Antimutagenic Activity of Mitochondria-Targeted Plastoquinone Derivative
V. A. Chistyakov*, M. A. Sazykina, A. A. Alexandrova, N. I. Belichenko, E. V. Mashkina, L. V. Gutnikova, P. V. Zolotukhin, and T. P. Shkurat
Research Institute of Biology, Southern Federal University, pr. Stachki 194/1, 344090 Rostov-on-Don, Russia; E-mail: vladimirchi@yandex.ru* To whom correspondence should be addressed.
Received November 2, 2009
The ability of cationic plastoquinone derivative 10-(6′-plastoquinonyl) decyltriphenylphosphonium (SkQ1) to modify processes of spontaneous and induced mutagenesis was studied. It is shown that daily introduction of this compound into male Wistar rats in doses of 25 and 250 nmol/kg during two weeks decreases spontaneous level of chromosome aberrations in anaphase in the eye cornea from 0.39 ± 0.09 to 0.13 ± 0.08 and 0.14 ± 0.05, respectively. The level of 8-hydroxy-2′-deoxyguanosine in blood serum of the investigated animals decreases from 32.12 ± 1.55 to 25.90 ± 2.26 and 25.76 ± 1.50 ng/ml, respectively. These facts indicate that the decrease in spontaneous clastogenesis is caused by decreased level of DNA damage by endogenous reactive oxygen species. A higher dose of SkQ1 also decreases to control level chromosome aberrations caused by oxygen under pressure of 0.5 MPa for 60 min. It is also shown in experiments with bacterial biosensors that SkQ1 is able to efficiently protect cells against genotoxic effect of UV radiation at 300-400 nm.
KEY WORDS: SkQ1, penetrating cations, antioxidants, mutagenesis, 8-OH-dG, chromosome aberrations, DNA damage, hyperbaric oxygenation, UV radiationDOI: 10.1134/S0006297910030028
Abbreviations: HBO, hyperbaric oxygenation; 8-OH-dG, 8-OH-2′-deoxyguanosine; ROS, reactive oxygen species; SkQ1, 10-(6′-plastoquinonyl) decyltriphenylphosphonium.
Among numerous hypothetical constructions called to explain or at least
describe the mechanism of aging, the concepts associated with
Harman’s idea concerning the involvement of free-radical
oxidation mechanisms [1] are characterized by the
highest theoretical potential. As biology of oxidative stress
developed, it became clear that mitochondria, figuratively called
“the dirtiest place in the cell”, are the main
intracellular source of reactive oxygen species (ROS) [2]. Therefore Harman’s free radical theory was
transformed into the mitochondrial theory of aging. The development of
ideas concerning the involvement of free-radical oxidation in launching
the programmed mechanisms of cell death, including apoptosis [3], led V. P. Skulachev to propose the phenoptosis
concept [4], methodologically representing
synthesis of ideas about stochastic and deterministic (in cybernetic
sense) bases of aging. According to the phenoptosis theory, generation
of cell-toxic ROS launches the process of gradual degradation of the
organism, which has adaptive character at the population level. The
main practical result and simultaneously the main point of verification
of the phenoptosis concept is the idea suggesting the use of
antioxidants for switching off the aging program. Reasons of poor
fitness for this purpose of presently known natural antioxidants and
those synthetic antioxidants imitating natural ones are described in
detail in works by V. P. Skulachev [5, 6]. Theoretical bases for constructing lipophilic
organic cations capable of addressed accumulation in mitochondria were
elaborated under his leadership in the 1970s. Following the proposal of
D. Green, such molecules were called “Skulachev ions”.
In 2003-2005 a number of Skulachev ions “charged” with plastoquinone, a chloroplast metabolite exhibiting strong antioxidant properties and ability for reduction upon interaction with mitochondrial respiratory chain, were synthesized [5].
Investigation of the adaptive properties of antioxidant-charged Skulachev ions imparting this geroprotective activity became the task of the largest Russian biotechnological project [5].
