Mitochondria-Targeted Plastoquinone Derivative SkQ1 Increases Early Reproduction of Drosophila melanogaster at the Cost of Early Survival
E. A. Tsybul’ko, N. V. Roshina, O. Yu. Rybina, and E. G. Pasyukova*
Institute of Molecular Genetics, Russian Academy of Sciences, pl. Kurchatova 2, 123182 Moscow, Russia; fax: (499) 196-1909; E-mail: egpas@rambler.ru* To whom correspondence should be addressed.
Received November 24, 2009
It was previously found that 20 pM SkQ1 (10-(6′-plastoquinonyl) decyltriphenylphosphonium) solution increased lifespan of virgin females and males of the fruit fly Drosophila melanogaster with maximal and highly reproducible effect on early survival of females. In this paper we demonstrate that SkQ1 solution of the same concentration does not increase lifespan of mated females and males, early effect on female survival being absent, whereas early fertility and the total number of progeny are elevated in treated flies. Increase in fertility observed in young mated females instead of increase of survival typical for young virgin females might illustrate the trade-off between the fly’s lifespan and reproduction.
KEY WORDS: SkQ1, antioxidant, lifespan, reproductionDOI: 10.1134/S0006297910030016
Correlations between oxidative damage and aging are well documented for Drosophila. For example, flies with altered expression of genes affecting reactive oxygen species production [1-3] and detoxification [4, 5] or with genetic alterations associated with modified mitochondrial genome [6] or structure [7] have modified lifespan. However, there are some results that cause doubts on the universal role of oxidative damage in regulation of normal aging [8-11]. In particular, external oxidative and antioxidative agents affect lifespan, but not always [12-14]. As it is still impossible to decide whether the oxidative stress hypothesis of aging is true, accumulation of new data elucidating this problem holds great value.
Discrepancies between different experimental data can be attributed to the effects of interaction of oxidative stress with other factors affecting lifespan. For example, reproduction status can affect survival [15, 16], and a complex interplay between reproduction and lifespan can interfere with effects of other factors, particularly oxidative stress, on lifespan.
It was previously found that the antioxidant SkQ1 (10-(6′-plastoquinonyl) decyltriphenylphosphonium) at extremely low concentrations increased lifespan of fungi (Podospora anserina), crustaceans (Ceriodaphnia affinis), insects (Drosophila melanogaster), and mammals (mouse) [17]. SkQ1 was effective both in virgin females and males, with maximal and highly reproducible effect on early survival of females. In this paper we demonstrate that SkQ1 solution of the same 20 pM concentration does not increase lifespan of mated D. melanogaster females and males, early effect on female survival being absent, whereas early fertility and the total number of progeny are elevated in treated flies.
MATERIALS AND METHODS
The isogenic w1118 Drosophila melanogaster line was used in the experiments. The flies were maintained on a wheat/sugar/raisin/agar medium at 25°C.
Fresh 20 mM stock solution of SkQ1 in 96% ethanol was prepared for each trial and stored at –20°C. Working 20 pM solution of SkQ in distilled water was prepared ex tempore. As a control, 96% ethanol diluted 109 times in distilled water was used (ethanol concentration in the SkQ1 working solution). In each case, 0.1 ml of the solution was applied to the medium surface and dried overnight at 25°C.
Three virgin females and three males collected from parental vials with controlled density during one day were placed together in a vial containing culture medium with appropriate solution. Flies were transferred to fresh medium with appropriate solution once in three days. Dead flies were registered every day. Progeny was scored in each vial during 10 days.
Sample size of one experimental trail was equal to 60 females and 60 males. In total, lifespan of 180 females and 180 males was scored. Data for males and females were analyzed separately. To compare lifespan of treated and control flies, two-way analysis of variance with trial and treatment as the main fixed effects and Student’s t-test were used. To compare survival curves, the Kaplan–Meier logrank test was performed using GraphPad Prism Version 4.00 for Windows (GraphPad Software, San Diego, California, USA, www.graphpad.com). To compare numbers of progeny of treated and control flies, Student’s t-test was used.
