restored by ATP. c) Oligomycin entirely prevented the “recoupling” effect of ATP. Incubation medium as in Fig. 7.
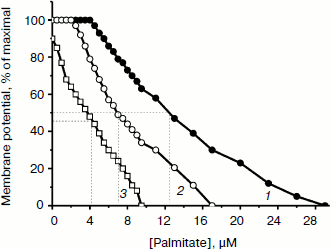
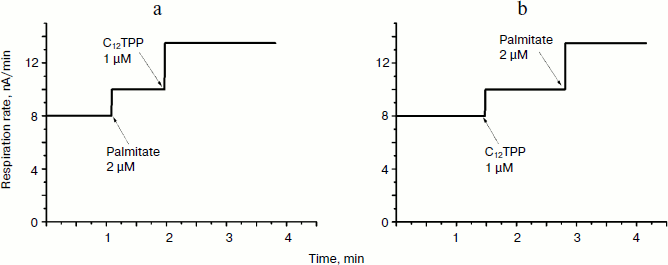
TPP and C12TPP added at low, non-uncoupling concentrations substantially promoted the uncoupling action of fatty acids (with palmitate as an example) in Y. lipolytica mitochondria. A very low, non-uncoupling TPP concentration lowered the palmitate concentration needed for half-maximal Δψ decrease by a factor of two, and TPP at a concentration exerting 10% uncoupling effect decreased this value approximately 3-fold (Fig. 10). Very similar results were obtained upon concerted action of C12TPP and palmitate taken at low concentrations. Figure 11 presents the first derivatives of amperometric curves of oxygen consumption by yeast mitochondria upon successive additions of low concentrations of C12TPP and palmitate (Fig. 11a) as well as palmitate and C12TPP (Fig. 11b). Synergism of their action is clearly seen. Similar results were obtained with other fatty acids, both saturated (stearic, pentadecanoic, lauric) and unsaturated (oleic and linoleic acids) (not shown in figures).Fig. 9. TPP (a) and C12TPP (b) at high concentrations exhibit detergent action. The incubation medium contained 0.5 M mannitol, 0.2 mM Tris-phosphate, pH 7.2-7.4, 1 mM EGTA, 10 mM Tris-succinate, 40 mM KCl, and mitochondria (0.5 mg/ml protein).
Fig. 10. TPP promotes the uncoupling effect of fatty acids. 1) Depolarization of the inner mitochondrial membrane as a function of palmitate concentration. 2) Depolarization of the inner mitochondrial membrane by palmitate in the presence of non-uncoupling TPP concentration. 3) Depolarization of the inner mitochondrial membrane by palmitate in the presence of TPP at a concentration exerting 10%-uncoupling effect. Incubation medium as in Fig. 7.
Thus, at low concentrations, TPP and C12TPP are “relatively weak, mild uncouplers”, do not shunt the electron flow in the respiratory chain, do not damage the mitochondrial membrane, and do potentiate the uncoupling effect of saturated and unsaturated fatty acids. At higher concentrations they partially inhibit respiration in state 3 and depolarize the membrane. At much higher concentrations they induce swelling of mitochondria, possibly due to their detergent action.Fig. 11. C12TPP and palmitate mutually promote the uncoupling effect of each other. Incubation medium as in Fig. 4.
In this paper we not only supported the previously obtained (on animal mitochondria) data on the uncoupling effect of TPP [1-3], but also demonstrated, to our knowledge for the first time, that low concentrations of TPP do not shunt the electron flow in the respiratory chain and do not disturb the mitochondrial membrane. In yeast mitochondria TPP, as in animal mitochondria, promotes the uncoupling action of fatty acids. Also, novel data on the distribution of TPP and C12TPP in a planar lipid membrane were obtained.
This work was supported by the Russian Foundation for Basic Research, grant No. 09-04-01238.
REFERENCES
1.Liberman, E. A., Topaly, V. P., Tsofina, L. M.,
Jasaitis, A. A., and Skulachev, V. P. (1969) Nature, 222,
1076-1078.
2.Skulachev, V. P., Sharaf, A. A., and Liberman, E.
A. (1967) Nature, 16, 718-719.
3.Skulachev, V. P. (1970) FEBS Lett.,
11, 301-308.
4.Green, D. E. (1974) Biochim. Biophys. Acta,
346, 27-78.
5.Skulachev, V. P., Anisimov, V. N., Antonenko, Yu.
N., Bakeeva, L. E., Chernyak, B. V., Erichev, V. P., Filenki, O. F.,
Kalinina, N. I., Kapelko, V. I., Kolosova, N. G., Kopnin, B. P.,
Korshunova, G. A., Lichinitser, M. R., Obukhova, L. A., Pasyukova, E.
G., Pisarenko, O. I., Roginsky, V. A., Ruuge, E. K., Senin, I. I.,
Severina, I. I., Skulachev, M. V., Spivak, I. M., Tashlitsky, B. N.,
Tkachuk, V. A., Vyssokikh, M. Yu., Jaguzhinsky, L. S., and Zorov, D. B.
(2009) Biochim. Biophys. Acta, 1782, 437-461.
6.Smith, R. A., Porteous, C. M., Coulter, C. V., and
Murphy, M. P. (1999) Eur. J. Biochem., 263, 709-716.
7.Murphy, M. P., Echtay, K. S., Blaikie, F. H.,
Asin-Cayuela, J., Cocheme, H. M., Green, K., Buckingham, J. A., Taylor,
E. R., Hurrell, F., Hughes, G., Miwa, S., Cooper, C. E., Svistunenko,
D. A., Smith, R. A., and Brand, M. D. (2003) J. Biol. Chem.,
278, 48534-48545.