The goal of investigations carried out within the frame of this project in the Research Institute of Biology of the Southern Federal University was studying the ability of the most active compound of this group, 10-(6′-plastoquinonyl) decyltriphenylphosphonium (SkQ1), to modify processes of spontaneous and induced mutagenesis with following biochemical investigation of the revealed mechanisms of the phenomenon. Oxygen under pressure (experiments on animals) and UV radiation at 300-400 nm characteristic of sunlight near the Earth’s surface (experiments on bacteria) were used as mutagens.
MATERIALS AND METHODS
Forty-five-day-old outbred male Wistar rats obtained from the vivarium of a branch of the Shemyakin–Ovchinnikov Institute of Bioorganic Chemistry (Russian Academy of Sciences) were injected daily for 14 days with SkQ1 (25 and 250 nmol/kg). The calculated dose dissolved in 100 µl 0.2% ethanol solution in distilled water was injected into cheek pouches of the animals. Animals of the control groups obtained 100 µl 0.2% ethanol daily for 14 days. Some rats underwent hyperbaric oxygenation (HBO) after the course of injections was finished. Each animal was placed in a separate 60 liter pressure chamber supplied with a carbon dioxide regeneration system. After pure medical oxygen flow through for 3 min, pressure in the chamber was adjusted to 0.5 MPa. Compression and decompression were carried out at the rate of 0.2 MPa/min. The animals were kept under this pressure for 60 min. Pressure chambers were purged with oxygen flow for 2 min at 15 min intervals. Oxygen-treated animals were sacrificed in 24 h after treatment and the last introduction of SkQ1.
Cornea squash preparations for cytogenetic investigations were obtained using standard technique with orcein staining [7]. Ready preparations were randomized and studied further by a “blind” method. In cornea preparations anaphase chromosome aberration frequency was analyzed during which chromosome and chromatid fragments as well as chromosome and chromatid bridges were taken into account. Mitotic index was calculated as percentage of dividing cells in the total number of analyzed cells that was no less than 3000 for each eye. Altogether, six groups of 10 animals each were useed for cytogenetic studies (Table 1).
Table 1. Modification of spontaneous and
HBO-induced clastogenesis in rat corneal epitheliocytes by SkQ1
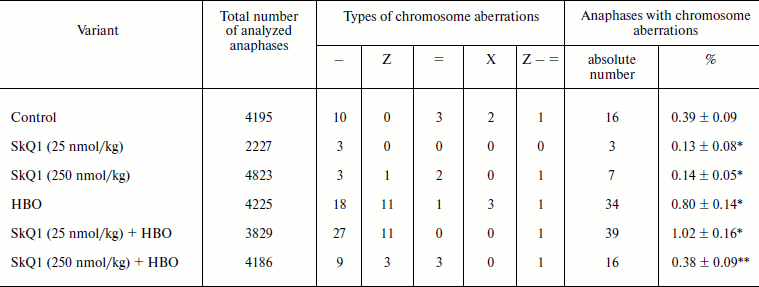
Notes: Types of chromosome aberrations: –, chromatid fragment;
Z, chromatid bridge; =, paired acentric fragment; X, chromosome
bridge; Z–=, multiple rearrangements.
* Statistically significant differences compared to control, p
< 0.05.
** Statistically significant differences compared to HBO, p
< 0.05.
Animals with characteristics resembling those described above were used for biochemical experiments. SkQ1 was introduced according to the scheme above described. The rats were decapitated in 60-90 min after the last SkQ1 injection. The blood serum was separated from the erythrocyte mass by centrifugation at 2000g. Serum specimens were randomized and then studied by the “blind” method. Extracellular 8-OH-2′-deoxyguanosine (8-OH-dG) was studied in the blood serum by immunoenzyme analysis using a test system for its determination in the blood, saliva, and urine – DNA Damage ELISA Kit No. EKS-350 (Biochem Acquires Assay Designs, USA; www.assaydesigns.com) on an Alisei automatic immunoenzyme analyzer (Italy).