RESULTS AND DISCUSSION
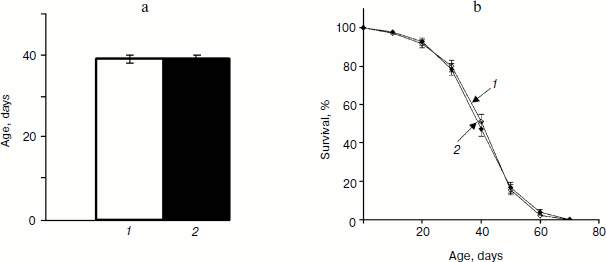
Analysis of variance did not reveal significant variation between trials within each treatment for mated males, which allowed combining trials for further analysis. Treatment term also was not significant (P = 0.8899) indicating that SkQ1 in concentrations 20 pM did not affect lifespan of mated males. Comparison of mean values of male lifespan (treated, 39 ± 1 days; control, 39 ± 1 days, P = 0.8626) and male survival curves (P = 0.9478) confirmed this result (Fig. 1).
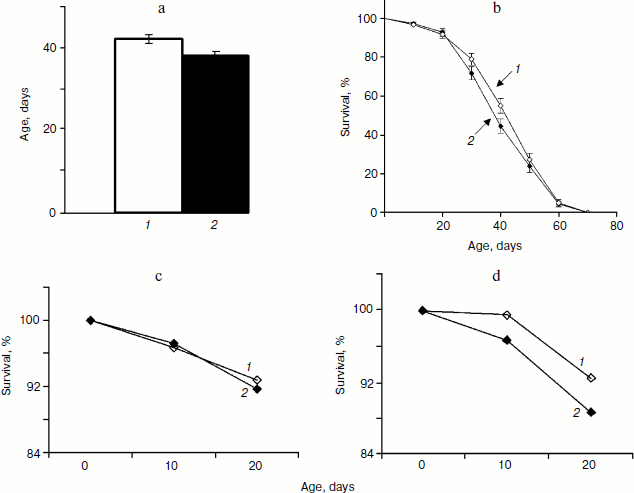
Analysis of variance did not reveal significant variation between trials within each treatment for mated females, which allowed combining trials for further analysis. Treatment term also was not significant (P = 0.1209) indicating that SkQ1 in concentrations 20 pM did not affect lifespan of mated females. Comparison of mean values of female lifespan (treated, 42 ± 3 days; control, 38 ± 1 days, P = 0.0868) and female survival curves (P = 0.1721) confirmed this result (Fig. 2, a and b). The most pronounced effect of SkQ1 on lifespan of virgin females consists in substantial increase of early survival [17]. This effect lays the groundwork for the total effect of SkQ1 on lifespan of virgin females. Such an effect is completely absent when lifespan of mated females is assayed (compare Figs. 2c and 2d).Fig. 1. a) Mean lifespan of treated (1) and control males (2). b) Survival curves of treated (1) and control males (2) (180 males per treatment were analyzed).
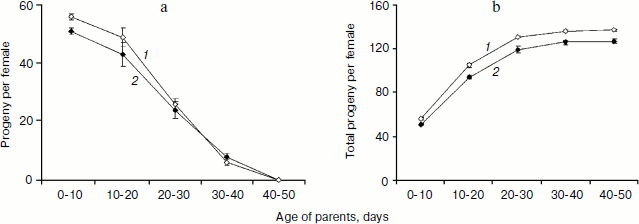
In total, three trials produced 23,011 and 20,855 flies in the progeny of treated and control parents, correspondingly. Taking into consideration reduction of the number of parents during the course of the experiments, the number of progeny per female and the accumulated (starting from the first day) number of progeny per female was calculated for 10 day intervals (Fig. 3). Significantly higher number of progeny was registered for 10-day-old treated parent as compared with control (P = 0.0307). This difference became insignificant at 20 days and disappeared completely later in life (Fig. 3a). However, the early effect of SkQ1 on reproduction was sufficient to provide significant increase in the number of progeny of treated flies during their entire life (P = 0.0161, 0.0143, 0.0142, and 0.0198 for 20, 30, 40, and 50 days, respectively) (Fig. 3b).Fig. 2. a) Mean lifespan of treated (1) and control females (2). b) Survival curves of treated (1) and control females (2). c) Survival curves of treated (1) and control (2) females early in life. d) Survival curves of treated (1) and control (2) virgin females early in life [17] (180 females per treatment were analyzed).