8.Brown, S. E., Ross, M. F., Sanjuan-Pla, A., Manas,
A. R., Smith, R. A., and Murphy, M. P. (2007) Free Radic. Biol.
Med., 42, 1766-1780.
9.Antonenko, Y. N., Avetisyan, A. V., Bakeeva, L. E.,
Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Y.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Y., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vassiliev, J. M., Vyssokikh, M.
Y., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Biochemistry (Moscow), 73, 1273-1287.
10.Skulachev, M. V., Antonenko, Y. N., Anisimov, V.
N., Chernyak, B. V., Cherepanov, D. A., Chistyakov, V. A., Egorov, M.
V., Kolosova, N. G., Korshunova, G. A., Lyamzaev, K. G., Plotnikov, E.
Y., Roginsky, V. A., Savchenko, A. Y., Severina, I. I., Severin, F. F.,
Shkurat, T. P., Tashlitsky, V. N., Shidlovsky, K. M., Vyssokikh, M. Y.,
Zamyatnin, A. A., Jr., Zorov, D. B., and Skulachev, V. P. (2011)
Curr. Drug Targets, 12, 800-826.
11.Severin, F. F., Severina, I. I., Antonenko, Y.
N., Rokitskaya, T. I., Cherepanov, D. A., Mokhova, E. N., Vyssokikh, M.
Y., Pustovidko, A. V., Markova, O. V., Yaguzhinsky, L. S., Korshunova,
G. A., Sumbatyan, N. V., Skulachev, M. V., and Skulachev, V. P. (2010)
Proc. Nat. Acad. Sci. USA, 107, 663-668.
12.Kovaleva, M. V., Sukhanova, E. I., Trendeleva, T.
A., Zyl’kova, M. V., Ural’skaya, L. A., Popova, K. M.,
Saris, N.-E., and Zvyagilskaya, R. A. (2009) J. Bienerg.
Biomembr., 41, 239-249.
13.Kovaleva, M. V., Sukhanova, E. I., Trendeleva, T.
A., Popova, K. M., Zylkova, M. V., Uralskaya, L. A., and Zvyagilskaya,
R. A. (2010) Biochemistry (Moscow), 75, 297-303.
14.Trendeleva, T., Sukhanova, E., Ural’skaya,
L., Saris, N.-E., and Zvyagilskaya, R. (2011) J. Bienerg.
Biomembr., 43, 623-631.
15.Trendeleva, T., Sukhanova, E., Ural’skaya,
L., Saris, N.-E., and Zvyagilskaya, R. (2011) J. Bienerg.
Biomembr., 43, 633-644.
16.Severina, I. I. (1982) Biochim. Biophys.
Acta, 681, 311-317.
17.Van der Spoel, D., Lindahl, E., Hess, B.,
Groenhof, G., Mark, A. E., and Berendsen, H. J. (2005) J. Comput.
Chem., 26, 1701-1718.
18.Brooks, B. R., Brooks, C. L., III, Mackerell, A.
D., Jr., Nilsson, L., Petrella, R. J., Roux, B., Won, Y., Archontis,
G., Bartels, C., Boresch, S., Caflisch, A., Caves, L., Cui, Q., Dinner,
A. R., Feig, M., Fischer, S., Gao, J., Hodoscek, M., Im, W., Kuczera,
K., Lazaridis, T., Ma, J., Ovchinnikov, V., Paci, E., Pastor, R. W.,
Post, C. B., Pu, J. Z., Schaefer, M., Tidor, B., Venable, R. M.,
Woodcock, H. L., Wu, X., Yang, W., York, D. M., and Karplus, M. (2009)
J. Comp. Chem., 30, 1545-1615.
19.Price, D. J., and Brooks, C. L. (2004) J.
Chem. Phys., 121, 10096-10103.
20.Torrie, G. M., and Valleau, J. P. (1977)
Comput. Phys., 23, 187-193.
21.Zvyagilskaya, R., Andreishcheva, E., Soares, I.
M., Khozin, I., Berhe, A., and Persson, B. L. (2001) J. Basic
Microbiol., 41, 283-303.
22.Zvyagilskaya, R. A., Parchomenko, O. A., and
Persson, B. L. (2000) IUBMB Life, 50, 151-155.
23.Zvyagilskaya, R. A., Parchomenko, O., Abramova,
N., Allard, P., Panaretakis, T., Pattison-Granberg, J., and Persson, B.
L. (2001) J. Membr. Biol., 183, 39-50.
24.Zvyagilskaya, R., and Persson, B. L. (2003)
IUBMB Life, 55, 151-154.
25.Zvyagilskaya, R. A., and Persson, B. L. (2004)
Biochemistry (Moscow), 69, 1310-1317.
26.Zvyagilskaya, R. A., and Persson, B. L. (2005)
Cell Biol. Int., 29, 87-94.
27.Zvyagilskaya, R. A., Parkhomenko, O. A.,
Gordeeva, A. V., Deryabina, Y. I., and Persson, B. L. (2004) Biosci.
Reps., 24, 117-125.
28.Andreishcheva, E. N., Isakova, E. P., Sidorov, N.
N., Abramova, N. B., Ushakova, N. A., Shaposhnikov, G. L., Soares, M.
I., and Zvyagilskaya, R. A. (1999) Biochemistry (Moscow),
64, 1061-1067.
29.Chance, B., and Williams, G. R. (1955)
Nature, 175, 1120-1121.
30.Akerman, K. E. O., and Wikstrom, M. K. F. (1976)
FEBS Lett., 68, 191-197.
31.Bradford, M. M. (1976) Anal. Biochem.,
72, 248-254.
32.Murphy, M. P., Krueger, M. J., Sablin, S. O.,
Ramsay, R. R., and Singer, T. P. (1995) Biochem. J., 306,
359-365.