For evaluation of SkQ1 protective effect against genotoxic action of UV radiation in Escherichia coli, the SOS-lux test (used earlier for investigation of protective effect of a number of synthetic and natural compounds against UV radiation) was used. The Lux operon served as the SOS-response reporter. The E. coli strain C600 transformed by plasmid pPLS-1 in which the bioluminescence operon is under control of SOS promoter C600 (pPLS-1) was used. The experimental protocol is described in detail elsewhere [8]. The SOS response induction factor (Is) was calculated using the formula: Is = Le/Lc – 1, where Lc and Le is luminescence intensity in control and experiment, respectively. The antimutagenic potential index (A, %) was calculated using the formula: A = (1 – Ia/Ip)·100%, where Ia and Ip is factor of SOS response induction by the effect under study in the absence and presence of protector, respectively [8].
The SkQ1 preparation was adjusted to necessary concentration with ethanol. The equivalent amount of ethanol was added to control samples. The data were statistically processed using standard formulas, and standard error of the mean was used as criterion of data scattering for all experiments [9].
RESULTS
The experimental results showed that the two-week course of SkQ1 introduction into male Wistar rats at doses of 25 and 250 nmol/kg statistically significantly decreases the level of chromosome aberrations in corneal epitheliocytes almost three-fold from 0.37 to 0.13 and 0.14%, respectively. Most of chromosome rearrangements (60%) were chromatid fragments (Table 1). On the whole, 88% of all chromosome aberrations were due to DNA fragmentation.
An increased level of chromosome aberrations, more than double compared to control, was registered in the corneal epitheliocytes of the animals in response to the HBO regime of 0.5 MPa for 60 min. In this case the increase in number of both chromatid fragments and chromatid bridges was observed. No statistically significant inhibition of HBO-induced mutagenesis in response to introduction of the lower SkQ1 dose (25 nmol/kg) was found. The higher dose of SkQ1 (250 nmol/kg) decreased to control level the frequency of chromosome aberrations induced by the oxidative stress.
Proliferative activity of corneal epithelium determined by mitotic index was in control 1.6 ± 0.17%. The HBO treatment did not cause statistically significant changes in proliferative activity, the mitotic index being 1.8 ± 0.14%. In the case of introduction of 25 nmol/kg SkQ1 mitotic index did not differ from control (1.5 ± 0.12%). In the case of the higher dose a significant (t-criterion, p < 0.05) increase in mitotic index to 2.7% (1.68-fold) was registered. The HBO treatment against the background of the lower SkQ1 dose did not cause statistically significant changes in this index. After HBO treatment of animals that obtained the higher SkQ1 dose, a statistically significant decrease in the mitotic cell pool to 1.2 ± 0.1%, constituting 75% of intact control, was registered. According to contemporary concepts, such deviations are within the limits of the physiological norm [7].
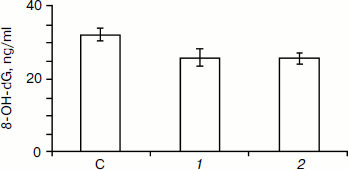
The results of 8-OH-dG determination in blood serum of the studied animals revealed that the content of this substance in control was 32.12 ± 1.55 ng/ml. Injection of SkQ1 dose of 25 nmol/kg decreased this value to 25.90 ± 2.26 ng/ml, while the higher dose of SkQ1 (250 nmol/kg) decreased it to 25.76 ± 1.50 ng/ml.
Data on the protective effect of SkQ1 against UV irradiation of E. coli are given in Table 2. SkQ1 at 1 nM concentration caused practically complete inhibition of the genotoxic effect of UV radiation. SkQ1 at 0.1 nM concentration did not exhibit any observable protective effect.
Table 2. SOS-response induction in E.
coli by ultraviolet radiation at 300-400 nm at dose 390
J/m2 in the presence of SkQ1
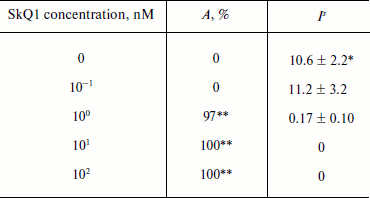
* Mean value and its standard error for induction factor in all
experiments on bacteria were calculated on the basis of results of
three independent experiments.
** Statistically significant protective effect, p <
0.05.