Detailed analysis of the SkQ1 effect on lifespan of virgin females revealed that it is much more pronounced early in life. Mortality during the first 10 days of life was almost completely abolished by SkQ1, whereas later in life the effect of SkQ1 treatment became less marked and disappeared completely in old females [17]. This effect is paralleled by the effect of SkQ1 on locomotor activity of virgin females. Significant increase in locomotion was observed in 10-day-old SkQ1 treated virgin females as compared with controls. After 10 days, the difference between treated and control flies was conserved at the same level and finally disappeared in old females (E. G. Pasyukova, unpublished results). In this paper we demonstrate that SkQ1 did not affect early survival of mated Drosophila females, but early reproduction appeared to be increased in treated flies. Altogether, these results indicate that SkQ1 can specifically affect different traits at young ages, probably because Drosophila is a post-mitotic organism. Alternatively, SkQ1 effect on lifespan, locomotion, and reproduction predominantly at young ages might reflect some basic characteristics of this antioxidant and mechanisms of its interaction with any organism. Indeed, the same tendency of predominant early-in-life SkQ1 activity was observed for other animals [17]. Taking into consideration (i) a complex action of SkQ on different traits including lifespan, locomotion, and reproduction and (ii) its activity primarily observed in young flies, it is possible to suggest that SkQ1 could increase the quality of life rather than to prolong lifespan.Fig. 3. a) Fertility of treated (1) and control females (2). b) Total progeny of treated (1) and control females (2). Differences between vials were used to calculate standard errors.
Increase in reproduction ability observed in young mated females instead of increase of survival typical for young virgin females might illustrate the trade-off between lifespan and reproduction. Our experiments do not provide any explanation why this negative correlation is observed in the case of SkQ1 treatment. It was shown that the female survival cost of mating is not associated with elevated feeding observed in females following mating [18]. Anyway, it is clear that SkQ1 effect on lifespan is not related to dietary composition and, at least for mice, to caloric restriction [17]. It is also evident that egg production (fecundity) and lifespan do not always show a trade-off [19, 20]. Of all the traits, frequency of mating is significantly associated with the extent of the female survival cost of mating [18]. It remains to be assessed experimentally whether mating frequency is affected by SkQ1 treatment. We know that locomotion is affected by SkQ1, and a reasonable speculation would be that fly activity in general is raised due to SkQ1 treatment. Mating frequency could be a part of this general effect. Another possibility is that SkQ1 effect on reproduction is more specific and based on interaction with the metabolism of sex peptides. These male seminal fluid proteins can profoundly change female gene expression and physiology, including egg production and frequency of mating [21].
Increased reproduction ability of SkQ1 treated flies can also be explained by better survival of the embryos, larvae, and pupa in their progeny because they developed in vials where SkQ1 was added. This explanation does not involve trade-off between reproduction and lifespan; rather, it predicts that SkQ1 affects viability at least at one of the developmental stages of Drosophila, and this prediction can also be evaluated experimentally.
The authors are grateful to V. P. Skulachev, V. N. Anisimov, L. S. Yaguzhinsky, I. I. Severina, B. V. Chernyak, N. G. Kolosova, F. F. Severin, M. Y. Vyssokikh, V. N. Tashlitsky, M. V. Skulachev, and other colleagues for helpful discussions.
This work was supported by the Research Institute of Mitoengineering of M. V. Lomonosov Moscow State University.