DISCUSSION
A number of facts in favor of a relationship between mutagenesis and aging have been accumulated. First, genetic anomalies accumulate with age [10, 11]. Second, progeric effect is observed for practically all cases disturbing activity of the genetic apparatus, while its extent depends on the severity of damage [12]. Third, progeric effect is characteristic of mutations associated with defects in DNA repair [13, 14]. The description of the action of any potential geroprotector without accounting for its effect on mutagenesis would be incomplete. Ageing of multicellular organisms is accompanied and possibly provided by progressing decrease in cell number. Owing to this, in our study of the effect of SkQ1 on spontaneous mutagenesis, we followed formal logic and paid attention to the most cell-destructive genetic anomalies – chromosome aberrations revealed in the anaphase test. Both studied SkQ1 doses decreased the spontaneous level of such events. This phenomenon can be stimulated by different factors. Chromosome aberrations are generated by tens dozens of enzymes using the energy of ATP [15]. Therefore, in principle, the decrease in their frequency is an indication of inhibition of metabolism. Data on proliferation suggest that the contribution of this mechanism is insignificant. No dose inhibiting spontaneous mutagenesis decreases mitotic index, i.e. it does not inhibit cell division.
“Positive” factors inhibiting spontaneous mutagenesis can be divided into two groups. The first group is associated with changes in adjustment of repair mechanisms. The transformation of DNA damage to morphologically distinguishable chromosome anomaly is mediated by the mismatching of repair activities. Therefore, the decrease in aberration frequency can be associated with activation of appropriate repair. Another group of factors is associated with lowering the level of primary damage to DNA. The ability of products of one-electron reduction of oxygen and lipid peroxidation to induce lesions resulting in formation of chromosome aberrations has been found [16]. These substances are continuously generated by mitochondria. It is logical to suppose that the preparation under study decreases the background generation of mitochondrial ROS [5, 6, 17] and thus decreases the content of endogenous DNA-damaging compounds.
Both studied doses cause statistically significant decrease in 8-OH-dG content in blood serum (see figure). Note that the repair of oxidative DNA damage involves excision of 8-OH-dG [18, 19]. Therefore, if SkQ1 worked as a repair activator we would observe the reverse effect.
Experiments on the modification of induced mutagenesis by SkQ1 were carried out to search for adaptogenic properties of this preparation not directly associated with geroprotector activity.Thus, it can be supposed that the main mechanism of suppression by SkQ1 of the background level of chromosome aberrations is lowering the level of generation of genotoxic products of ROS interaction with biomolecules.
Hyperbaric oxygenation is rather widely used in clinical practice. Many healthy people undergo oxygen pressure when diving. In this case the partial pressure of oxygen approaches the level of therapeutic HBO regimes. Mutagenic effect in this case is described in a number of works [20-22]. As is shown by our studies, Skulachev ions with antioxidant charge can be used for protection against oxygen-induced mutagenesis.
Ultraviolet radiation at 300-400 nm, specific of sunlight near the Earth’s surface, causes DNA damage due to ROS generation [23]. As shown by our experiments, SkQ1 at 1 nM concentration practically completely inhibits the genotoxicity of this kind of radiation for E. coli. No studied natural or synthetic antioxidant exhibited an equivalent effect in a similar system [8, 24]. Minimal effective dose for the thiazine dye methylene blue, that showed the best result, was 10 nM. It is interesting that this compound, like SkQ1, is a lipophilic cation capable of in vivo formation of an oxidation/reduction cycle [25]. Thus, the UV-protector properties of Skulachev ions with antioxidant charge can be also of practical interest.
This work was supported by the Research Mitoengineering Institute (Moscow State University) and the Russian Federation Ministry of Science and Education (grant “Development of Higher School Scientific Potential for 2009-2010” No. 2.1.1/5628).
REFERENCES
1.Harman, D. (1956) J. Gerontol.,
11, 298-300.
2.Skulachev, V. P. (2005) IUBMB Life,
57, 305-310.
3.Manskikh, V. N. (2007) Tsitologiya,
49, 909-915.
4.Skulachev, V. P. (1999) Biochemistry
(Moscow), 64, 1418-1426.
5.Skulachev, V. P. (2007) Biochemistry
(Moscow), 72, 1385-1396.
6.Skulachev, V. P. (2009) Ross. Khim. Zh.,
53, 125-140.
7.Darlington, S. D., and La Cure (1980)
Chromosomes. Methods of Investigation [Russian translation],
Atomizdat, Moscow.
8.Chistyakov, V. A., Sazykina, M. A., Kolenko, M. A.,
Chervyakov, G. G., and Usatov, A. V. (2009) Genetika, 45,
304-307.
9.Glants, S. (1998) Medico-Biological
Statistics [in Russian], Praktika, Moscow.
10.Dolle, M. E. T., Snyder, W. K., Gossen, J. A.,
Lohman, P. H. M., and Vijg, J. (2000) Proc. Natl. Acad. Sci.
USA, 97, 8403-8408.
11.Dolle, M. E. T., Snyder, W. K., Dunson, D. B.,
and Vijg, J. (2002) Nucleic Acids Res., 30, 545-549.
12.Grillari, J., Katinger, H., and Voglauer, R.
(2007) Nucleic Acids Res., 35, 7566-7576.
13.Vogel, H., Lim, D.-S., Karsenty, G., Finegold,
M., and Hasty, P. (1999) Proc. Natl. Acad. Sci. USA, 96,
10770-10775.
14.Weeda, G., Donker, I., de Wit, J., and Morreau,
H. (1997) Curr. Biol., 7, 427-439.
15.Shiloh, Y. (2003) Nature Rev. Cancer,
3, 155-168.
16.Marnett, L. J. (2000) Carcinogenesis,
21, 361-370.
17.Antonenko, Yu. N., Avetisyan, A. V., Bakeeva, L.
E., Chernyak, B. V., Chertkov, V. A., Domnina, L. V., Ivanova, O. Yu.,
Izyumov, D. S., Khailova, L. S., Klishin, S. S., Korshunova, G. A.,
Lyamzaev, K. G., Muntyan, M. S., Nepryakhina, O. K., Pashkovskaya, A.
A., Pletjushkina, O. Yu., Pustovidko, A. V., Roginsky, V. A.,
Rokitskaya, T. I., Ruuge, E. K., Saprunova, V. B., Severina, I. I.,
Simonyan, R. A., Skulachev, I. V., Skulachev, M. V., Sumbatyan, N. V.,
Sviryaeva, I. V., Tashlitsky, V. N., Vasiliev, Yu. M., Vyssokikh, M.
Yu., Yaguzhinsky, L. S., Zamyatnin, A. A., Jr., and Skulachev, V. P.
(2008) Biochemistry (Moscow), 73, 1273-1287.
18.Imlay, J. A. (2008) Annu. Rev. Biochem.,
77, 4.1-4.22.
19.Busuttil, R. A., Garcia, A. M., Reddick, R. L.,
Dolle, M. E., Calder, R. B., Nelson, J. F., and Vijg, J. (2007) PLoS
ONE, 12, e876.
20.Guskov, E. P., and Shkurat, T. P. (1985)
Genetika, 21, 1361-1367.
21.Guskov, E. P., Shkurat, T. P., and Kamynina, M.
V. (1985) Genetika, 21, 1693-1699.
22.Guskov, E. P., Shkurat, T. P., Shimanskaja, E.
I., and Guskova, S. I. (1990) Mutat. Res., 241,
341-347.
23.Gomes, A. A., Asad, L. M. B. O., Felzenszwalb,
I., Leitјo, A. C., Silva, A. B., Guillobel, H. C. R., and Asad,
N. R. (2004) Radiat. Environ. Biophys., 43, 219-222.
24.Sazykina, M. A., Chistyakov, V. A., Kolenko, M.
A., and Azarin, K. V. (2009) Ekol. Genet., 7,
44-46.
25.Atamna, H., Nguyen, A., Schultz, C., Boyle, K.,
Newberry, J., Kato, H., and Ames, B. N. (2008) The FASEB J.,
22, 703-712.