REFERENCES
1.Walker, D. W., Muffat, J., and Benzer, S. (2006)
Curr. Biol., 16, 674-679.
2.Copeland, J. M., Cho, J., Lo, T., Jr., Hur, J. H.,
Bahadorani, S., Arabyan, T., Rabie, J., Soh, J., and Walker, D. W.
(2009) Curr. Biol., 19, 1591-1598.
3.Zid, B. M., Rofers, A. N., Katewa, S. D., Vargas,
M. A., Kolipinski, M. C., Lu, T. A., Benzer, S., and Kapahi, P. (2009)
Cell, 139, 149-160.
4.Parkes, T. L., Hilliker, A. J., and Phillips, J. P.
(1999) Neurobiol. Aging, 20, 531-535.
5.Sun, J., Folk, D., Bradley, T. J., and Tower, J.
(2002) Genetics, 161, 661-672.
6.Clancy, D. J. (2008) Aging Cell, 7,
795-804.
7.Walker, D. W., and Benzer, S. (2004) Proc. Natl.
Acad. Sci. USA, 101, 10290-10295.
8.Mockett, R. J., Orr, W. C., Rahmandar, J. J.,
Benes, J. J., Radyuk, S. N., Klichko, V. I., and Sohal, R. S. (1999)
Arch. Biochem. Biophys., 371, 260-269.
9.Mockett, R. J., Bayne, A. C., Kwong, L. K., Orr, W.
C., and Sohal, R. S. (2003) Free Radic. Biol. Med., 34,
207-217.
10.Orr, W., Mockett, R. J., Benes, J. J., and Sohal,
R. S. (2003) J. Biol. Chem., 278, 26418-26422.
11.Perez, V. I., Bokov, A., Rammen, H. V., Mele, J.,
Ran, Q., Ikeno, Y., and Richardson, A. (2009) Biochim. Biophys.
Acta, 1790, 1005-1014.
12.Anisimov, V. N., Mylnikov, S. V., and Khavinson,
V. K. (1998) Mech. Ageing Dev., 103, 123-132.
13.Izmailov, D. M., and Obukhova, L. K. (1999)
Mech. Ageing Dev., 106, 233-240.
14.Magwere, T., West, M., Riyahi, K., Murphy, M. P.,
Smoth, R. A. J., and Partrige, L. (2006) Mech. Ageing Dev.,
127, 356-370.
15.Sgro, C. M., and Partridge, L. (1999)
Science, 286, 2521-2524.
16.Partridge, L., Gems, D., and Withers, D. J.
(2005) Cell, 120, 461-472.
17.Anisimov, V. N., Bakeeva, L. E., Egormin, P. A.,
Filenko, O. F., Isakova, E. F., Manskikh, V. N., Mikhelson, V. M.,
Panteleeva, A. A., Pasyukova, E. G., Pilipenko, D. I., Piskunova, T.
S., Popovich, I. G., Roshchina, N. V., Rybina, O. Y., Saprunova, V. B.,
Samoylova, T. A., Semenchenko, A. V., Skulachev, M. V., Spivak, I. M.,
Tsybul’ko, E. A., Tyndyk, M. L., Vyssokikh, M. Y., Yurova, M. N.,
Zabezhinsky, M. A., and Skulachev, V. P. (2008) Biochemistry
(Moscow), 73, 1329-1342.
18.Barnes, A. I., Wigby, S., Boone, J. M.,
Partridge, L., and Chapman, T. (2008) Proc. Roy. Soc. B,
275, 1675-1683.
19.Hwangbo, D. S., Gersham, B., Tu, M. P.,
Palmer, M, and Tatar, M. (2004) Nature, 429,
562-566.
20.Mair, W., Sgro, C. M., Johnson, A. P., Chapman,
T., and Partridge, L. (2004) Exp. Gerontol., 30,
1011-1019.
21.Wolfner, M. F. (1997) Insect Biochem. Mol.
Biol., 27, 179-192.